Raised $800M+! How is this Biotech using AI to tackle "undruggable" cancer targets?
Recently, Parabilis Medicines (formerly FogPharma, hereinafter referred to as Parabilis) announced the completion of a new round of $305 million in Series F financing.
This round of financing was co-led by RA Capital Management, Fidelity Management & Research Company, and Janus Henderson Investors, with other participants including star institutions such as Frazier Life Sciences and Soleus Capital. As a clinical-stage biopharmaceutical company, Parabilis has been deeply engaged in the field of oncology since its establishment. To date, Parabilis has completed five rounds of financing, accumulating a total of $801 million in funding.
What is the charm of Parabilis Medicines, Inc. that continuously attracts the attention of capital?
1Harvard Chemist Who Founded Over 10 Biotechs Teams Up with Former Global R&D Head of Johnson & Johnson to Solve the Dilemma of "Target Recognizable, Drug Hard to Develop"
In the field of cancer treatment, "target identifiable, drug hard to develop" has been a long-standing challenge for the global pharmaceutical industry. With the continuous advancement of medical research, scientists have discovered a large number of disease targets closely related to tumor occurrence and development. However, more than 80% of these targets are considered "undruggable" and cannot be effectively intervened through traditional drugs. The core technical bottlenecks that make these targets difficult to drug lie in two aspects: location issues and structural issues.
Most key tumor targets are located inside cells. Due to their large molecular weight, antibodies cannot penetrate the cell membrane, making these intracellular targets inaccessible. At the same time, these targets are mostly protein-protein interaction targets with flat surfaces that lack the "deep binding pockets" required for small molecule drugs, making it difficult for traditional small molecule drugs to achieve precise and efficient binding. Even if they can penetrate the cell membrane, they are easily degraded by enzymes in the body, rendering them unable to exert targeted therapeutic effects.
For many years, pharmaceutical companies and research institutions around the world have been seeking ways to break free from this dilemma, attempting to develop new drugs capable of penetrating cell membranes and precisely targeting flat intracellular sites. In 2015, Parabilis Medicines, Inc. was founded—headquartered in Cambridge, Massachusetts, this company boasts a core team with both top-tier scientific research capabilities and extensive industry experience:
Co-founder Dr. Gregory Verdine, in addition to being a scientist, is also an entrepreneur and investor. He became a tenured professor in the Chemistry Department at Harvard University at the age of 35. With decades of dedication to chemical biology and new drug development, he has founded more than 10 biotech companies, five of which have successfully gone public. Among them, Gloucester Pharmaceuticals was acquired by biopharmaceutical giant Celgene. He led the development of three FDA-approved drugs: Romidepsin (Istodax®), Paritaprevir (a component of Viekira Pak®), and Glecaprevir (a component of Mavyret®).
Chairman and CEO Dr. Mathai Mammen, formerly a member of the Executive Committee and Global Head of R&D at Johnson & Johnson, has long been deeply involved in the biopharmaceutical field. With extensive experience in corporate management and the coordination of new drug development, he has led company financing, pipeline advancement, and global strategic planning. Under his leadership, Parabilis Medicines, Inc. successfully completed multiple rounds of significant financing, advancing its core pipeline to the clinical stage.
2From Natural Protein Helices to Designable "Stapled Peptides"
The core asset of Parabilis Medicines, Inc. is its self-developed Helicon™ platform. The underlying technology stems from the groundbreaking research by Professor Verdine and his laboratory team. In the late 1990s, the team achieved a breakthrough in long-term research: the protein alpha-helix, which can both embed into cell membranes for efficient transmembrane delivery and bind to flat protein surfaces, possesses key characteristics essential for overcoming the challenge of "undruggable" targets.
The α-helix is not an arbitrary protein structure but a natural conformation of proteins. It possesses a certain degree of rigidity, along with sufficient flexibility and inherent stability.
In 2000, the Verdine team, based on this insight, invented the "stapled peptides" technology (stapled peptides), the prototype of Helicon technology—using chemical cross-linking technology to stabilize short peptides into an α-helical conformation, preventing their structure from becoming loose, while precisely controlling the amino acid sequence of the peptide chain, allowing it to maintain the transmembrane ability of the α-helix and accurately match the structure of intracellular targets.
Specifically, the team selects specific amino acids (non-natural amino acids) and connects them through a proprietary linker to form a stable α-helix structure. This allows the structure to easily penetrate cell membranes while enabling tight binding with intracellular targets via amino acid side chains on the surface. Additionally, the "stapling" design prevents the peptide chain from being degraded by proteases in the body, allowing the drug to remain effective for an extended period.
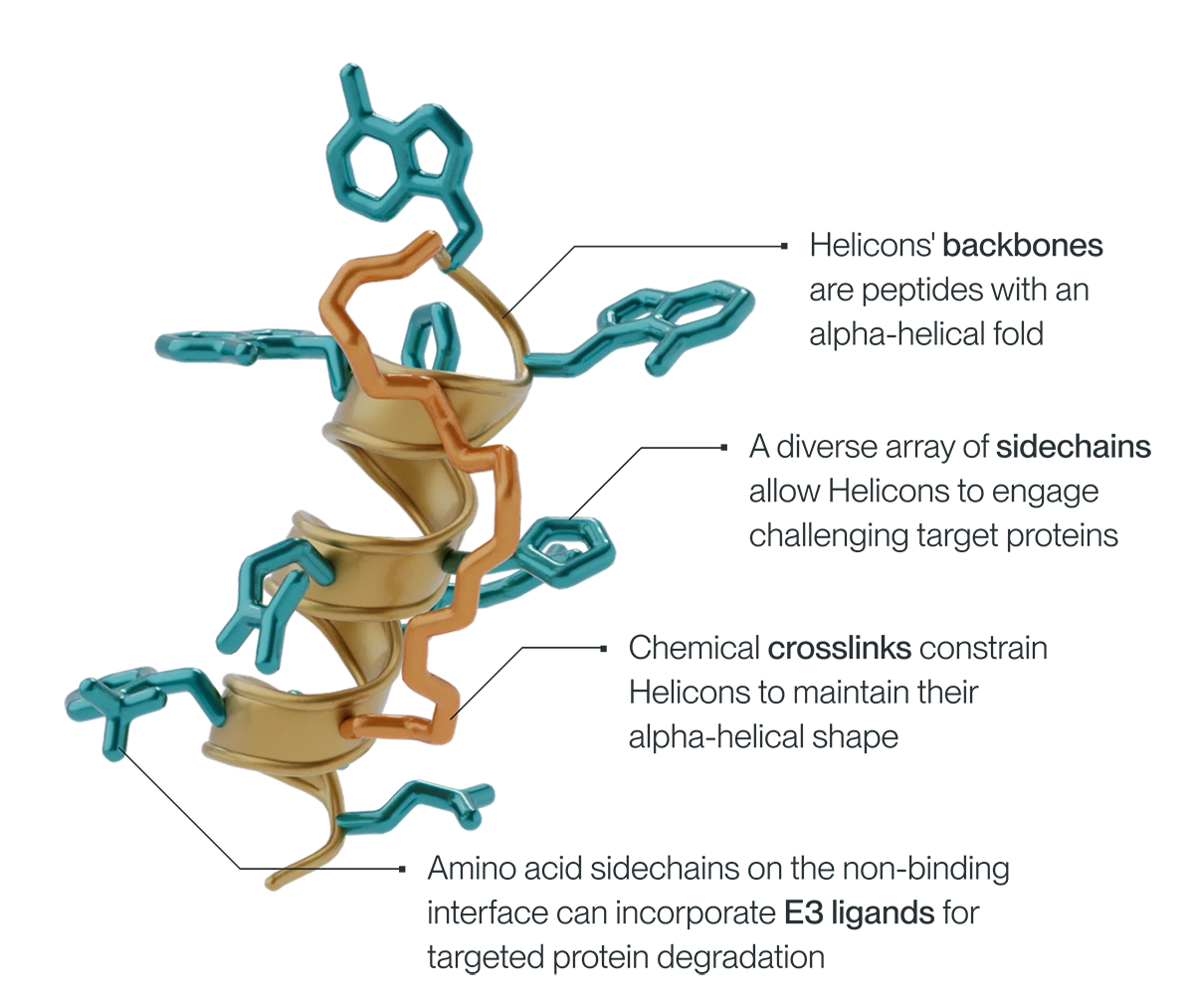
Helicon™ Technology Principle (Image Source: Parabilis Official Website)
3Integration of Modular Chemistry + High-Throughput Validation to Build an AI + Closed-Loop Platform
With the continuous iteration of biochemistry technology and computational science, Parabilis has been深耕ing in cutting-edge peptide technologies such as stapled peptides, eventually building up the unique Helicon™ platform. Parabilis defines it as a novel drug creation system that integrates AI and experimental techniques, centered on three core pillars: AI and physical computation, modular chemistry, and multiple experimental platforms. This significantly improves the efficiency of new drug development and makes "undruggable" targets truly intervenable and treatable.
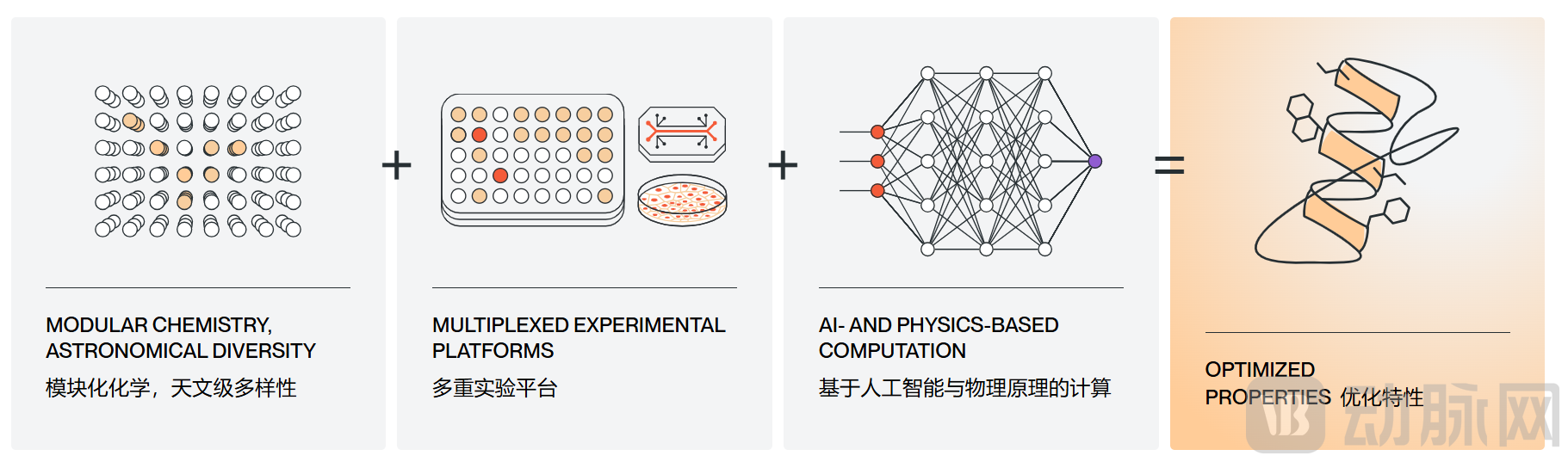 The Three Core Components of the Helicon™ Platform (Source: Parabilis Official Website)
The Three Core Components of the Helicon™ Platform (Source: Parabilis Official Website)
Chemical Modification Modularization: Based on the natural α-helix structure of proteins and a proprietary "stapling" chemical cross-linking technology, Parabilis has built a custom library of over 1,000 non-natural amino acids, combined with hundreds of proprietary linkers. Using a modular chemical assembly approach, the platform achieves diversity that is 20 orders of magnitude higher than natural peptides. While retaining the α-helix’s membrane-penetrating ability, it precisely exposes the side chains of amino acids on the peptide surface to form specific binding with flat intracellular targets, addressing the three major pain points of traditional peptides: "structural instability, low membrane penetration efficiency, and weak binding affinity."
AI Physical Computing Model: The Helicon™ platform integrates advanced AI algorithms with physical computational models to build a proprietary peptide design and screening system. Unlike the traditional drug discovery approach of individual screening and exhaustive trial-and-error, the Helicon™ platform's AI model is based on Parabilis' unique dataset of non-natural amino acids, combined with cutting-edge physical modeling, enabling rapid prediction of key attributes of α-helical peptides formed by different amino acid combinations and various "stapling" methods. These attributes include membrane permeability, target binding affinity, in vivo stability, metabolic half-life, immunogenicity, and ADME properties.
High-Throughput Validation Closed Loop: To ensure the selected candidate drugs have clinical application value, Parabilis has established a comprehensive multi-high-throughput experimental verification system. Using highly automated experimental equipment, it can simultaneously evaluate thousands of α-helical peptide molecules in parallel. The AI model can also continuously optimize prediction accuracy through a self-learning mechanism combined with high-throughput experimental data, forming a closed loop of "experiments generate data → train AI → AI guides design → experimental verification." In addition, the AI model, combined with publicly available biological and patient datasets, further optimizes clinical design, shortens the candidate drug screening cycle, and significantly reduces R&D costs.
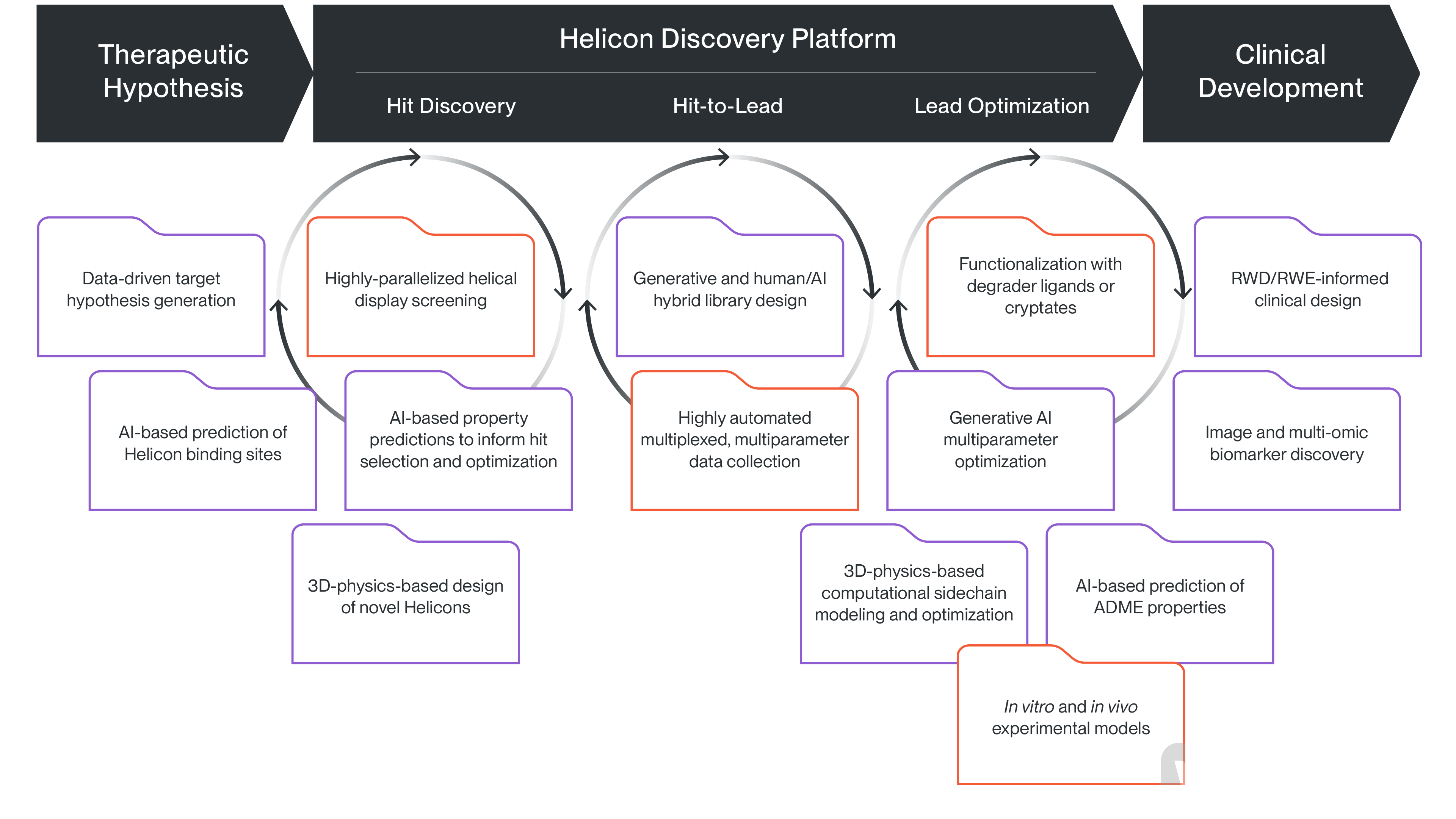 Helicon™ Platform Operation Process (Image Source: Parabilis Official Website)
Helicon™ Platform Operation Process (Image Source: Parabilis Official Website)
4Self-developed Small Molecules Enter Clinical Trials, Collaborative Development of Radioligand Therapy
Leveraging the technical advantages of the Helicon™ platform, Parabilis Medicines, Inc. has currently built a diversified R&D pipeline, covering clinical and preclinical stages, focusing on "undruggable" intracellular targets, and spanning various malignant tumors.
The world's first and only investigational drug directly inhibiting β-catenin:TCF interaction, zolucatetide (FOG-001)
Abnormalities in the Wnt/β-catenin pathway (Wnt/β-catenin signaling pathway, a classic tumor-associated signaling pathway) are key drivers of various solid tumors such as colorectal cancer, hepatocellular carcinoma, gastric cancer, desmoid tumors, and ameloblastoma. However, the interaction between β-catenin and the T-cell factor (TCF, the core transcriptional activation complex of the classic Wnt signaling pathway) family has long been considered an undruggable target.
Zolucatetide, as a competitive inhibitor of β-catenin:TCF interaction, blocks its interaction by directly targeting the core, independent of specific mutation types, thereby shutting down downstream oncogenic signals and fundamentally blocking tumor progression.
Currently, zolucatetide is in Phase 1/2 clinical trials and was granted Fast Track designation and Orphan Drug designation by the FDA for the desmoid tumor indication in November 2025 and March 2026, respectively:
Desmoid Tumor: Among 12 desmoid tumor patients treated with zolucatetide, according to RECIST 1.1 criteria, all evaluable patients (n=10) experienced tumor shrinkage; meanwhile, among those completing more than one post-baseline scan (n=5), an 80% objective response rate (ORR) was observed, with a favorable safety profile and no grade 4/5 treatment-related adverse events.
Adamantinomatous Craniopharyngioma (ACP): Three ACP patients received zolucatetide treatment at dose levels of 144 mg/m² (n=1) and 360 mg/m² (n=2). All patients (n=3) experienced tumor shrinkage, with two patients achieving partial responses, showing tumor volume reductions of 56.0% and 48.0%, respectively; one patient had stable disease, with a tumor volume reduction of 19.2%.
Familial Adenomatous Polyposis (FAP)A patient with FAP and desmoid tumors showed significant improvement after 60 weeks of treatment with zolucatetide; compared to the assessment prior to treatment nearly two years ago, a substantial reduction in both the number and size of polyps was observed, with downstaging, and the diameter of the desmoid tumor decreased by 52.2%.
Others: Monotherapy activity has been observed in hepatocellular carcinoma (HCC), ameloblastoma, salivary gland cancer, and solid pseudopapillary neoplasm (SPN).
Currently, Parabilis Medicines, Inc. has multiple "undruggable" target pipelines in reserve and is building a product matrix of small molecules + nuclear medicine around "stapled peptides."
Multiple Small Molecule Pipeline Reserves
β-catenin degrader (β-catenin degrader): Using Helicon™ technology, a bifunctional degrader approach is employed to bind soluble β-catenin and target it for proteasomal degradation. This can form a complementary mechanism with zolucatetide, focusing on shutting down the Wnt pathway, covering 80%-90% of colorectal cancer cases.
ERG Degrader (ERG Degradation Agent): Targeting ERG fusion proteins (common fusion proteins in prostate cancer), precisely binding to the unique region of ERG, and carrying E3 ligands to specifically degrade ERG through the ubiquitin-proteasome system without affecting structurally similar ETS family proteins; it can target the TMPRSS2-ERG fusion present in approximately 40% of prostate cancers, providing a new treatment option for castration-resistant prostate cancer.
Androgen Receptor Degrader (Non-ligand Binding Site): Targeting the androgen receptor AR (binding to the non-ligand-binding region), bypassing high-frequency drug-resistant mutations in the AR ligand-binding domain, and directly degrading the activated state of AR; addressing the issue of drug resistance in existing treatments for metastatic castration-resistant prostate cancer (mCRPC).
Join Hands with ARTBIO to Create a New Track for Radionuclide + Targeted Therapy
HEARTs (Helicon-enabled alpha radioligand therapies): In collaboration with radiopharmaceutical company ARTBIO, the development of Helicon-enabled α-radioligand therapies (HEARTs) targeting multiple sites is underway. Both parties leverage their respective core technological strengths: Parabilis provides the Helicon™ platform to address target binding challenges, while ARTBIO utilizes the AlphaDirect™ platform to offer radio-ligand development capabilities with the potentially best-in-class isotope lead-212 (212Pb).
Lead-212, with its ideal clinical characteristics such as a short half-life, precise delivery of high energy to tumors, high stability, and compatibility with SPECT/CT imaging, can be combined with the targeting ability of Helicon™ peptides to achieve precise tumor delivery and visualized treatment.
5AI + Protein Design Reshapes Drug Development Paradigm, Chinese and Foreign Enterprises Accelerate to Seize Opportunities
With the development of modern technology, especially artificial intelligence, the deep integration of AI and protein design is sparking an industrial revolution. Many companies around the world have already focused on this area for research, and the industry is accelerating from the technical validation stage toward the value realization stage.
Insilico Medicine (03696.HK) has achieved a full-process closed loop of "AI discovering new targets + AI designing drug molecules" through its Pharma.AI platform. Its core pipeline, Rentosertib (an inhibitor of TNIK, a novel lung fibrosis target discovered by Pharma.AI), significantly reduced R&D costs while accelerating the development process. In 2025, Phase IIa clinical trial data was published in *Nature Medicine*, showing its potential anti-fibrotic and anti-inflammatory effects.
XtalPi (02359.HK) has built a technology platform by applying artificial intelligence and robotics based on vertical data. According to the disclosure on XtalPi’s official website, its PepiX™ AI Integrated Peptide Drug Platform can achieve de novo peptide design without considering whether the target has known protein-protein interactions, and can produce low nanomolar high-affinity peptides within months, significantly improving R&D efficiency compared to traditional models. In February 2026, XtalPi partnered with Yaotang Bio to build an mRNA/CAR-T AI screening platform, aiming to reduce treatment costs while enhancing the efficiency and success rate of drug development.
MoleculeMind, founded by Professor Jinbo Xu, a computational biologist with relevant research in the AI protein field, has recently collaborated with research institutions such as Tianjin University, Hainan University, Southern University of Science and Technology, and Duke University. Through self-developed AI protein algorithms like ComplexDDG, they have successfully doubled the insecticidal efficacy of scorpion toxin LqhαIT. Meanwhile, their self-developed MoleculeOS large model has completed a generational upgrade, integrating the NewOrigin Darwin large model to solve industrial challenges not covered by AlphaFold 3. It can be widely applied in scenarios such as antibody design, enzyme engineering, and peptide optimization.
In the multinational pharmaceutical company camp, leading companies such as Eli Lilly, Sanofi, and Servier have also extended olive branches, rapidly seizing the high ground in the AI + protein design track through large-scale strategic cooperation.
In addition to the $2.75 billion strategic partnership with Insilico Medicine, Eli Lilly also announced a joint investment of $1 billion with NVIDIA to establish an AI drug discovery lab, aiming to shorten the new drug development cycle. Meanwhile, Eli Lilly's self-developed AI platform covers antibody and small molecule drug design, focusing on cancer and neurodegenerative diseases. According to Eli Lilly’s official website, its launched AI drug discovery model possesses proprietary data valued at over $1 billion.
Sanofi Focuses on Autoimmune/Inflammatory Diseases, Announces Up to $2.56 Billion Collaboration with Earendil Labs, a Subsidiary of Huashen Zhiyao, in January 2026 to Discover Novel Bispecific Antibodies Using AI Platforms, Optimize Antibody Design with AI Technology, Advance Peptide Drug Development, and Break Through Traditional R&D Bottlenecks. Servier Deepens Its Commitment to Oncology, Signing a Multi-Year Collaboration with Insilico Medicine in January 2026 Worth Up to $888 Million to Jointly Develop Challenging Target Drugs in the Oncology Field, with Insilico Medicine Responsible for Target and Molecular Design, and Servier Leading Clinical and Commercialization Efforts.
From Challenges to Blue Ocean, from Layout to Breakthrough, the Global Biomedical Industry is Redefining the Boundaries of Treatment through Technological Innovation. In the future, continuous breakthroughs in technologies such as AI-driven drug discovery, protein degradation, and stable peptides may lead to more targets being conquered one by one.
