One Device Covers Phacoemulsification, Vitrectomy, and Laser! Sierra Medical's All-in-One Ophthalmic Anterior and Posterior Segment Surgical Platform SIERRA VISION SYSTEM® Approved

Sierra Medical
Innovative Medical Device Developer
China-Made High-End Ophthalmic Surgical Equipment Achieves Milestone Breakthrough!
On April 14, 2026, SIERRA VISION SYSTEM independently developed by Sierra Medical®Ophthalmology Anterior and Posterior Segment Surgical Platform Officially Obtains NMPA Class III Medical Device Registration Certificate (GuoXieZhuZhun):20263160759)。As the first domestically produced ophthalmic anterior and posterior segment comprehensive surgical platform that highly integrates core functions such as phacoemulsification, vitrectomy, laser, electrocoagulation, and illumination, it can cover the entire process of anterior and posterior segment surgeries without additional equipment, providing a truly "one-stop" surgical solution for clinical use.
SIERRA VISION SYSTEM®The approval marks a leapfrog upgrade for domestically produced high-end ophthalmic surgical equipment, transitioning from single-point breakthroughs to comprehensive solutions. It not only effectively fills the long-standing gap in the domestic market for integrated phacoemulsification and vitrectomy machines but also, with internationally competitive quality, propels Chinese ophthalmic devices into a new phase of high-end, platform-oriented development, offering patients better, more efficient, and safer treatment options.

SIERRA VISION SYSTEM®
With the intensification of global aging trends, the incidence of fundus diseases continues to rise, becoming the leading cause of irreversible blindness. Fundus surgeries are complex and their demand is increasing daily, not only placing higher demands on the professional skills of fundus doctors but also raising the bar for innovation in surgical equipment and instruments.
The Phaco-Vitrectomy Machine is a high-end ophthalmic surgical device that integrates the functions of phacoemulsification and vitrectomy. It is mainly used to treat various blinding eye diseases such as cataracts, retinal detachment, and diabetic retinopathy. It can complete anterior and posterior segment combined treatment in a single surgery, significantly improving the efficiency and safety of complex eye surgeries, reducing patient trauma and recovery time, and is an indispensable core piece of equipment for modern minimally invasive precision treatment of eye diseases.
However, the phacoemulsification and vitrectomy integrated machine involves multidisciplinary intersections such as optics, fluid dynamics, precision machinery, and energy control, with extremely high technical barriers. It also requires long-term clinical validation to build trust among doctors. Therefore, the market has long been dominated by overseas manufacturers like Alcon, Bausch + Lomb, and Zeiss, which have decades of technical accumulation and a global clinical network. More realistically,Imported equipment not only has high acquisition costs, but also the prices of its matching consumables are equally expensive. Moreover, the uncertainty of international trade frictions in recent years has posed challenges to the stability of the equipment supply chain.
At the same time, the state has implemented clear restrictions on certain imported medical devices in the government procurement sector. The policy of "prioritizing domestically produced products" is rapidly evolving from a guiding principle into a quantifiable rigid rule. There is a growing demand in clinical settings for domestically produced alternatives that match international performance standards, ensure supply chain stability, and align with policy directives.
Based on these factors, domestically produced brands have gradually started to make their moves. However, the solutions are not without flaws: while some phacoemulsification and vitrectomy integrated machines possess the core functions of both phacoemulsification and vitrectomy,But functions such as lasers still rely on external devices or module expansions, leading to issues like device switching, parameter coordination, space management, and process integration.
Sierra Medical SIERRA VISION SYSTEM®The approval undoubtedly meets the aforementioned needs. As a genuine anterior and posterior segment integrated solution, it provides clinical practice with a more systematic, stable, efficient, and convenient new option.
As a high-end ophthalmic anterior and posterior segment comprehensive surgical platform, SIERRA VISION SYSTEM®Relying on Sierra Medical's full-stack self-developed"Fluid + Optics + Energy"Three major technology platforms can complete the entire process from cataract phacoemulsification to vitrectomy, laser photocoagulation, and intraocular filling without additional equipment.One-stop solution to meet multi-scenario surgical needs, with comprehensive performance aligned with international top-tier standards.
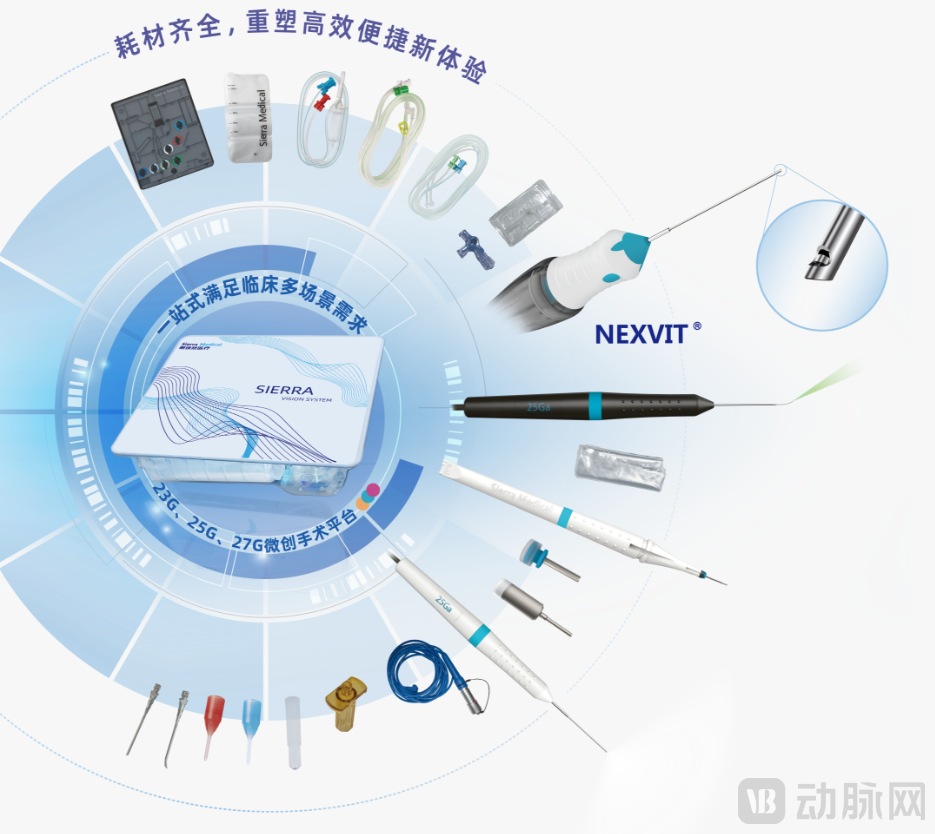
Supporting the strength of this product is Sierra Medical's sustained high-intensity investment in research and development. As of now, Sierra MedicalTotal financing of nearly 300 million yuan, ranking at the forefront of the track.The investors include top institutions such as Qianji Capital, Shunwei Capital, Boxin Capital, and Hony Capital, as well as industry funds like Aier Venture Capital and Aibo Qing Shi. The company places high importance on technological innovation.R&D investment exceeds 100 million yuan.This "heavy research and heavy investment" development strategy has built an insurmountable technical barrier for Sierra Medical——The company currently has dozens of patent layouts, and its core consumables such as vitrectomy heads, ophthalmic trocars, and intraocular laser fibers have all been approved for marketing.ByLeveraging leading R&D progress and comprehensive productionProduct Matrix, Firmly Ranking in the First Tier of the Track.
For domestically produced high-end medical equipment to achieve true domestic substitution, it is not enough to just "produce it"; it must also "perform well and consistently." Clinical trust stems from real-time feedback during surgeries. Especially for sophisticated devices like phacoemulsification and vitrectomy machines, their performance, maturity, ease of use, and system stability are the core factors that determine whether they can truly enter clinical application.
Sierra Medical Adheres to the Path of Large-Scale Clinical Evidence-Based Research——From NEXVIT®Series of vitrectomy heads to the newly approved SIERRA VISION SYSTEM®,Sierra Medical always collaborates with top ophthalmic centers in China to conduct large-scale, multi-center clinical trials for each product, continuously refining and iteratively optimizing in complex surgical scenarios, and verifying the safety and efficacy of the products with solid clinical data.
At the same time,Sierra Medical also has a research and development center and production base of nearly 4,000 square meters.(including hundreds of square meters of Grade D GMP clean room), established a comprehensive and stringent quality system, forming standardized intelligent manufacturing capabilities,Ensure the stable, safe, and scalable delivery of equipment. This full-chain quality control from the laboratory to the operating room not only ensures the compliance of each device but also guarantees excellent performance stability and reliability.
If we talk about SIERRA VISION SYSTEM®The approval of Sierra Medical marks a powerful breakthrough in the deep waters of technology. The platform strategy behind it, however, reveals the company's broader industrial ambitions — within Sierra Medical’s strategic logic,The breakthrough of single-device equipment is just the starting point; the real barrier lies in the profound mastery of the overall ophthalmic surgery.This goes beyond the role of a traditional device manufacturer,To become a comprehensive solution provider covering the entire process and all scenarios of ophthalmic anterior and posterior segment surgeries.
Sierra Medical is strengthening its system capabilities through a dense product matrix. In addition to the recently approved SIERRA VISION SYSTEM®Outside of the phacoemulsification and vitrectomy machine, the company is alsoSynchronously develop core equipment such as phacoemulsification machines, digital microscopes + iOCT, and continuously improve the matching consumables system., achieving an integrated intelligent collaboration of "hardware + software + consumables" to build an overall solution covering the entire process of ophthalmic surgery.
As this product matrix becomes increasingly complete, Sierra Medical's systematic capabilities have gradually become prominent, leading to a shift in the industry's competitive logic. Traditional ophthalmic equipment companies often focus on a specific niche, whereas Sierra Medical has built a comprehensive service network covering the entire chain of equipment, software, and consumables. Upon achieving self-research and production of core equipment and consumables,The platform can create stronger integrated advantages in surgical procedures, operational experience, and clinical collaboration, which piecemeal procurement cannot match.At the same time, unified operation training, parts supply, and after-sales service systems have significantly reduced the procurement costs for hospitals and the usage barriers for doctors. This "one-stop" solution offers far greater long-term value than competition based solely on the price of individual devices.
It can be foreseen that with the development of Sierra Medical's SIERRA VISION SYSTEM®With the popularization and promotion, the competition between domestically produced high-end ophthalmic surgical equipment and imported brands will no longer be a simple contest of cost-performance ratio, nor a race to surpass single technical specifications, but will evolve into a system-level competition of "comprehensive solutions." For Sierra Medical, this is just the beginning; for China's high-end ophthalmic equipment industry, it may signify the official dawn of a brand-new era.
