Three milestones in one day! Mabwell races toward "A+H" innovative drug company status

Mabwell
Innovative Biopharmaceutical Company
On April 16, Mabwell (688062.SH), a company listed on the STAR Market, achieved three milestones in a row.
·Original Class 1 Innovative Drug,The world's first TCE (T-cell engager) bispecific antibody targeting LILRB4/CD3 to enter clinical trials 6MW5311 for Injection, for the clinical trial application (IND) of [hematological tumors (acute myeloid leukemia, chronic myelomonocytic leukemia, and multiple myeloma)] indications, has been officially accepted by the NMPA.
·The supplemental indication application for China's first approved Xgeva® biosimilar, Maiweijian®, has been accepted by the NMPA — adding the indication for [bone metastases from solid tumors and multiple myeloma], for use in patients with bone metastases from solid tumors or in patients with multiple myeloma to delay or reduce the risk of skeletal-related events (pathological fracture, spinal cord compression, bone radiation, or bone surgery).
·Regarding its two denosumab injection products — Mailishu® (Prolia® biosimilar) and Maiweijian® — Mabwell has signed licensing and commercialization agreements with its strategic partner in the Malaysian market. The partner will be responsible for the registration and sales of the two products in Malaysia, while Mabwell will be responsible for product development, manufacturing, and commercial supply.
Listed on the STAR Market of the Shanghai Stock Exchange in January 2022, Mabwell filed its first application for listing on the Hong Kong Stock Exchange on January 6, 2025, and received the overseas issuance and listing filing notice from the China Securities Regulatory Commission on November 21, 2025. In early April 2026, after three filings, Mabwell formally passed the listing hearing of the Hong Kong Stock Exchange, with Citic Securities and Haitong International serving as joint sponsors.
Once successfully listed, Mabwell will become another innovative biopharmaceutical company with an "A+H" dual listing.
1Three-time Filing with HKEX, ADC+TCE Bispecific Antibody Advancing on Multiple Fronts
Founded in 2017 and positioned as an innovative pharmaceutical company, Mabwell has continuously developed and upgraded its technology platforms, including an ADC drug development platform, an integrated high-efficiency antibody discovery platform, and a TCE bispecific/trispecific antibody development platform.
In this Hong Kong listing, the company plans to issue no more than 62.664 million overseas listed ordinary shares. The proceeds will be primarily used for the clinical development of its core product 9MW2821, the advancement of its oncology and age-related disease pipelines, and commercialization build-out, further expanding international financing channels and accelerating R&D and globalization efforts.
Mabwell currently has four marketed products and ten drug candidates (one at the NDA stage, eight in clinical stages, and one in preclinical stage), covering therapeutic areas such as oncology, immunology, ophthalmology, and orthopedics, and has accumulated 140 patents globally.
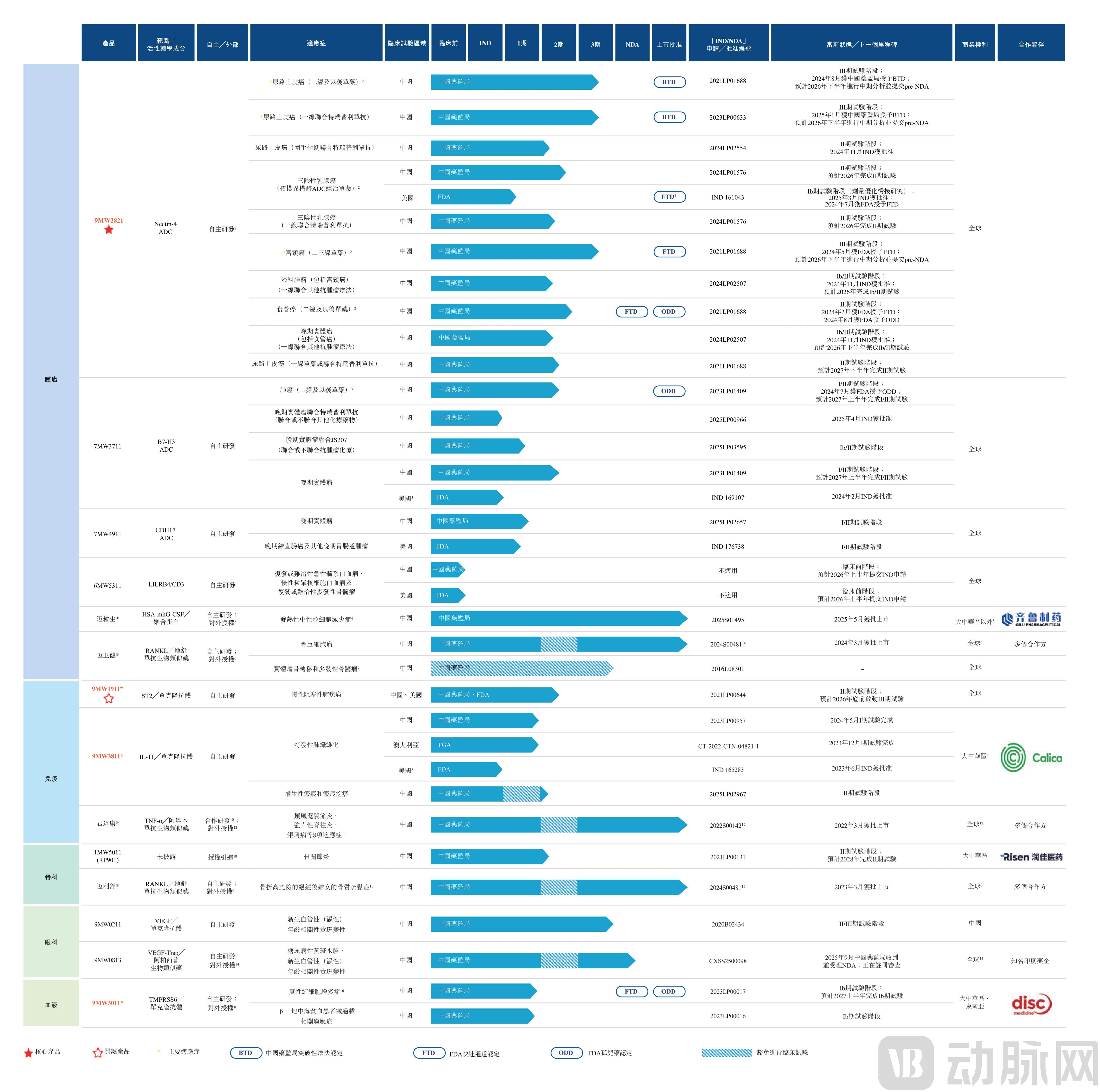
Its core product, 9MW2821, is the most advanced Nectin-4-targeting ADC in China, having received three Fast Track Designations (FTD) and one Orphan Drug Designation (ODD) from the FDA, as well as two Breakthrough Therapy Designations (BTD) from the NMPA for both monotherapy and combination use. Clinical trials are currently underway for multiple indications, including urothelial carcinoma, cervical cancer, triple-negative breast cancer, and advanced esophageal cancer.
Among Nectin-4 ADC products globally for the treatment of urothelial carcinoma, 9MW2821 ranks second only to Padcev, the only approved product in this class, developed by Astellas/Seagen (Pfizer). Additionally, it is the first product of its kind globally to enter pivotal Phase III clinical trials for cervical cancer. According to the prospectus, 9MW2821 is planned to submit an NDA (New Drug Application) to the NMPA in 2027 for monotherapy as a second-line or later-line treatment for locally advanced or metastatic urothelial carcinoma.
Furthermore, its ADC pipeline also includes 7MW3711 (a B7-H3-targeting ADC, in Phase I/II clinical trials) and 7MW4911 (a CDH17-targeting ADC, in Phase I clinical trials).
6MW5311 for injection, whose IND was formally accepted by the NMPA today, is a Class 1 innovative drug independently developed by Mabwell and the world's first TCE bispecific antibody targeting LILRB4/CD3 to enter clinical submission, intended for the treatment of hematologic malignancies (acute myeloid leukemia, chronic myelomonocytic leukemia, and multiple myeloma).
Through a unique "2+1" asymmetric molecular structure, 6MW5311 simultaneously targets LILRB4 and CD3, bridging tumor cells and T cells to form an immune synapse, activating T cells and efficiently killing tumor cells. Incorporating a unique steric hindrance design significantly reduces CD3 antibody binding activity to T cells in the absence of tumor cells, while demonstrating potent killing activity only in the microenvironment where tumor cells and T cells coexist — thereby greatly improving safety while enhancing efficacy.
Another key antibody product, 9MW1911, is a ST2-targeting monoclonal antibody for the treatment of autoimmune diseases. It is planned to be developed as the first large-molecule drug for chronic obstructive pulmonary disease (COPD) targeting a non-Th2 pathway. A Phase Ib/IIa trial is currently underway in China, and it has also received FDA IND approval for Phase IIa clinical trials.
Additionally, 9MW3811 (an IL-11-targeting humanized monoclonal antibody) is being developed for the treatment of idiopathic pulmonary fibrosis and advanced malignancies. It is also the first IL-11-targeting drug in China to enter clinical development for pathological scars (including hypertrophic scars and keloids).
One Class 1 innovative drug with wholly independent intellectual property rights has been approved for market launch — Mailisheng® (Albipagrastim alfa for injection, 8MW0511) was approved in May 2025. It is the first next-generation long-acting granulocyte colony-stimulating factor (G-CSF) innovative drug developed using albumin long-acting fusion technology to be marketed in China. It is indicated for [reducing the incidence of infection manifested as febrile neutropenia in adult patients with non-myeloid malignancies receiving myelosuppressive anticancer therapy that is likely to cause febrile neutropenia].
In June 2025, Mabwell granted Qilu Pharmaceutical an exclusive license to develop, manufacture, modify, utilize, and commercialize the licensed product within Greater China. Mabwell's wholly-owned subsidiary T-mab BioPharma is entitled to receive upfront and sales milestone payments of up to 500 million RMB, plus additional royalty fees. Effective January 1, 2026, Mailisheng® was successfully included in the National Reimbursement Drug List (2025 edition) less than six months after its approval, becoming a new long-acting G-CSF treatment option.
2Self-Sustaining R&D, First-to-Market Biosimilars, and Licensing Deals Covering Over 27 Countries Worldwide
From a financial perspective, Mabwell's revenue for 2024 and 2025 was approximately 199.6 million RMB and 658.6 million RMB, respectively, representing a year-on-year increase of 229.8% in 2025. The core driver of this growth was licensing revenue — for 2024 and 2025, licensing revenue amounted to 55.03 million RMB and 409 million RMB, accounting for 27.6% and 62.0% of total revenue, respectively.
Overall, Mabwell remains in a loss-making position, with net losses of approximately 1.047 billion RMB and 972 million RMB for 2024 and 2025. However, there are positive signs of cash flow stability — year-end cash and cash equivalents stood at approximately 1.228 billion RMB and 1.526 billion RMB, respectively.



Summary of Comprehensive Income Statement / Summary of Cash Flow Statement
Nevertheless, Mabwell has already launched commercial products and established self-sustaining capabilities. Drug sales revenue for 2024 and 2025 reached 145 million RMB and 250 million RMB, accounting for 72.4% and 38.0% of total revenue, respectively. Notably, the prospectus indicates that the increase in sales revenue was primarily driven by increased sales of Mailishu®. The Southeast Asian market may be an important growth driver in this regard.
Mabwell has three biosimilars approved for marketing in China, all of which have been included in the National Reimbursement Drug List (NRDL) and are not subject to volume-based procurement (VBP):
·Mailishu® is the second Prolia® denosumab biosimilar approved in China for the treatment of osteoporosis. It was approved in March 2023 for the treatment of osteoporosis in postmenopausal women at high risk of fracture. Sales revenue for 2024 and 2025 reached 124.4 million RMB and 202.8 million RMB, respectively.
·Maiweijian® is the first Xgeva® denosumab biosimilar approved in China, granted approval in March 2024 for the treatment of giant cell tumor of bone that is unresectable or where surgical resection is likely to result in severe morbidity. As announced today, its supplemental application for the treatment of bone metastases from solid tumors and multiple myeloma has been formally accepted by the NMPA. Sales revenue for 2024 and 2025 reached 14.6 million RMB and 3.7 million RMB, respectively.
·Junmaikang® is an adalimumab biosimilar. It was approved in March 2022 for the treatment of rheumatoid arthritis, ankylosing spondylitis, and psoriasis, and in November of the same year received approval for supplemental indications including Crohn's disease, uveitis, polyarticular juvenile idiopathic arthritis, pediatric plaque psoriasis, and pediatric Crohn's disease.
·Additionally, an aflibercept (Eylea) biosimilar, 9MW0813, had its NDA accepted by the NMPA in September 2025. It is expected to be approved for the treatment of ophthalmic diseases characterized by abnormal vascular growth and increased vascular permeability, such as wet age-related macular degeneration (AMD).
Since 2025, Mabwell has entered a period of explosive growth in the overseas commercialization of its products. In August, Mailishu® and Maiweijian® received marketing approval from the Drug Regulatory Authority of Pakistan, becoming the first biosimilars in their category in Pakistan. In December, Junmaikang® was approved for marketing in Indonesia.
Looking back, Mabwell has long taken a strategic approach to global expansion. For emerging markets and countries along the Belt and Road Initiative, the company primarily adopts a model of "licensing + local partnership" (e.g., supplying active ingredients for local filling, or providing cell lines for local manufacturing). Mabwell has established an international business department responsible for screening local partners and advancing registration filings, while delegating registration, filling, sales, and promotion in target markets to local partners with on-the-ground experience. This approach leverages their registration capabilities and commercial networks to accelerate product launches and increase the share of overseas revenue.
For example, with Maiweijian® and Mailishu®, Mabwell has entered into commercialization agreements (including commercial licenses) with leading pharmaceutical companies in multiple countries, including Brazil, Peru, the Philippines, and the Middle East and North Africa region. Separately, the company has entered into commercialization agreements (retaining ownership rights) in 27 countries, including Colombia, Indonesia, and Thailand.
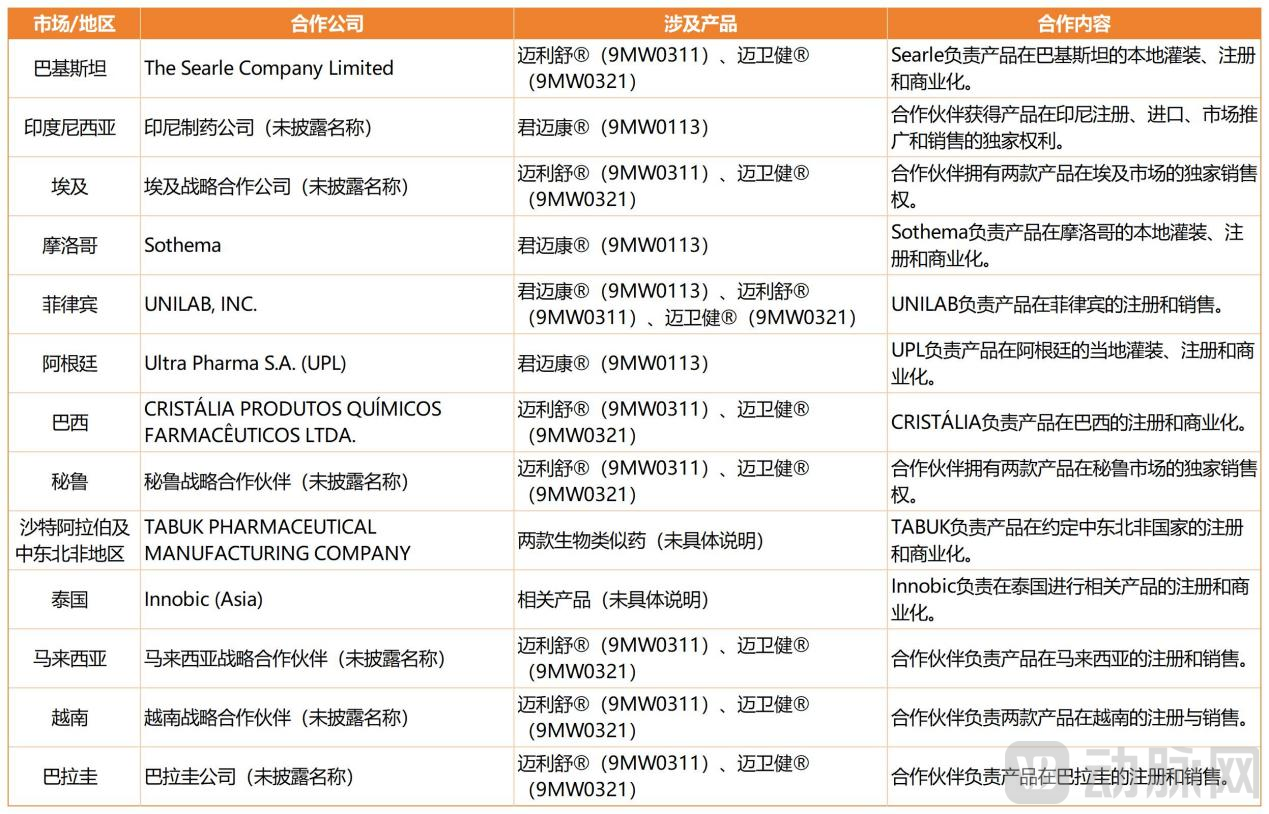
According to Incomplete Statistics from VCBeat, Mabwell's Overseas Map
Meanwhile, the innovative drug business segment has also reached multiple licensing deals for global expansion, continuously exploring business development opportunities for its ADC technology platform and innovative pipeline products:
In January 2023, Mabwell entered into an exclusive license agreement with Disc for 9MW3011 (an anti-TMPRSS6 monoclonal antibody for the treatment of iron overload anemia), under which it is entitled to receive up to $412.5 million in upfront payments, milestone payments, and royalty fees.
In June 2025, Mabwell entered into an exclusive license agreement with Calico for 9MW3811 (an IL-11-targeting humanized monoclonal antibody), under which it is entitled to receive over $600 million in upfront payments, milestone payments, and royalty fees.
In October 2025, Mabwell entered into an exclusive license agreement with Kalexo Bio for the development of novel dual-targeting siRNA drug candidates.
