67% Objective Response Rate, Discontinuation Rate <2%, World's First Targeted Therapy for High-Grade Glioma Receives FDA Breakthrough Therapy Designation
When a BRAF inhibitor no longer just "inhibits" but actively "disrupts" the abnormal activation of cancer-causing signaling pathways, the survival ceiling for brain cancer patients may be rewritten.
Recently, FORE Biotherapeutics, a developer of targeted cancer therapies, announced that its self-developed novel BRAF inhibitor, plixorafenib, has received Breakthrough Therapy Designation from the U.S. Food and Drug Administration (FDA) for the treatment of adult patients with high-grade glioma (HGG) harboring the BRAF V600E mutation.
As a novel oral small-molecule BRAF inhibitor, Plixorafenib exhibits high selectivity for BRAF mutations. This is also the world's first targeted therapy for HGG to receive this designation.
1Dual Mechanism of Dimer Disruption and Paradoxical Destruction to Overcome Limitations of Previous Drugs
Behind the Breakthrough Therapy Designation is the unique molecular mechanism of plixorafenib, which distinguishes it from first- and second-generation BRAF inhibitors.
Before breaking down the mechanism of action of plixorafenib, it is necessary to first understand the impact of the BRAF target on the development of malignant brain tumors.
BRAF is a kinase in the mitogen-activated protein kinase (MAPK) pathway that links growth factor signals to transcriptional changes, thereby regulating cell proliferation, survival, and differentiation. It is the kinase with the highest frequency of mutations in cancer, among which the V600E mutation accounts for 90% of BRAF variants. BRAF V600E occurs in 7% of pediatric primary brain tumors and 4% of adult primary brain tumors, particularly in epithelioid glioblastoma (GBM), pleomorphic xanthoastrocytoma (PXA), and ganglioglioma. In pediatric low-grade gliomas (pLGG), BRAF V600E leads to poor efficacy of conventional therapies and increases the risk of progression to malignant high-grade gliomas (HGG), especially in the context of CDKN2A deletion. Additionally, the BRAF V600E mutation causes constitutive MAPK activation and loss of feedback inhibition, promoting oncogenic transformation.
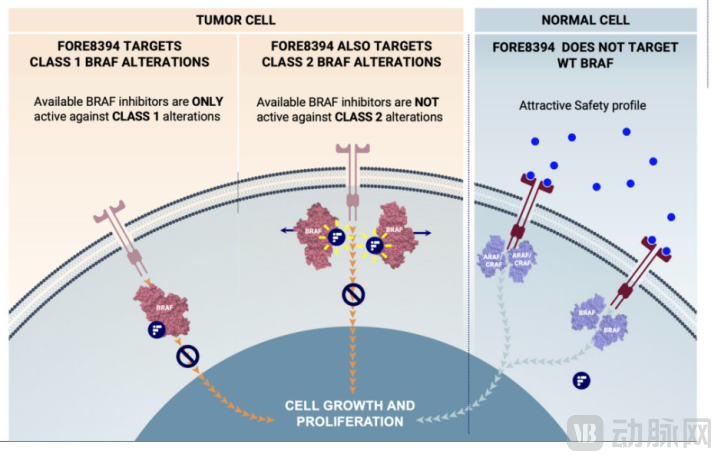
Based on the BRAF mutation being a key actionable driver in the molecularly integrated clinical decision-making paradigm for CNS tumors, the design mechanism of plixorafenib targeted two major limitations of the first two generations of drugs from the outset. First, traditional BRAF inhibitors lack antitumor activity when facing BRAF dimers or Class 2 mutations (including BRAF fusions). Second, "paradoxical activation" triggered by dimer activation—where the drug, while inhibiting mutant BRAF, paradoxically activates wild-type BRAF and its downstream MEK/ERK signaling pathway—transduces extracellular signals into the cell, causing uncontrolled cell growth and division again. They can even amplify the oncogenic activity of certain mutant cells, ultimately leading to treatment resistance."The paradox of cancer recurrence caused by cancer treatment" became the unsolvable dilemma for the first generation of RAF inhibitors.
The core innovation of plixorafenib lies in its dual properties of "dimer-breaking" and "paradox-breaker." As a "paradox-breaker," plixorafenib effectively blocks the formation of BRAF dimers, thereby preventing the abnormal activation of the MAPK pathway caused by traditional RAF inhibitors at the source. Meanwhile, it exhibits high selectivity for both BRAF V600 monomers and BRAF-containing dimers, meaning it can precisely target tumors with the classic BRAF V600 mutation while also addressing BRAF Class 2 variants (including fusions) that are difficult for first-generation drugs to tackle, significantly expanding the scope of patients who can benefit.
Source: Fore Bio official website
Overall, as a "paradoxical disruptor," plixorafenib does not induce paradoxical activation of the RAF/MEK/ERK pathway, disrupts BRAF dimerization, thereby blocking the growth and proliferation of tumor cells, while avoiding paradoxical activation and circumventing some mechanisms of acquired resistance, demonstrating better safety and more durable efficacy compared to previous-generation RAF inhibitors.
2Objective response rate reached 67%, clinical benefit rate exceeded 75%, and discontinuation rate was less than 2%.
The FDA granted this designation primarily based on two core clinical studies of plixorafenib: the completed Phase 1/2a clinical trial (treatment data from approximately 25 patients) and the ongoing FORTE basket trial.
First, look at the Phase 1/2a clinical trial. The data shows that in the pre-specified subgroup of refractory, MAPK inhibitor-naïve BRAF V600-mutant primary CNS tumor patients, plixorafenib achieved a 67% objective response rate (ORR), with a clinical benefit rate exceeding 75%.
A more straightforward understanding is that the emergence of plixorafenib means that 67% of patients experienced significant tumor shrinkage or complete disappearance, and 75% of patients had no further tumor progression, achieving either volume reduction or stabilization.
Another set of data further validates the clinical efficacy of plixorafenib. In a broader patient population with all V600 mutations who have not been treated with MAPK inhibitors, plixorafenib achieved a 42% response rate and durable duration of response, with a median DOR of 17.8 months and a clinical benefit rate exceeding 70%. Additionally, in terms of safety, compared to existing standard treatment regimens for various BRAF-mutant tumors, plixorafenib demonstrates favorable safety and tolerability across different tumor types.The discontinuation rate due to drug-related adverse events was less than 2%., patient compliance has significantly improved.
Another study that helped plixorafenib secure the FDA Breakthrough Therapy Designation is the global Phase 2 clinical trial named FORTE. This trial employs a basket study design with four sub-protocols to evaluate the application of plixorafenib across different cancer patient populations. The three single-agent indications currently being assessed include: recurrent or progressive BRAF V600 primary CNS tumors, solid tumors with BRAF fusions, and rare BRAF V600 mutant solid tumors. The CNS tumor types encompass HGG, low-grade glioma (LGG), as well as other primary brain and spinal cord tumors in adults and children.
According to the interim analysis data released by FORE in the third quarter of 2025, the BRAF V600E primary CNS basket study of the FORTE trial has met the pre-specified interim analysis criteria, further validating the potent anti-tumor activity of plixorafenib across various BRAF-mutant tumor types and CNS histologies.
The Independent Data Monitoring Committee (IDMC) supports the continuation of the FORTE study as planned, based on the response data assessed by Blinded Independent Central Review (BICR). However, the IDMC will continue to monitor safety. After the preliminary analysis of positive results from the CNS basket study is completed, FORTE will submit a New Drug Application for plixorafenib to the U.S. FDA under the accelerated approval pathway.
Notably, positive clinical data has led to further regulatory milestones.Plixorafenib has previously received Fast Track designation for the treatment of cancer patients with BRAF Class 1 (V600) and Class 2 (including fusions) mutations who have exhausted prior therapies, as well as Orphan Drug designation for the treatment of primary brain and CNS malignant tumors.
The label "orphan drug" is backed by a set of harsh figures: according to research data from an article in the *International Journal of Surgery* journal, from 1990 to 2021, the global prevalence and incidence rates of central nervous system cancers increased by 124.50% and 106.53%, respectively, with corresponding rises in age-standardized incidence rates (ASR). By 2035, the estimated number of prevalent cases, new cases, deaths, and DALYs globally will be 1.22 million, 430,000, 310,000, and 10.27 million, respectively, marking increases of 24%, 19%, 19%, and 15% from 2021 to 2035. The severe situation of rising incidence and persistently high mortality will continue long-term. This designation granted by the FDA means that Plixorafenib has already obtained "priority review rights" for these two indications.
3Monotherapy Breaks the Dilemma of Efficacy and Side Effects, Safer and More Tolerable
In the treatment journey of malignant tumors such as primary brain tumors and gliomas, traditional chemotherapy and radiotherapy often force patients to make a difficult trade-off between efficacy and side effects due to their "kill a thousand enemies at the cost of eight hundred allies" limitations. The emergence of targeted therapy has opened a new window for long-term survival for cancer patients. The recently approved combination of Dabrafenib and Trametinib—a "golden duo" consisting of a BRAF inhibitor and a MEK inhibitor—is a highly representative example of targeted combination therapy. However, the maintenance of efficacy heavily relies on the combined use of both drugs, which means facing higher toxicity and more complex adverse reaction management. Moreover, most BRAF V600E mutant gliomas cannot be treated with BRAFi+MEKi, and residual tumors may lead to rebound, progression, drug resistance, and high mortality.
High-grade gliomas are aggressive primary brain tumors, and "keeping it under control" is already the most basic, yet also the most heart-wrenching, demand for patients with this condition. The treatment of this cancer is highly challenging, characterized by a high disability rate and a high recurrence rate. As a result, despite adopting multimodal treatments, patient prognosis remains poor. In addition to limited survival prospects, patients also endure immense suffering caused by the disease itself and the toxicity of current treatments. There is an urgent need in clinical practice for novel therapies that are both effective and better tolerated.
Therefore, from a clinical perspective, plixorafenib, with its unique mechanism of action targeting BRAF, offers another solution to the current treatment challenges. While precisely attacking the lesion as a single-agent therapy, it avoids the limitations of previous BRAF inhibitors, such as rapid disease recurrence and the need to be combined with MEK inhibitors, thereby providing patients with a more "straightforward," better-tolerated, and quality-of-life enhancing treatment option.
4Triple Boost from Fast Track, Orphan Drug, and Breakthrough Therapy Designations Accelerates Commercialization
From fast track to orphan drug, and now to breakthrough therapy, plixorafenib is gradually building a complete regulatory incentive map, which is not uncommon in the new drug development cycle. How to capitalize on this wave of policy dividends is the core proposition that plixorafenib needs to address next.
Currently, RAF inhibitors have evolved to the third generation, with tovorafenib, a representative of the third-generation RAF inhibitors, already approved and on the market. This means that if plixorafenib’s development process and FDA approval proceed smoothly, it will enter a market that has already been validated, offering relatively clear future commercial returns.
With the breakthrough designation as a trump card, plixorafenib can enjoy intensive guidance from senior FDA reviewers and is eligible for rolling review and priority review, significantly shortening the time to market. How plixorafenib writes the story of a new generation of RAF inhibitors is worth looking forward to.
