Frequent suspension of delistings: ADC finally faces this critical issue

ICE Bioscience
CRO Service Provider
Since 2024, the delisting and clinical halts caused by interstitial pneumonia, ocular toxicity, and hematological toxicity have elevated the safety concerns of ADCs to a higher priority. This has profoundly impacted the R&D pace of pharmaceutical companies. Unlike the previous approach of "development first, evaluation later," ADC companies are now advancing safety and efficacy studies concurrently in the early stages of R&D. The focus of research and development has shifted from solely pursuing efficacy to minimizing toxicity while achieving therapeutic effects.
According to Dr. Ma Xingquan, co-president of Chempartner, the safety improvement in ADC development has gradually shifted from being an added advantage in the past to one of the core issues that need to be prioritized. In the past, the industry focused more on whether ADCs had sufficient killing power and whether they could deliver the payload to tumor cells. However, in recent years, it has become increasingly clear that what determines a project's ability to be sustainably advanced is not only "whether it hits the target accurately" but also "whether it can control off-target toxicity, systemic exposure, and normal tissue damage within a reasonable range while hitting the target accurately." Especially during the clinical advancement of some projects, issues such as premature payload release, off-target exposure, and a less-than-ideal therapeutic window often affect dose design, indication expansion, and even the subsequent development potential of the project.
ICE Bioscience, a CRO company with a professional evaluation platform in the ADC field, has also clearly felt the changes in customer needs. According to Dr. Li Li, head of the company's ADC business, since last year, there has been a significant increase in demand for toxicity consultations involving pulmonary toxicity (prediction of interstitial pneumonia), hematological toxicity, ocular toxicity, and others, which has brought continuous order revenue. This reflects that ADC R&D companies are experiencing a rapid increase in demand for safety evaluation, but the corresponding evaluation tools are relatively lacking.
From the perspectives of both corporate R&D and customer needs, safety improvements are no longer peripheral issues but one of the key topics that need to be considered in platform construction and project design.
Based on the safety challenges faced in ADC development, researchers have carried out a series of improvements around antibodies, linkers, and payloads. Currently, site-specific conjugation technology has gained widespread recognition.
The core logic of site-specific conjugation technology is to reduce the non-specific distribution of impurities and thereby lower toxicity by precisely controlling the drug-to-antibody ratio (DAR) and conjugation sites.
Traditional randomly conjugated ADCs have a DAR value distribution between 0 and 8, forming a complex mixture. This heterogeneity not only makes it difficult to control the quality between batches, but more importantly, components with high DAR are prone to aggregation and higher toxicity, while those with low DAR lack efficacy.
ADCs with uniform DAR (such as DAR2, DAR4) obtained through site-specific conjugation exhibit more predictable pharmacokinetic behavior. More importantly, it can effectively reduce the "premature release" of payloads in the bloodstream, thereby significantly lowering off-target toxicity and improving patient tolerance to the drug.
From the perspective of R&D trends, the penetration rate of site-specific conjugation technology in ADCs entering clinical trials globally has risen sharply. From an industrial perspective, large pharmaceutical companies (such as Daiichi Sankyo, Astellas, and Innovent Biologics) have successively adopted the method of either introducing or self-developing site-specific conjugation platforms as the "underlying technology" for their ADC pipelines. From a regulatory perspective, an ADC molecule with uniform composition and controllable manufacturing processes also has a more straightforward registration pathway.
Taking Boan Biotech' BA1301 as an example, this ADC targeting CLDN18.2 utilizes C-Lock site-specific conjugation technology. Phase I clinical data presented at the 2025 ESMO Congress showed that the product's hematological (anemia and neutropenia incidence rate was only 1.7%) and gastrointestinal (vomiting incidence rate was 1.7%, nausea 0%) toxicities were significantly lower than other ADCs in the same target class. Pharmacokinetic data revealed an extremely low payload release rate of just 0.002%. This set of data very intuitively demonstrates the high stability brought by site-specific conjugation technology has successfully translated into a safety advantage in clinical settings.
"Advances in antibody engineering technology have laid the foundation for the realization of site-specific conjugation," said Dr. Yang Yang, CEO of GlycanLink, in an interview with VCBeat. Research has found that there is a highly conserved glycosylation site at the 297th asparagine position of humanized IgG antibodies. This site can introduce toxic payloads through enzymatic modification, making it an excellent choice for site-specific conjugation. In the future, with the continuous iteration of related technologies, more new site-specific conjugation strategies will emerge. While these technologies help researchers solve the problem of conjugation uniformity, they will also strongly promote the improvement of drugs in terms of safety and stability.
Dr. Yang Yang believes that GlycanLink's technology platform can provide more feasible solutions for innovative pharmaceutical companies to build "antibody-drug conjugate systems": first, by verifying the feasibility of molecular design through process development and optimization, and then achieving the desired biological functions through multi-dimensional combinations of payload, linker, and antibody.
And site-specific conjugation technology is the key support to achieve this core value.
Beyond site-specific conjugation technology, other improvement strategies are also attracting researchers' attention.
1) The development strategy of bispecific ADC has been validated.
The core logic of bispecific ADCs is to enhance tumor selectivity by simultaneously targeting two antigens or two epitopes of the same antigen, reducing collateral damage to normal tissues that express only one target, thereby improving safety. This logic is also easy to understand: if one lock is prone to accidental opening, adding another lock provides double verification.
The safety improvement of bispecific ADC development strategies has been clinically validated. BL-B01D1, developed by Biokin Pharmaceutical as a bispecific antibody-drug conjugate (ADC) targeting EGFR/HER3, is the world's first bispecific ADC to publish Phase III study results. The design concept of BL-B01D1 demonstrates unique and controllable safety characteristics, with its most prominent advantage being a lower incidence of interstitial lung disease (ILD) compared to similar ADC drugs.
ILD is a serious toxicity event that has drawn significant attention in ADC drug development. In the clinical study of BL-B01D1, only a 0.6% incidence rate of ILD was reported, all of which were Grade 1. For reference, the incidence rates of ILD for some other ADCs are much higher. For example, trastuzumab emtansine reported an ILD incidence rate of 9.49%, while trastuzumab deruxtecan was significantly higher at 68.43%. Therefore, the extremely low risk of ILD associated with BL-B01D1 is a notable advantage. BL-B01D1 has successfully validated the feasibility of the revolutionary bispecific ADC technology platform. Although cross-trial comparisons should be approached with caution, such a stark difference still leaves the industry highly optimistic about the safety potential of bispecific ADCs.
In 2023, Biokin Pharmaceutical's product reached an $84 billion collaboration with BMS, holding a significant position in the history of Chinese products going overseas, demonstrating its enormous commercial value.
Beyond the bispecific ADC strategy, many researchers are actively attempting approaches such as engineering the Fc region of antibodies to reduce toxicity to immune cells, and designing more hydrophilic and stable linkers.
In Dr. Li Li's view, these categories are currently the more mainstream safety improvement strategies. In addition, prodrug-type ADCs have also begun to be explored by some companies.
2) Prodrug-type ADCs are being actively explored.
The core logic of prodrug-type ADCs is to add "masking groups" to antibodies that can be specifically cleaved by the tumor microenvironment, keeping the antibody in a "shielded" state during circulation and "unlocking" to restore activity after entering tumor tissues. This means being "shielded" in circulation, "unlocked" in the tumor microenvironment, and exerting therapeutic effects at the tumor site.
CytomX is one of the pioneers in the field of masked antibodies and has established strategic partnerships with giants such as AbbVie, Astellas, and Bristol-Myers Squibb. However, in recent years, these collaborations have been successively terminated, and CytomX has now concentrated all its resources on its ADC drug Varseta-M. Not long ago, Varseta-M achieved positive results in colorectal cancer: the objective response rate (ORR) in the high-dose group reached 32%, and the disease control rate (DCR) was 88%, indicating that the majority of patients had their tumors controlled. The median progression-free survival (PFS) for Varseta-M was 7.1 months, significantly better than the current standard third-line or higher monotherapy (typically PFS of 2–3.7 months). These data suggest that Varseta-M could become a new treatment option for later-line mCRC. CytomX's stock price surged over 44% on the same day.
Currently, prodrug-type ADCs are still in the early stages of development, with few participants. Adagene utilizes its SAFEbody®The platform provides precise masking for antibodies. The platform has reached a cooperation agreement with Exelixis to develop novel masked ADCs targeting the points nominated by Exelixis.
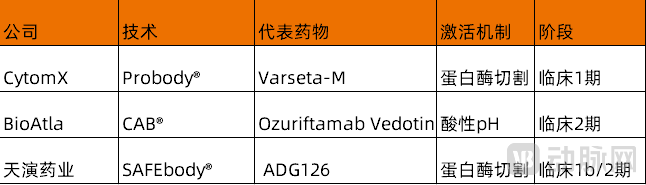
Introduction to New Masking Technology (Source: VCBeat)
As a new technology platform, masking technology still requires continuous iteration and validation. As leading products from companies like CytomX and Adagene advance into later-stage clinical trials, we will see more critical clinical data. This data will verify whether masking technology can truly translate into survival benefits for patients.
The safety improvement of ADC has now entered the stage of overall synergy.
From a technical perspective, since site-specific conjugation can produce ADCs with uniform DAR values, it offers better safety compared to traditional random conjugation and stronger predictability in pharmacokinetic behavior. Therefore, site-specific conjugation technology is expected to become a "standard feature" in ADC development rather than a differential advantage.
In addition, as the advantages of improved strategies such as bispecific ADCs, Fc engineering, and novel linkers are gradually clinically validated, these technology platforms will demonstrate sustained competitiveness. In the longer term, prodrug-type ADCs, if their safety window advantages can be validated across multiple indications, are expected to provide new solutions for highly toxic payload ADCs.
In practical applications, these strategies are not mutually exclusive but rather work in synergy. Future innovative ADCs are highly likely to be based on site-specific conjugation, with additional features such as bispecificity, prodrug forms, and novel linkers, achieving a "modular combination." The current R&D has already reflected this trend.
JSKN021, independently developed by Alphamab Oncology, had its IND application accepted by the NMPA in March this year. This product is the world's first ADC designed using the dual-antibody dual-payload approach. As an EGFR/HER3-targeting ADC, JSKN021 utilizes glycan-specific conjugation technology to site-specifically and quantitatively conjugate two different cytotoxic payloads to the Fc region of the antibody, thereby achieving a molecule with higher homogeneity.
Through the combination design of site-specific conjugation and bispecific antibodies, JSKN021 demonstrated unique high stability and safety in preclinical studies.
Of course, the integration of technology is only one aspect of safety improvement; more importantly, it is the systematic nature of the overall ADC design concept.
"If we look at the development stage of the entire industry, the optimization of ADC safety has transitioned from a past reliance on experience-driven approaches to the current phase, which emphasizes systematic engineering and platform collaboration." ChemPartner's Dr. Ma Xingquan told VCBeat. Currently, people are no longer just focusing on making local improvements at a single point, but are looking at the therapeutic window as a whole, considering targets, antibodies, linkers, payloads, conjugation methods, analytical characterization, process development, and clinical translation. "This change is very important because it means that the competition in ADCs is shifting from who can make them to who can achieve efficacy, safety, and developability all together."
According to Dr. Ma Xingquan, the existing development system of ChemPartner is not limited to a single point but integrates linker design, conjugation construction, analytical characterization, in vivo and in vitro evaluation, and early CMC into a complete chain of evidence. It is reported that ChemPartner has established a new generation CP-C2LP™ technology platform. As a two-dimensional or even multi-dimensional toxin release control model, this technology platform can improve the safety of ADCs while maintaining efficacy. It can also enhance bioconjugation efficiency for certain special payloads, further improving safety and efficacy.
This shows that the safety improvement of ADC has been incorporated into the overall R&D system by pharmaceutical companies for comprehensive consideration. Safety improvement has become a full-chain systematic project.
The progressive enhancement of safety ultimately points to a simple fact: what patients need is not "stronger efficacy," but better therapeutic drugs.
With the development of technology, the field of conjugated drugs is also undergoing profound changes, evolving from a "magic bullet" that pursues extreme cell killing to a programmable, modular precision drug delivery system.
ADCs initially only carried highly toxic chemotherapy drugs to kill rapidly dividing tumor cells. Now, the payload has expanded to include various non-cytotoxic substances, such as glucocorticoid receptor modulators used for autoimmune diseases and oligonucleotides capable of gene regulation. Additionally, new forms like PDCs and RDCs have also demonstrated clinical value. The diversification of conjugated drugs has broadened the scope of treatment from oncology to a wider range of indications, offering potential treatments for autoimmune diseases, genetic disorders, and more.
But no matter how the form of conjugated drugs evolves, safety is the premise for expanding across indications.
