One approved, six on the way: is the pVAD market already becoming crowded in its first year of commercialization?

Core Medical
Artificial Heart Series Product Developer

Abiomed
Cardiac System Medical Device Developer

Johnson & Johnson
Medical Device R&D and Manufacturer
The Interventional Artificial Heart (pVAD) is Finally About to be Implemented.
In December 2025, HXCOREMED's interventional artificial heart received approval from the National Medical Products Administration (NMPA) for marketing in China, becoming the first domestically approved product of its kind. It can be used for high-risk percutaneous coronary intervention (PCI), providing minimally invasive and convenient left ventricular assistance to adult patients with severe coronary artery disease, reduced left ventricular ejection fraction, and stable hemodynamics. In February 2026, the product successfully completed its first surgery post-market launch, officially entering the clinical application stage.
Meanwhile, Abiomed's Impella series of interventional artificial hearts has recently been introduced to China through the clinical urgent need channel. Previously, the product had entered the innovative medical device green channel in 2024.
In addition to HXCOREMED and Abiomed, more than ten companies in China have entered the field of interventional artificial hearts. The core products of several companies have completed clinical trials or are about to enter the final stage, and are highly likely to be approved for marketing by 2026.
According to this trend, 2026 is expected to become the "collective approval year" for interventional artificial hearts, with the industry welcoming a window period for scaled implementation.
Why does the market have high expectations for interventional artificial hearts?
Looking at Abiomed, a leading company in the field, over 95% of its revenue comes from the Impella series of interventional artificial heart products. Relying solely on this product, Abiomed's revenue exceeded $1 billion in 2022, making it one of the 14 tenfold pharmaceutical stocks between 2009 and 2019.
At the same time, in 2022, Johnson & Johnson acquired Abiomed for a staggering $16.6 billion. The core asset supporting this hundred-billion-dollar valuation is precisely the Impella series of interventional artificial hearts.
Subsequent performance also proved the correctness of Johnson & Johnson's decision. The sales volume of the Impella series continued to rise, with steady revenue growth: $1.306 billion in fiscal year 2023, $1.496 billion in fiscal year 2024, and climbed to $1.751 billion in fiscal year 2025, demonstrating extremely strong growth resilience.
Abiomed's rapid growth has also brought substantial returns to Johnson & Johnson. Its Q2 2023 earnings report showed that, following the acquisition, Abiomed added $660 million in bottom-line revenue to Johnson & Johnson MedTech. After the quarterly report was released, Johnson & Johnson's stock price surged over 6% in a single day. For the entire year of 2023, Abiomed's performance exceeded Johnson & Johnson's internal model and surpassed the expectations set by Wall Street analysts prior to the deal's completion. In fiscal year 2025, driven by Abiomed and electrophysiology products, Johnson & Johnson's cardiovascular business achieved robust growth of 15.8%, with sales reaching $8.928 billion.
Currently, the clinical application scope of Abiomed's Impella series continues to expand, widely used in scenarios such as cardiogenic shock rescue and high-risk PCI (percutaneous coronary intervention). Globally, the cumulative applications have exceeded 400,000 cases, with annual sales surpassing 1.7 billion US dollars, establishing it as a benchmark product in the field of cardiovascular critical care.
The Impella series can support a 100-billion valuation, achieve tens of billions in annual revenue, and accumulate hundreds of thousands of clinical applications. The core reason lies in the vast market space, and previously, this product was the only one approved globally for a long time, enjoying an exclusive monopoly. Now, with the approval and implementation of domestically produced interventional artificial hearts, this hundred-billion-dollar market pattern is set to be reshaped.
An interventional artificial heart is an interventional ventricular assist device, primarily used in scenarios such as cardiogenic shock and high-risk percutaneous coronary intervention (PCI), providing medium- to short-term ventricular circulatory support for patients with critical cardiovascular conditions.
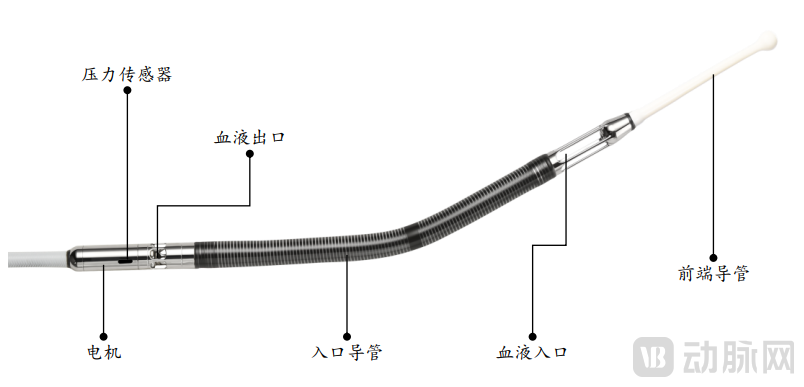
(HXCOREMED Interventional Ventricular Assist System - Catheter Pump Body)
Specifically, when performing surgical treatment in high-risk cardiovascular patients, clinicians can insert an interventional artificial heart through the skin into the main blood vessels and advance it to the heart. The integrated blood pump directly pumps blood from the failing heart into the aorta or pulmonary artery, thereby improving systemic blood flow and perfusion of vital organs.
There is also an implantable artificial heart on the market. However, the implantable artificial heart is mainly aimed at end-stage heart failure patients, designed to help improve symptoms, enhance quality of life, and extend patient survival. Unlike interventional artificial hearts, implantable artificial hearts are inserted into the patient's heart through open-chest surgery, causing greater trauma, providing longer assistance, involving more complex products, and incurring higher costs.
Compared with implantable artificial hearts, interventional artificial hearts correspond to a wider patient population and a larger market space. According to the prospectus of HXCOREMED, in 2024, the number of patients with cardiogenic shock in China was 308,000, and the number of PCI surgeries in China reached 1.906 million, of which about 10-15% are high-risk PCI, indicating broad and urgent clinical needs. According to Frost & Sullivan, the market size of China's medium and short-term artificial heart market is expected to exceed 11 billion RMB by 2033. Meanwhile, the market size of implantable artificial hearts is expected to be 5.7 billion RMB by 2033.
It should be noted that, in addition to interventional artificial hearts, interventional ventricular assist devices also include products such as intra-aortic balloon pump systems (IABP) and extracorporeal membrane oxygenation equipment (ECMO).
Intra-Aortic Balloon Pump (IABP) is a pulsatile pump assist device that can increase cardiac output by 10%-20%. It features easy implantation and a low complication rate, making it one of the most commonly used percutaneous mechanical circulatory support methods in China.
Extracorporeal Membrane Oxygenation (ECMO) is an extracorporeal life support technology that significantly enhances cardiac output but increases left ventricular afterload, does not markedly improve coronary perfusion, and is complex to operate. It is suitable for emergency situations such as heart failure combined with respiratory failure or cardiac arrest.
As an innovative medical device that the market expects more, the percutaneous ventricular assist device (pVAD) can provide larger flow support such as 2.5 liters, 3.5 liters, and 5 liters. Moreover, this product can reduce the left ventricular afterload and effectively increase coronary perfusion.
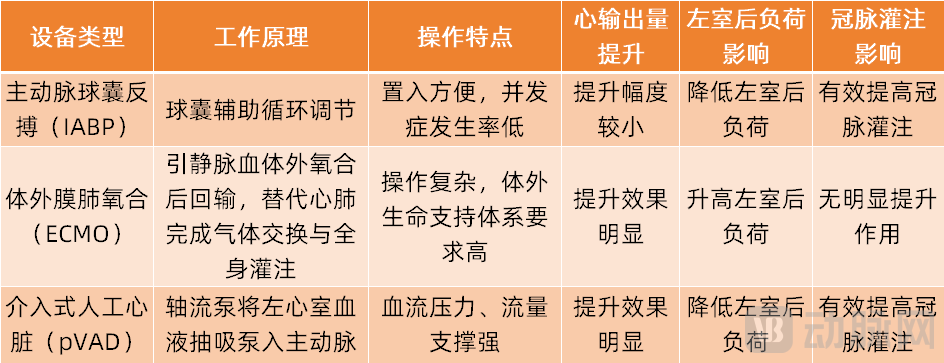
By comparison, pVAD represented by the Impella system is currently the most widely used and fastest-growing interventional ventricular assist device globally. In the field of high-risk PCI patient rescue, pVAD has the most substantial clinical research evidence, and authoritative guidelines also provide clear recommendations.
In 2025, international societies such as ACC/AHA updated and released guidelines for the management of patients with Acute Coronary Syndrome (ACS) based on multiple clinical research findings on pVAD. The guidelines clearly recommend that pVAD is suitable for patients with acute ST-segment elevation myocardial infarction complicated by severe or refractory cardiogenic shock who meet the indications, as well as ACS patients with mechanical complications. It also points out that routine use of IABP or VA-ECMO is not recommended for patients with acute myocardial infarction complicated by cardiogenic shock.
With its solid clinical outcomes, clear guideline recommendations, and substantial patient demand, the percutaneous ventricular assist device (pVAD) is expected to become mainstream in the future hundred-billion-dollar interventional ventricular assist market.
Even for interventional artificial hearts with high technical barriers, more than ten enterprises in China are simultaneously laying out their strategies.
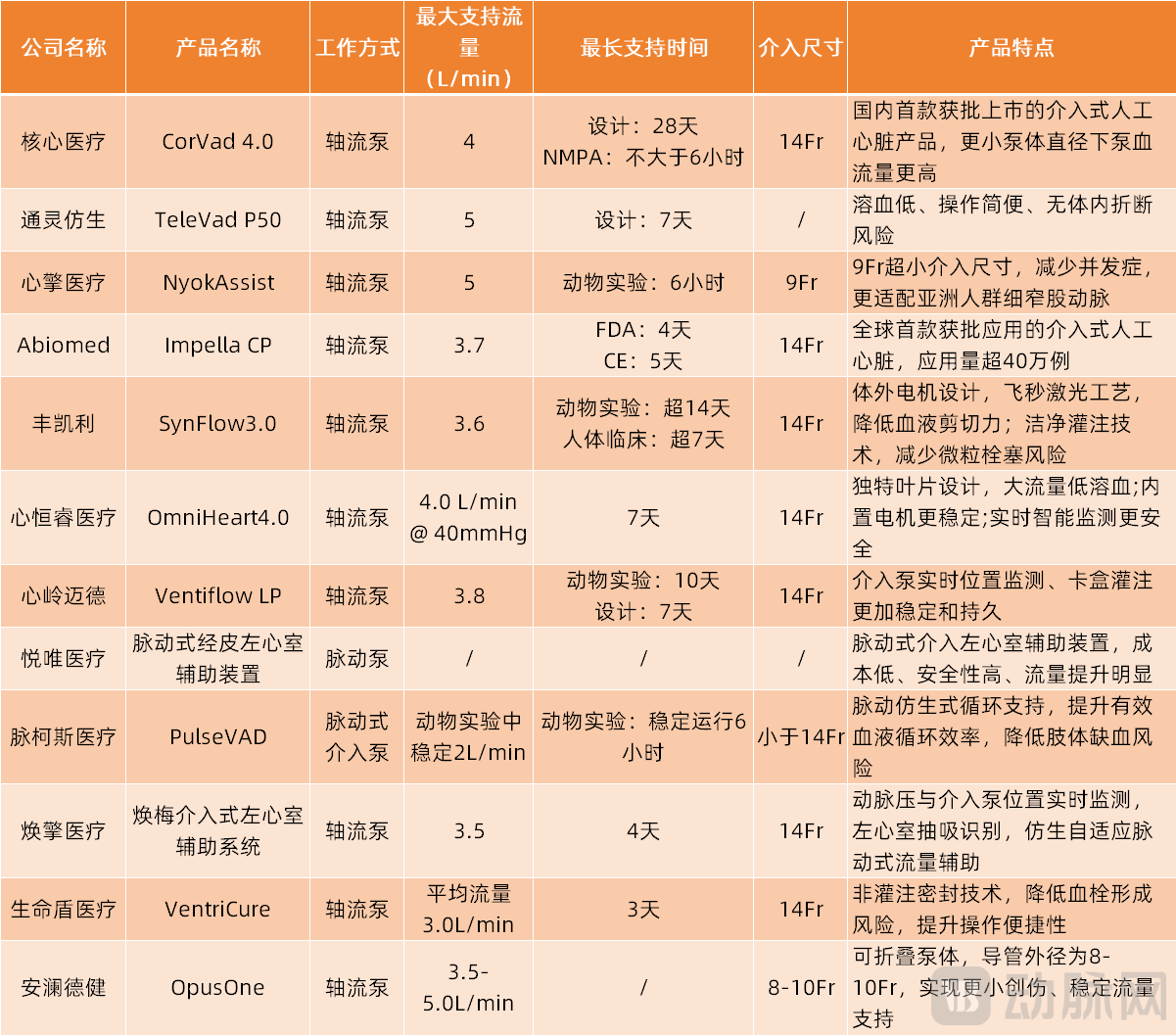
From the above table, the invasive artificial hearts of various companies in China have their own unique features. For instance, HXCOREMED's CorVad is designed to provide support for up to 28 days; magAssist's NyokAssist has an extremely small intervention size of 9Fr; ForQaly® Medical has innovated a clean perfusion technology; Xinhengrui Medical has incorporated intelligent design; and HeartHill's innovative cartridge perfusion system offers greater stability and longevity…
It should be noted that in invasive artificial hearts, higher flow support is not always better. Instead, multiple dimensions such as product size, support duration, and perfusion sealing technology need to be considered to provide tailored solutions for corresponding patients. For instance, the Impella series includes several models such as Impella CP, Impella 5.0, and Impella 2.5, offering different levels of maximum flow support at 3.7L/min, 5L/min, and 2.5L/min respectively, to meet the needs of different patients.
These products have their own unique features because each company has adopted different technical routes and achieved technological innovation in various directions. According to the different positions of the power unit, interventional artificial hearts can be divided into two types: built-in motor and external motor.
Among them, HXCOREMED, Tongling Bionic, Phigine Medical, and HeartHill adopt the built-in motor approach, while magAssist, ForQaly® Medical, and
Enginprime Medical take the external motor approach.
The principle of the motor-integrated route is as follows: The impeller and micro motor of the interventional artificial heart are delivered to the ascending aorta via the femoral artery; a catheter is delivered to the left ventricle, with its inflow port located in the left ventricular outflow tract and its outflow port in the ascending aorta. When the micro axial flow pump at the tip of the catheter operates, the catheter draws blood from the left ventricle and delivers it to the ascending aorta.
This approach requires companies to overcome technical challenges such as miniature motors since the motor is placed inside the body. With the motor positioned close to the impeller, its power transmission efficiency is higher, causing less damage to the myocardium and heart valves. The fluid flow is more stable with fewer vortices, resulting in minimal harm to blood cells, a lower risk of hemolysis and tissue damage, and better support for long-term stable operation within the body. This is also the key reason why HXCOREMED's CorVad can provide support for up to 28 days.
Previously, HXCOREMED, which chose the motor-integrated technology route, released clinical research results on CorVad. Clinical data showed that the CorVad system was successfully placed in 118 (100%) high-risk PCI patients, none of whom experienced hemodynamic impairment. Surgical success was achieved in 116 (98.3%) patients, and acute kidney injury occurred in 14 (11.9%) patients. No severe hemolysis, device malfunction, or life-threatening infections were reported. Compared to baseline, left ventricular ejection fraction significantly increased at 48 hours, before discharge, and 30 days post-PCI.
HXCOREMED also conducted a randomized controlled trial comparing CorVad with ECMO. The data showed that the incidence of adverse events at 30 days was 15.0% in the CorVad group and 29.1% in the ECMO group; at 90 days, the incidence was 15.8% in the CorVad group and 33.9% in the ECMO group. This indicates that for pVAD support during high-risk PCI, CorVad is non-inferior and superior to ECMO in terms of adverse events at both 30 and 90 days.
In addition to HXCOREMED, the classic Impella series products, which have been applied in over 400,000 cases in the market, also use the built-in motor route, a technology that has been extensively validated in clinical practice.
The principle of the external motor route is as follows: the impeller of the interventional artificial heart is placed in the left ventricle, and the power unit (motor) is positioned outside the femoral artery (external to the body). A flexible drive shaft connects the power unit and the impeller. When the device operates, the catheter draws blood from the left ventricle into the aorta. This technical route significantly reduces the cost of the motor by placing it outside the body, while also decreasing the size of the interventional artificial heart. For instance, the external motor route adopted by magAssist features its NyokAssist system, which has a size of 9Fr, smaller than the 14Fr of the Impella series.
However, since the motor is placed outside the body, such products require larger impellers to generate sufficient flow support and need reliable drive shafts to connect the power unit with the impeller. This demands that companies overcome technologies like foldable impellers and flexible drive shafts.
Although not yet widely used in clinical practice, products based on this technical route have achieved excellent results in clinical research. For example, data from the randomized controlled study of SynFlow 3.0, a percutaneous transvalvular micro-axial pump by ForQaly® Medical, showed that at 30 days, the incidence of major adverse events was 7.34% in the SynFlow 3.0 group versus 11.5% in the ECMO group, achieving non-inferiority with statistical significance. This suggests that SynFlow 3.0 is non-inferior to ECMO and can be a choice for pVAD in high-risk PCI patients. Moreover, compared with ECMO, SynFlow 3.0 was associated with less intraoperative blood transfusion, shorter hospital stays, and fewer device-related adverse events.
In addition to the differences in the location of the power unit, various enterprises also differ in terms of blood flow support. In the market, most companies choose Impella series products of the same type of micro axial flow pump to provide patients with continuous and stable circulatory support.
Some other companies choose pulsatile pumps or pulsating pumps. This is because research has found that cardiac blood circulation has pulsatile characteristics, and pulsatile pumping is more consistent with the human physiological circulation mechanism. It can better simulate physiological blood perfusion and improve the efficiency of blood flow support.
In response, Yuewei Medical launched the pulsatile percutaneous left ventricular assist device. Pulsevad, the interventional artificial heart from Mcsmed, adopts a pulsatile design. The Huanmei interventional left ventricular assist system from Phigine Medical is equipped with a bionic self-adaptive pulsatile flow assistance function… At the same time, these innovative pulsatile artificial hearts have achieved a small catheter diameter and greater blood flow support. For example, the PulseVAD from Mcsmed has a catheter diameter of less than 14Fr.
Overall, there are already more than 10 domestically produced interventional artificial hearts in China. In the future, who will take the lead in the market still needs to be tested by the market and clinical practice.
Based on the progress of these more than ten interventional artificial hearts, the seven fastest-progressing domestically produced interventional artificial hearts in China have been identified.
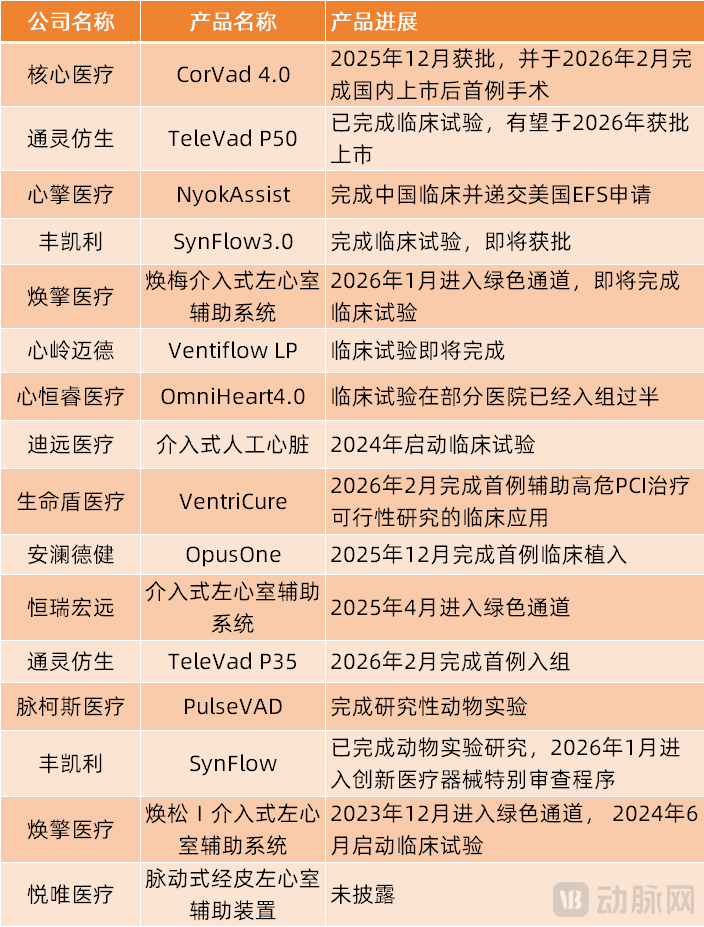
(Progress of Chinese-produced Interventional Artificial Heart)
First is HXCOREMED's CorVad 4.0, the product was approved for marketing by the National Medical Products Administration (NMPA) in December 2025, becoming the first domestically produced product of its kind to receive approval in China, filling the technical gap in interventional cardiac assist devices. In February 2026, the product successfully completed its first surgery post-market launch, officially entering the clinical application stage.
Secondly, products from companies such as Tongling Bionic, magAssist, ForQaly® Medical, Phigine Medical, HeartHill, and Xinhengrui Medical have completed or are about to complete clinical trials, and are expected to receive marketing approval in 2026.
As of now, the above-mentioned seven products are the fastest-progressing interventional artificial hearts in China. Except for HXCOREMED's CorVad 4.0, which has already been approved, the other six are expected to be approved by 2026. This also means that 2026 could become the collective approval year for domestically produced interventional artificial hearts.
Finally, some innovative companies' invasive artificial hearts made progress in 2026. Products such as LifeShield Medical, Enginprime Medical, and TeleVad P35 by Tongling Bionic completed their first clinical trials or feasibility studies for clinical application in 2026. Meanwhile, Hengrui Medical's Hengrui Hongyuan interventional artificial heart and another product by ForQaly® Medical, SynFlow, entered the Innovative Medical Device Green Channel in 2026.
Overall, as domestically produced interventional artificial hearts are accelerating their approval and market entry, high-risk PCI and cardiogenic shock patients in China will have access to higher-quality innovative treatment options. The market landscape for short-term interventional ventricular assist devices is also set to undergo significant changes. VCBeat will continue to monitor how the market evolves.
