The "Cardiovascular Answer" of Small Molecule GLP-1: Eli Lilly's Orforglipron Gets the Ticket, But the Real War Has Just Begun
On April 16, 2026, just 15 days after Eli Lilly's Orforglipron (brand name Foundayo) was approved by the FDA for obesity indications as the "world's first oral small-molecule GLP-1 receptor agonist," another set of data subtly shifted the balance of this victory.
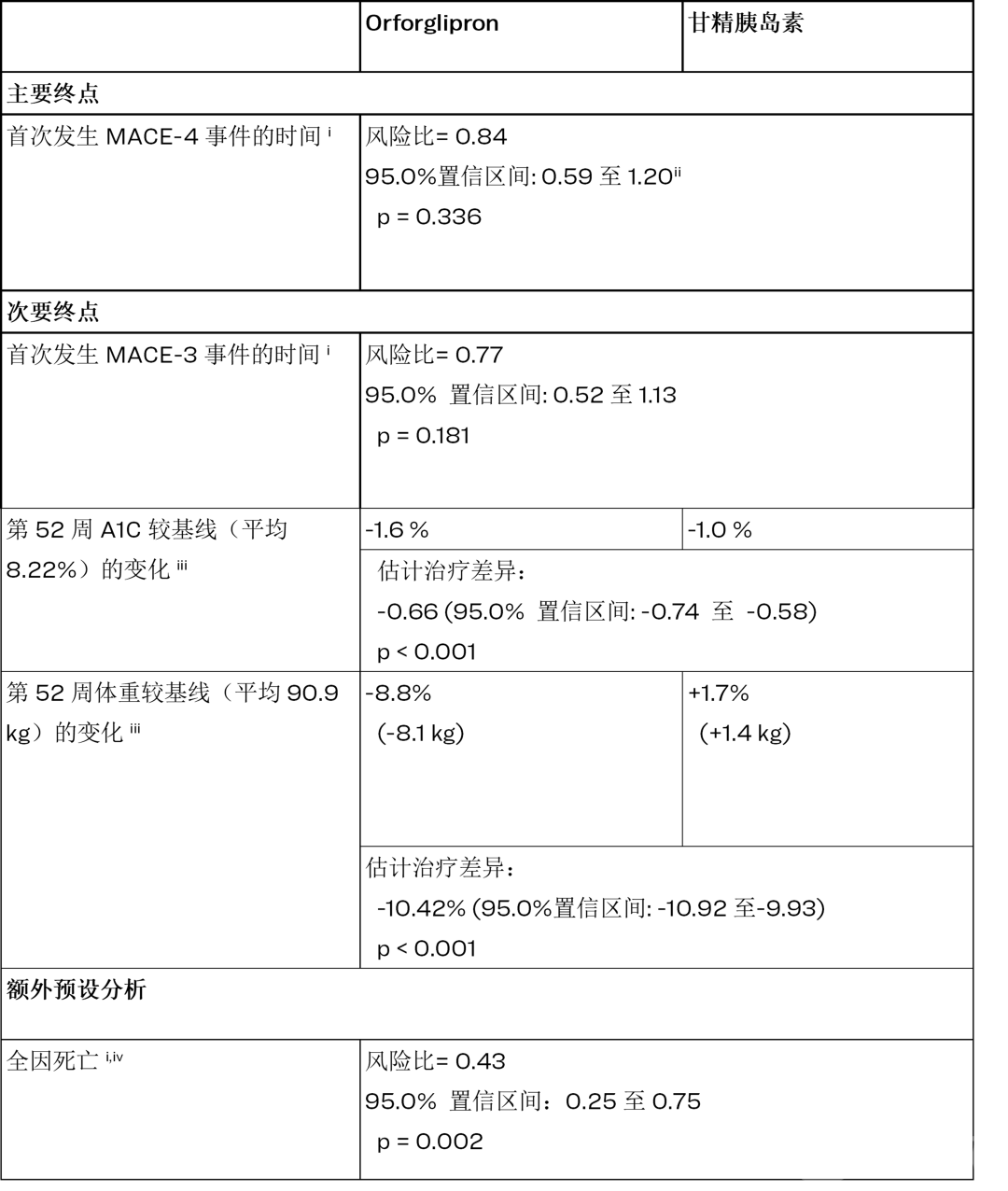
ACHIEVE-4 Trial Results Announced: In Patients with Type 2 Diabetes and Overweight/Obesity, Elevated Cardiovascular Risk, Foundayo Showed a 16% Reduction in Primary Endpoint MACE-4 Risk Compared to Insulin Glargine (Hazard Ratio=0.84, 95% Confidence Interval: 0.59-1.20, p=0.336), Meeting Non-Inferiority Criteria. More Remarkably, All-Cause Mortality — Risk Reduced by 57% (Hazard Ratio=0.43, 95% Confidence Interval: 0.25-0.75, p=0.002) — Becoming the Only Secondary Endpoint in the Trial to Reach Statistical Significance.
ACHIEVE-4 Topline Results
At the moment of the data release, the entire metabolic disease field fell silent. Because everyone knew that behind this number lay an unanswered question: Can oral small-molecule GLP-1 replicate the story already proven by injectables in cardiovascular outcomes?
Lilly gave a "yes, but not loud enough" answer.
In the GLP-1 market dominated by Novo Nordisk's injectable Semaglutide (Wegovy/Ozempic), Eli Lilly has chosen a seemingly more challenging path — developing small molecules.
This is not a "compromise solution," but rather a structural judgment.
The core pain point of injectable GLP-1 has never been efficacy, but accessibility. Cold chain transportation, cold chain storage, and once-a-week injections—these barriers are clinical convenience issues at the prescription end, capacity and supply bottlenecks at the payment end, and compliance challenges for patients. If small-molecule oral formulations can approach efficacy and surpass injectables in dosing convenience, they can unlock incremental markets in two dimensions: capturing patients with a stronger preference for oral administration from the existing injectable user base, and activating the "needle-phobic" population that GLP-1 has never reached.
Orforglipron is not an "oral modification" of peptide GLP-1 but a de novo designed non-peptide small molecule that activates the receptor by binding to a non-natural ligand-binding pocket of the GLP-1 receptor.
Lilly has built a global Phase III project matrix centered on Orforglipron: the ATTAIN series covers obesity/overweight indications, and the ACHIEVE series covers Type 2 diabetes indications. By 2026, Orforglipron has become the oral metabolic drug closest to commercialization in Lilly's pipeline.
Lilly Defined ACHIEVE-4 in the Official Press Release as"Foundayo Study: The Largest and Longest Study on Type 2 Diabetes to Date", recruiting over 2,700 participants across 15 countries. This statement on the scale of the trial reflects Lilly's high regard for cardiovascular data — it is not only a safety report but also a key asset in securing T2DM indication approval.
If the赛道 is defined as "oral GLP-1 receptor agonists," there are currently not many products worldwide that have truly entered the late clinical stage or have been launched, but the competition density is rapidly increasing.
Let's first look at the two listed products:
Foundayo (Orforglipron) / Eli Lilly, a non-peptide small molecule GLP-1RA, administered orally once daily. In the ATTAIN-1 trial for obesity, the 36mg dose demonstrated a 12.4% weight loss over 72 weeks (11.5% after placebo adjustment), with approximately 60% of participants in the 36mg group achieving ≥10% weight loss and about 40% achieving ≥15% weight loss. In terms of cardiovascular outcomes, the ACHIEVE-4 trial met non-inferiority criteria for MACE-4, with a 57% reduction in all-cause mortality risk (p=0.002). The company plans to submit a T2DM indication application to the FDA by the end of Q2 2026.
Wegovy Oral Pills (Semaglutide Oral Formulation) / Novo Nordisk, an oral formulation of the peptide GLP-1RA class, administered orally once daily. The OASIS 4 trial demonstrated a 16.6% weight loss over 72 weeks, and the SELECT trial confirmed a 20% reduction in MACE, with cardiovascular benefits clinically validated. Approved for weight loss indications by the end of 2025, the number of weekly prescriptions reached approximately 50,000 in the third week post-launch, of which about 45,000 were self-paid.
The key differences between the two lie in three dimensions:
• Efficacy Depth: The highest weight loss achieved with oral Wegovy is 16.6%, and with Foundayo it is 12.4%, a difference of approximately 4 percentage points. However, at the 36mg dose, about 40% of patients on Foundayo achieved ≥15% weight loss, making the "responder proportion" gap smaller than the absolute mean difference compared to oral Wegovy.
• Cardiovascular Evidence: The SELECT trial of Wegovy injection has demonstrated a 20% reduction in MACE with statistical significance; while the ACHIEVE-4 trial of Foundayo achieved non-inferiority for MACE-4 (p=0.336, did not reach superiority), the 57% reduction in all-cause mortality (p=0.002) presented a highly significant signal.
• Business Rhythm: Wegovy oral is expected to be approved by the end of 2025, Foundayo in April 2026, and Lilly about four months later. However, Foundayo may progress faster in the T2DM indication — the ACHIEVE-4 data provides direct support for the registration of the T2DM indication.
Next, let's look at the key players in the research pipeline:
Pfizer's Danuglipron (PF-06882961), a small-molecule GLP-1RA, is taken orally twice daily (a sustained-release formulation is under development). Phase II data showed a placebo-adjusted weight loss of approximately 5-9% over 16 weeks, and the Phase III program is currently ongoing.
Roche's Carmot Acquired Pipeline Includes Multiple Oral GLP-1 Candidates, Some in Phase Ⅰ/Ⅱ, Focusing on Peptide Optimization.
Chinese companies such as Huadong Medicine and Hengrui Medicine are also laying out plans for oral GLP-1, but most are in the early clinical stages and will not pose direct competition in the short term.
A Often-Overlooked Detail: Oral Administration ≠ Automatic Improvement in Adherence. Wegovy oral formulation requires fasting before administration, and no food intake is allowed for at least 30 minutes after taking the medication — this "medication window" may have a greater impact on adherence than the difference between "injection vs. oral" formulations. Whether Foundayo’s once-daily dosing will require similar fasting conditions will be a key variable in real-world use. Dr. Thomas Seck, Senior Vice President of Cardiovascular Metabolic Health Research and Development at Eli Lilly, stated in a press release: “Across seven Phase 3 studies involving over 11,000 participants, orforglipron has demonstrated consistent safety and efficacy. ACHIEVE-4 adds a new dimension to this evidence — cardiovascular safety, as well as a reduction in all-cause mortality risk observed in patients with elevated cardiovascular risk. Combined with the convenience of once-daily dosing without dietary or hydration restrictions, we believe orforglipron has the potential to become an important new treatment option for patients with type 2 diabetes.” The description of medication convenience without dietary or hydration restrictions suggests that there are virtually no specific requirements for taking the medication.
Back to Orforglipron itself. Its story can be summarized in three key milestones.
Node 1: ATTAIN-1 "Dose-Response" Curve
In August 2025, the results of the ATTAIN-1 trial were announced.
This is a randomized, double-blind, placebo-controlled Phase III trial enrolling overweight/obese adult subjects with a baseline weight of 103.2 kg, followed up for 72 weeks.
The results showed a clear dose-response relationship stratified by dose: weight change was -0.9% in the placebo group, -7.8% in the 6mg group, -9.3% in the 12mg group, and -12.4% in the 36mg group. The placebo-adjusted weight loss in the 36mg high-dose group was 11.5%.
More clinically significant is a set of "threshold data": about 60% of participants in the 36mg group achieved ≥10% weight loss, and about 40% achieved ≥15% weight loss. These two figures hold special meaning in the field of metabolic diseases—10% weight loss is considered by multiple guidelines as the threshold for "clinically significant benefit," while 15% approaches the lower limit of effectiveness for certain bariatric surgeries.
"Lilly wrote in the press release: 'Orforglipron represents the first oral GLP-1 receptor agonist to demonstrate clinically meaningful weight loss in a Phase 3 trial.'"
The wording of this sentence is very precise — "clinically meaningful weight loss" rather than "optimal weight loss." Lilly has not claimed to have the best weight loss effect, but instead emphasized the叠加 of the two qualifiers "oral administration" and "clinical significance." This is a deliberate market positioning strategy: when efficacy is not absolutely the first priority, the uniqueness of the route of administration is highlighted.
Node 2: FDA Approval, but Pricing Strategy Leaves Suspense
On April 1, 2026, the FDA approved Foundayo for the obesity indication.
The interval between the release of ATTAIN-1 data and FDA approval was approximately eight months — this approval speed is considered a regular pace for metabolic drugs, neither fast nor slow.
In terms of pricing, Eli Lilly's proposal is: patients covered by commercial insurance can start at $25 per month with discount coupons; for self-paying patients, the monthly cost ranges from $149 to $349 depending on the dosage.
Compared with the pricing of Wegovy oral at $149-299, Foundayo's out-of-pocket price ceiling is about $50 higher, but the lower limit is basically the same. The signal of this pricing strategy is: Eli Lilly hopes to quickly gain prescription volume by providing patients with a low-cost entry point covered by insurance, while leaving room for negotiation with payers through a higher out-of-pocket cap.
On April 6, Foundayo began accepting prescriptions and shipping through LillyDirect. It took only five days from approval to the first shipment—a noteworthy supply chain preparation speed for a new molecular entity.
Node Three: The Cardiovascular "Answer" of ACHIEVE-4
Then it is April 16, 2026.
The results of the ACHIEVE-4 trial were announced, a long-term study conducted across 15 countries involving more than 2,700 patients with type 2 diabetes who also have overweight/obesity and elevated cardiovascular risk, with insulin glargine as the comparator.
First, look at the primary endpoint: MACE-4 (a composite endpoint of cardiovascular death, non-fatal myocardial infarction, non-fatal stroke, and hospitalization for unstable angina) HR=0.84, 95% CI: 0.59-1.20, p=0.336. Non-inferiority was achieved, but superiority did not reach statistical significance.
This figure requires precise interpretation. A p-value of 0.336 means that the null hypothesis of "no difference" cannot be statistically ruled out—in other words, Foundayo's superiority over insulin glargine in terms of MACE-4 has not been demonstrated. However, the success of the non-inferiority design indicates that Foundayo is at least not inferior to insulin glargine. For a drug primarily indicated for weight loss and glucose reduction, this represents an acceptable cardiovascular safety profile along with a potential benefit signal.
Looking at the key secondary endpoint: MACE-3 (excluding hospitalization for unstable angina) HR=0.77, 95% CI: 0.52-1.13, p=0.181, with a 23% risk reduction, also not reaching statistical significance.
But the all-cause mortality data is striking: HR=0.43, 95% CI: 0.25-0.75, p=0.002, with a 57% reduction in risk, which is a secondary endpoint that reached statistical significance in ACHIEVE-4.
For a metabolic drug, the independent benefit signal of all-cause mortality is highly significant both clinically and in terms of regulatory approval. It implies that even if the primary cardiovascular composite endpoint fails to demonstrate superiority, Foundayo may not only help patients "live better" but also potentially "live longer"—although this conclusion requires validation through larger sample sizes.
Lilly stated in the press release: "These results demonstrate that Foundayo not only improves glycemic control and promotes weight loss, but also shows a compelling signal in all-cause mortality reduction."
Moreover, the data on A1C and weight changes at 52 weeks were equally impressive: A1C decreased by 1.6% with Foundayo (from a baseline of 8.22%) compared to a 1.0% reduction with insulin glargine, with a difference of -0.66% (p<0.001); Weight decreased by 8.8% (-8.1kg) with Foundayo (from a baseline of 90.9kg), while increasing by 1.7% (+1.4kg) with insulin glargine, resulting in a difference of -10.42% (p<0.001). The intergroup differences for both metrics reached a high level of statistical significance (p<0.001).
Notably, the insulin glargine group experienced a 1.7 kg weight gain – a classic side effect of insulin therapy.Foundayo not only had no such problem but also brought about an 8.8% weight loss.For T2DM patients with overweight/obesity, the attribute of "glycemic control and weight loss" may hold more clinical decision-making value than mere differences in cardiovascular endpoints.
Based on the data from ACHIEVE-4, Eli Lilly plans to submit the T2DM indication marketing application for Orforglipron to the FDA by the end of the second quarter of 2026.
On the first day after the ACHIEVE-4 data was released, discussions in the metabolic disease field focused on three key questions.
First, is non-inferior but not superior MACE-4 sufficient to support regulatory approval and clinical acceptance for the T2DM indication?
In the FDA's review standards for T2DM drugs, cardiovascular safety is the "threshold" (excluding additional risks through CVOT), while cardiovascular benefits are a "bonus." The non-inferiority achievement of ACHIEVE-4 means the threshold has been passed, but the lack of superiority indicates limited "bonus." However, the 57% reduction in all-cause mortality (p=0.002), a secondary endpoint signal, may play a more significant role in the review than expected.
Second, oral small molecules vs oral peptides, who is the real future?
Wegovy oral weight loss 16.6%, Foundayo up to 12.4%, with an absolute difference of about 4 percentage points. However, in the real world, compliance may be more important than absolute efficacy. If Foundayo's dosing conditions (whether fasting is required, fasting time after taking the medication) are more lenient than Wegovy oral, this compliance advantage may offset some of the efficacy gap.
Third, has the curtain on price wars already been raised?
Foundayo Self-Pay Patients $149-349/Month, Wegovy Oral Price $149-299/Month. The price ranges of the two highly overlap. Within three weeks of Wegovy Oral's launch, it reached approximately 50,000 prescriptions per week (of which about 45,000 were self-pay), demonstrating the market’s willingness to pay for "oral weight-loss drugs." Whether Foundayo can quickly capture a share in this already validated market depends on two variables: the speed of insurance coverage and clinicians' awareness and preference regarding the difference between "small molecules vs peptides."
The era of oral small-molecule GLP-1 has begun. Eli Lilly used the ACHIEVE-4 data to prove that this path is viable — at least, the bottom line of cardiovascular safety has been maintained, and the benefit signal for all-cause mortality is even more optimistic than many had anticipated.
But access does not equal success.
Weight loss of 12.4% vs. 16.6%, MACE-4 non-inferior but not superior, approved about four months later than Novo Nordisk – these gaps are "acceptable but not ideal" in clinical practice. The real decisive factor for Eli Lilly may not lie in the pharmacodynamic data itself, but in commercial execution: the speed of insurance coverage, penetration rate of patient assistance programs, progress in advancing T2DM indications, and – most crucially – whether Orforglipron can be transformed from "yet another GLP-1" into "the first non-injectable metabolic disease intervention tool."
Novo Nordisk's Wegovy injection has already built up brand momentum and accumulated clinical evidence. Eli Lilly's Orforglipron boasts the uniqueness of being a small molecule and is rapidly advancing in its indications matrix. The competition between the two has moved from "who can be the first to market" to "who can first establish prescribing habits."
On April 16, 2026, Eli Lilly obtained the entry ticket for cardiovascular safety. But the real competition begins after entering.
