China's first domestically developed carbon dioxide contrast injection device has been approved, and AMT Medical has completed a Series D financing round of over RMB 200 million
AMT Medical has recently completed a Series D financing round exceeding RMB 200 million. This round was led by the Zhesheng Collaborative Life and Health Fund, with participation from the Henan Key Intellectual Property Fund, Global Hedge Fund Management, and other investors. Following the completion of this financing round, AMT Medical's post-investment valuation has surpassed RMB 1.5 billion.
According to AMT Medical, the funds raised will primarily be allocated to the research and development of its core product line of 3D-printed biodegradable vascular stents, as well as related registration and certification efforts in both Chinese and international markets. Additionally, a portion of the financing will be directed toward the market promotion of its recently launched carbon dioxide contrast injection device in China.
Driven by multiple factors such as population aging, the number of patients with cardiovascular diseases is growing rapidly. In China alone, according to the Annual Report on Cardiovascular Health and Diseases in China 2024 by the National Center for Cardiovascular Diseases, the total number of prevalent cardiovascular disease cases in the country has reached 330 million. This includes 13 million stroke patients, 11.39 million patients with coronary heart disease, 45.3 million patients with peripheral artery disease, and 245 million patients with hypertension. Furthermore, the 2024 China Health Statistics Yearbook shows that cardiovascular diseases still rank first in the composition of causes of death among both urban and rural residents in China.
The rapid advancement of technologies such as minimally invasive interventions, coupled with increased disease awareness among patients, has led to a sharp rise in the demand for pan-vascular interventional diagnosis and treatment. This, in turn, has driven a rapid increase in the demand for medical devices used in diagnostic and treatment services, thereby accelerating the expansion of the pan-vascular interventional device market.
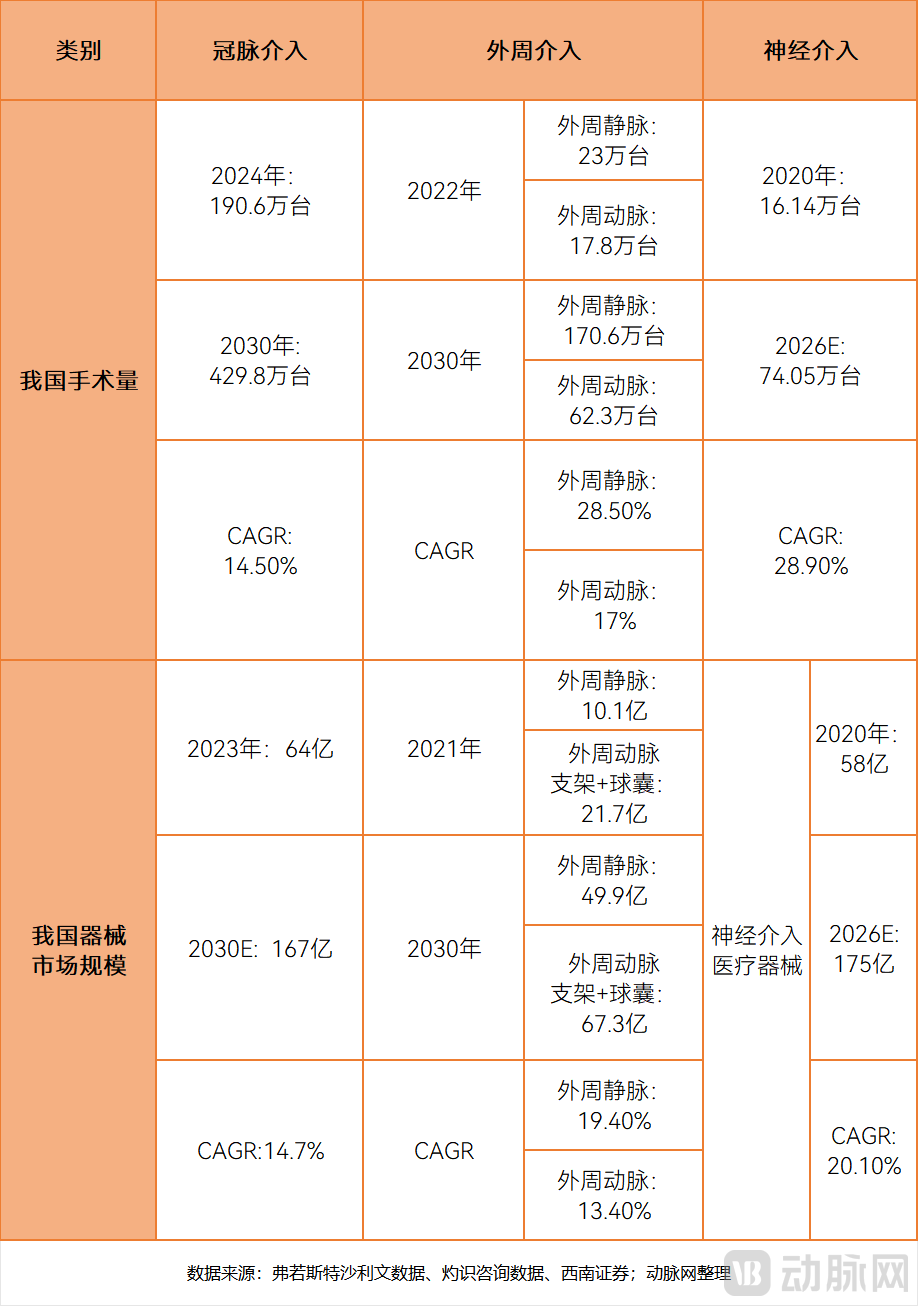 Note: Data from different statistical sources may vary. According to the National Quality Control Center for Cardiovascular System Diseases, the number of PCI procedures in 2024 is 2.21 million.
Note: Data from different statistical sources may vary. According to the National Quality Control Center for Cardiovascular System Diseases, the number of PCI procedures in 2024 is 2.21 million.
With the continuous advancement of medicine, the concept of "intervention without implant" has become increasingly accepted in clinical practice. For example, in the field of coronary intervention, implant-free products such as drug-coated balloons and biodegradable stents can dilate stenotic vessels while leaving no permanent implants behind, offering patients better treatment options. Moreover, biodegradable stents used in coronary intervention and other markets are currently not covered by centralized procurement in China, which provides innovative companies with broader room for market development and growth.
In view of this, AMT Medical has focused on the pan-vascular intervention field, including cardiovascular, peripheral vascular, and neurovascular areas, and has developed a range of biodegradable stents and related interventional and implantable auxiliary devices. To date, AMT Medical has obtained five medical device registration licenses, including several balloon dilatation catheter products, a domestically pioneered stent positioner designed to provide precise positioning for stent implantation in aortocoronary ostial lesions, and a domestically pioneered carbon dioxide contrast injection device. The products currently under rapid development primarily include second-generation fully absorbable rapamycin drug-eluting coronary stents (made of 3D-printed PLLA material, for cardiovascular intervention), and fully absorbable paclitaxel drug-eluting superficial femoral artery stents (made of 3D-printed PLCL material, for peripheral vascular intervention). Both have been granted access to the Innovative Medical Device Special Review and Approval Process and are currently in clinical trials or under regulatory application. In the field of neurovascular intervention, AMT Medical has also planned the only investigational biodegradable intracranial stent product in China.

It is particularly worth mentioning that AMT Medical has recently obtained approval for the Jetfall® Carbon Dioxide Contrast Pressure Injection Set, a Class III innovative medical device that is a domestic pioneer in China. Currently, iodinated contrast agents are commonly used for angiography in clinical practice. However, patients with iodine allergy, renal insufficiency, or thyroid diseases face contraindications to their use. As a result, some patients are unable to undergo interventional procedures. In contrast, the carbon dioxide contrast injection device enables the acquisition of clear images via DSA. Carbon dioxide is excreted through the lungs after circulating in the bloodstream, without causing gas embolism. It leaves no residue in the patient's body, has no hepatorenal toxicity, and is non-allergenic.
It is reported that AMT Medical has established partnerships with distributors across China and has secured tens of millions of RMB in prepayments. It is anticipated that with the launch of this carbon dioxide contrast injection device, the domestic angiography market in China is poised for a round of technological product substitution.
It is worth noting that the Jetfall® Carbon Dioxide Contrast Pressure Injection Set, as a product that entered the NMPA Innovative Medical Device Review Channel, received substantial support from the Center For Medical Device Evaluation . NMPA, the Beijing Municipal Medical Products Administration, and other agencies throughout the registration and application process. Relevant authorities assigned dedicated personnel to provide one-on-one responses to the company's inquiries during the application process, enabling the product to obtain its registration certificate in just nine months from the date of application acceptance. This efficient review and approval process fully reflects the strong encouragement and substantive support provided by national regulatory authorities for independently developed and innovative products of domestic medical device companies.
In overseas markets, AMT Medical had already obtained CE-MDR certification under EU regulations and FDA clearance in the United States prior to NMPA approval, and has also received medical device certifications in multiple countries and regions, including the United Kingdom, Brazil, and Turkey. AMT Medical began overseas sales as early as September last year.

In addition to the pan-vascular intervention field, AMT Medical has also established a presence in non-vascular implantable and interventional products, including biodegradable biliary stent products and medical-grade biodegradable polymer products made of poly(L-lactide-co-caprolactone) (PLCL). On the one hand, AMT Medical recognizes the growth potential of biliary stents; on the other hand, other types of products developed from medical-grade biodegradable polymer materials are expected to further expand the product portfolio of AMT Medical.
As the only enterprise in China with both fully biodegradable coronary stents and fully biodegradable peripheral stents in clinical trials, and the only domestic company capable of processing from raw material pellets directly into finished biodegradable stent products in a single step, AMT Medical relies on its core 3D Multi-Axial Printing (3D-MAP) technology platform.
In terms of materials, AMT Medical received project support from the Ministry of Industry and Information Technology (MIIT) and the National Medical Products Administration (NMPA) in 2023, and was selected as the leading entity for the "Revealing and Leading" initiative (first batch) under the Biomedical Materials Innovation Task, focusing on the development of materials such as PLCL. Its strategic layout in upstream raw materials is expected to enable domestic companies to achieve import substitution of medical-grade biodegradable polymers in the future, providing strong support for the R&D of high-end medical devices in China. PLCL, a novel biodegradable polymer medical material, is synthesized by block copolymerization of PLA and PCL monomers, achieving a molecular-level balance between rigidity and flexibility. PLCL exhibits improved biocompatibility, superior biodegradability, high strength, and high elasticity. It holds promising application prospects in ligament tissue engineering, bone tissue engineering, vascular tissue engineering, and other fields. In the future, AMT Medical is expected to reduce its dependence on imported materials by leveraging its independently developed medical-grade biodegradable polymer materials, achieving autonomous and controllable production and significantly lowering production costs.
In terms of the technology platform, the 3D Multi-Axial Printing platform enables one-step forming of biodegradable stent products. In the past, both domestic and overseas enterprises predominantly employed laser cutting technology, a subtractive manufacturing method requiring multiple steps, which resulted in low material utilization. Moreover, the relatively complex process led to low processing efficiency, low yield, and persistently high costs. Additionally, a large number of technology patents are held by overseas companies, constraining the development of local enterprises. AMT Medical's 3D-MAP platform has addressed the challenges previously encountered in 3D-printed vascular stents. Through additive manufacturing, it significantly improves material utilization, enables the fabrication of structures that laser cutting cannot achieve, achieves one-step forming, and enhances manufacturing efficiency. Furthermore, this platform greatly reduces material trial costs and time costs during the R&D process. At the same time, by circumventing the patent landscape of overseas companies in the laser cutting field, AMT Medical has secured a leading position in technology patents related to the innovative field of medical 3D printing. To date, AMT Medical has obtained multiple invention patents in China, the United States, and Europe in areas such as stent 3D printing methods, structural designs, and production equipment.
It is reported that biodegradable stent products printed using the 3D-MAP platform have demonstrated complete endothelialization within just two weeks in animal studies. In terms of degradation rate, coronary stents degrade on average in approximately three years, while peripheral stents degrade in approximately two and a half years. In clinical trials involving more than 200 patients receiving coronary stents, the longest follow-up period has reached six years, with a stent thrombosis rate of zero.
