After billions of dollars, how will China-produced IL-4Rα play its cards?
In the golden track of IL-4Rα, Dupilumab, a super blockbuster worth tens of billions of dollars, and Stapokibart from the Chinese company Keymed, once stood side by side as two giants. However, this seemingly stable pattern has quietly changed recently — with Akeso's Manfidokimab and 3S Guojian's SSGJ-611 successively filing for market approval, bringing the total number of pending products to five, all targeting the same point.
More notably, in this increasingly fierce race, several innovative drugs and generic drug products have quietly entered Phase III clinical trials. The competition for this target in China has already formed a "1 imported leader + 1 domestically produced already on the market + 5 filed for marketing approval + multiple in late-stage clinical trials" tier structure.
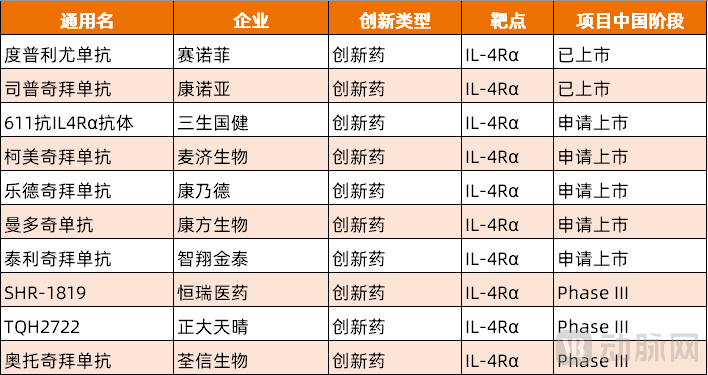
Figure 1. Pipeline of IL-4Rα monoclonal antibody innovative drugs with advanced clinical progress in China (Source: PharmaCube)
In the next two to three years, with more domestically produced drugs entering the market, IL-4Rα monoclonal antibody products in China are likely to face fierce competition. The future competition will be all-encompassing, Dr. Zhang Chenghai, founder of Mabgeek Biotechnology, told VCBeat.
A Chinese domestic "offense-defense" battle, evolving from single-product breakthrough to full-scale confrontation, is already on the verge of being launched.
Sanofi's Dupilumab is the "leader" in the global IL-4Rα field, with annual sales of Dupilumab reaching 15.714 billion euros in 2025. As a precise IL-4Rα inhibitor, Dupilumab comprehensively inhibits the type 2 inflammatory pathway from upstream by blocking the signaling of two core pathogenic factors, IL-4 and IL-13, ultimately achieving the therapeutic goal of controlling the disease, repairing the barrier, and improving symptoms. Dupilumab has currently been approved for the treatment of various type 2 inflammation-related diseases, including atopic dermatitis, prurigo nodularis, asthma, and chronic obstructive pulmonary disease.
Targeting IL-4Rα, pharmaceutical companies in China are actively promoting the commercialization of related products. These products work through cost-effectiveness, differentiated indications, and optimized dosing frequency and other methods to launch full-scale competition.
1) Cost-performance advantage is the core battlefield
As the only domestically produced drug currently approved and marketed in this field, Keymed's Stapokibart has demonstrated a significant cost-performance advantage. According to the price displayed on the JD.com app, compared with Dupilumab (trade name: Dupixent, 1432 yuan/vial), Stapokibart (trade name: Kangyue Da, 1039 yuan/vial) offers a price discount of nearly 30%.
Stapokibart was included in the medical insurance in early 2026, further alleviating the payment pressure on patients. The self-payment for employee insurance will be approximately 208-312 RMB, and for resident insurance, it will be about 416-624 RMB. For the large domestic patient population sensitive to price, especially in the primary healthcare market, cost-effectiveness will become an important factor influencing patient decisions.
2) Find differentiation from Indications
After Keymed's Stapokibart was approved by the NMPA for the treatment of adult patients with moderate to severe atopic dermatitis, it was subsequently approved for two additional indications: chronic rhinosinusitis with nasal polyps and seasonal allergic rhinitis. Stapokibart has also become the world's first IL-4Rα antibody drug for the treatment of seasonal allergic rhinitis.
It is reported that the prevalence of allergic rhinitis in China has exceeded 20%, meaning that one in every five people is affected, with the total number of patients approaching 240 million. Clinical studies have shown, for patients with poor control of conventional treatments (such as intranasal glucocorticoids combined with antihistamines), the addition of Stapokibart can significantly improve nasal symptoms and quality of life. Stapokibart has the characteristic of rapid onset, with significant improvement in nasal congestion symptoms after 2 days of treatment. Moreover, this product can continuously improve nasal symptoms. Compared with the 2-3 year course of immunotherapy, patients using Stapokibart show higher compliance.
Clinical results show that, compared with conventional therapy and immunotherapy, Stapokibart, as a biologic, directly targets the core drivers of type 2 inflammation (IL-4 and IL-13), providing a new precise treatment pathway for patients with moderate to severe allergic rhinitis. By targeting allergic rhinitis, Stapokibart has avoided direct competition with Dupilumab and found a differentiated market for itself.
Genrix Bio's Telikibart has also been submitted for marketing approval, covering multiple indications including adult seasonal allergic rhinitis and adult moderate to severe atopic dermatitis. In addition, the research on telikibart for chronic spontaneous urticaria (CSU) is currently in Phase III clinical trials, and Genrix Bio has similarly positioned telikibart in a differentiated niche market.
CSU is a skin disease characterized by recurrent episodes of urticaria and swelling, significantly impacting patients' quality of life. Currently, in China, antihistamines are the first-line treatment for CSU, with approximately 40%-50% of patients achieving partial or complete relief of symptoms. For patients who do not respond well to antihistamine control, omalizumab, a monoclonal antibody targeting IgE, has become the second-line treatment option for antihistamine-refractory CSU. As the dose of omalizumab is gradually increased, some CSU patients experience only localized symptom relief, and a portion of patients show no response to omalizumab.
Telikibart (anti-IL-4Rα), on the other hand, inhibits type 2 inflammation upstream and is more effective for CSU patients driven by type 2 cytokines such as IL-4 and IL-13. For patients who respond inadequately or are intolerant to omalizumab, telikibart provides a novel therapeutic option and fills the treatment gap for these patients. Telikibart is expected to become the first IL-4Rα-targeted therapy for CSU.
By selecting differentiated indications, Stapokibart and Telikibart have avoided the fiercely competitive atopic dermatitis indication, bringing new therapeutic scenarios for the products.
3) Optimize dosing frequency to improve patient compliance
For adult patients with atopic dermatitis, the initial dose of dupilumab is 600mg (administered as two injections of 300mg each), and the maintenance dose is 300mg every two weeks. If a longer-acting injectable product can be developed, it will improve patient compliance. Currently, domestic companies in China are conducting research in this direction.
SSGJ-611, developed by 3S Guojian, demonstrated promising results in a Phase III clinical trial conducted among Chinese adults with moderate to severe atopic dermatitis. At week 52 of SSGJ-611 maintenance therapy, 96.9% of patients in the short-interval group (300mg, Q2W) and 95.8% in the long-interval group (300mg, Q4W) achieved EASI-75 (at least a 75% improvement in the Eczema Area and Severity Index from baseline).
From the Phase III clinical data, there is no significant difference in the EASI-75 improvement effect between the short-interval group and the long-interval group. The clinical data supports SSGJ-611 maintenance treatment with dosing once every four weeks.
Rademikibart is an IL-4Rα monoclonal antibody developed by Connect Biopharma, which achieved positive results in clinical trials with a once-every-four-week dosing regimen. Mabgeek Biotechnology's Comekibart has directly branded itself as "the world's first long-acting IL-4Rα monoclonal antibody." Comekibart can achieve once-every-four-week dosing, requiring only 13 injections per year, halving both the injection frequency and dosage compared to drugs targeting the same pathway, such as Dupilumab, significantly improving medication adherence.
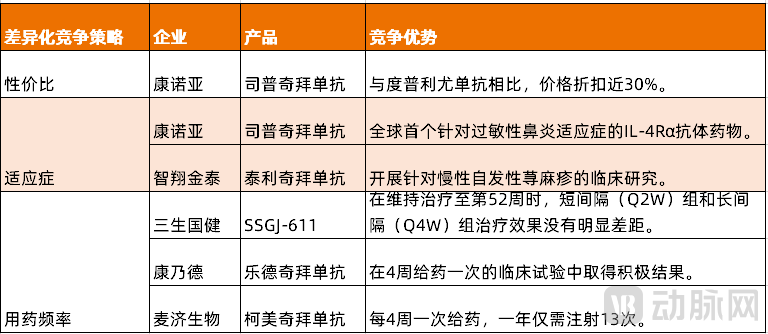
Figure 2. Differentiated Competition Strategies of IL-4Rα Monoclonal Antibodies in China (Source: VCBeat)
According to Dr. Zhang Chenghai, founder of Mabgeek Biotechnology, Comekibart not only exhibits excellent druggability and high bioavailability, but its Fc segment has also been engineered to further enhance the recycling of the antibody drug in the body. This structural improvement allows Comekibart to have a half-life twice as long as similar products, which is key to achieving its long-acting effect.
Currently, in the medication phase, patients tend to extend the dosing interval during the maintenance stage based on their medication habits or financial pressure, often at the cost of reduced efficacy. The long-acting design of Comekibart meets clinical needs by extending the dosing interval without compromising efficacy or increasing the single-dose amount. Dr. Zhang Chenghai believes that with the ongoing development of IL-4Rα monoclonal antibodies in China, products from various companies will gradually cover more indications. In this scenario, the gap in indications among different companies' products will gradually narrow. Indication coverage is only a matter of time, not availability. Meanwhile, reducing dosing frequency is the most powerful way to enhance the cost-effectiveness of a product. Ultimately, what will be compared in this field is how well each company can address patients' clinical needs through superior product performance and commercialization capabilities.
Facing the increasingly fierce competition in the IL-4Rα monoclonal antibody field, some innovative pharmaceutical companies have begun to proactively lay out plans for bispecific antibody products.
As the "leader" in this field, Sanofi has further developed bispecific antibody products. Currently, Sanofi's TSLP/IL-13 bispecific antibody Lunsekimig has entered Phase III clinical trials for COPD indications and is expected to break the efficacy ceiling of the Type 2 inflammation pathway. Compared with the IL-4Rα target, TSLP is located more upstream in the inflammatory response and is mainly released by damaged epithelial cells (such as those in the skin and airways). IL-4Rα is the core driver of Type 2 inflammation, while TSLP acts as the upstream "alarm." Therefore, a bispecific antibody targeting both TSLP and IL-4Rα can more comprehensively block the Type 2 inflammation pathway at both the "initiation" and "execution" levels.
Currently, Lunsekimig is exploring multiple indications, including asthma, atopic dermatitis, and COPD.
Chinese companies have also adopted similar development strategies, upgrading and iterating new generations of bispecific antibody products based on IL-4Rα monoclonal antibodies. Currently, CM512 from Keymed and AK139 from Akeso are both in Phase II clinical trials in China.
1) CM512, Keymed's TSLP/IL-13 bispecific antibody product, has a half-life of up to 70 days.
As the first self-developed IL-4Rα monoclonal antibody in China, the marketed Stapokibart demonstrates Keymed's R&D capabilities. Based on the company's core R&D platform, Keymed has further iterated to develop CM512, the world's first long-acting TSLP/IL-13 dual blocker. CM512 can simultaneously target TSLP and IL-13, effectively reducing Th2 cell differentiation and the release of cytokines such as IL-13, thereby treating type 2 inflammation-related diseases more efficiently.
CM512 demonstrated rapid disease control in patients with moderate-to-severe atopic dermatitis. At Week 6 after the first dose, 50% of patients in the 300 mg group (the proposed clinical recommended dose) achieved EASI-75; by Week 12, the EASI-75 response rate in the 300 mg dose group further increased to 58.3%.
More importantly, CM512 has a half-life of up to 70 days, far exceeding Sanofi's bispecific antibody product Lunsekimig (around 10 days). In Phase I clinical trials, CM512 demonstrated good safety and tolerability, with the overall incidence of treatment-emergent adverse events (TEAE) and no serious adverse events (SAE) reported being comparable to the placebo group.
2) Akeso's IL-4R/ST2 bispecific antibody product AK139 shows good safety.
Based on the development of Manfidokimab monoclonal antibody, Akeso Biopharma has further upgraded and iterated to create the IL-4R/ST2 bispecific antibody product AK139. IL-33, as an alarmin, binds to its specific receptor ST2, activating downstream signaling pathways and triggering multi-system inflammatory responses. AK139 simultaneously targets IL-4R and ST2, covering multiple key upstream and downstream factors related to Th2-type inflammation and non-Th2-type inflammation.
Currently, AK139 has entered Phase II clinical trials, covering multiple indications including chronic obstructive pulmonary disease (COPD), severe asthma, chronic spontaneous urticaria, allergic rhinitis, chronic rhinosinusitis with nasal polyps, moderate to severe atopic dermatitis, and prurigo nodularis. In the clinical Phase I dose escalation, the product demonstrated good safety. In addition, AK139 also adopts a design to extend the half-life, which can improve patient compliance.
Although bispecific antibody products under research have shown better clinical efficacy and longer half-life, whether these products will be approved for marketing and their future commercial prospects remain uncertain. After all, based on existing monoclonal antibody products, the market has higher expectations for bispecific antibody products: improved efficacy, longer dosing intervals, and better safety.
However, the current clinical sample size of bispecific antibody products under research is still relatively small, making it difficult to prove their safety and efficacy on a larger scale. Compared with dupilumab, which has been fully recognized for its safety (approved for pediatric indications) and has accumulated effective data from over 1 million patients globally, the commercial prospects of bispecific antibody products face significant uncertainty.
In the future, as more IL-4Rα monoclonal antibodies are approved for marketing, competition in this field will evolve from the current comparison of single advantages such as indications, cost-effectiveness, and dosing intervals to a stage of comprehensive competition. In addition, bispecific antibody products will also provide patients with more options.
For patients, different treatment options may be adopted at different stages of treatment: In the initial treatment stage, controlling itching is the core demand, and patients can use Dupilumab to improve symptoms; during the maintenance phase, patients may opt for monoclonal antibody products that are slightly less effective but have longer dosing intervals and better patient compliance; for those who do not respond well to monoclonal antibody drugs, bispecific antibody drugs are expected to achieve better clinical outcomes.
For enterprises, the IL-4Rα monoclonal antibody is merely an entry ticket; the real competition lies in the depth and breadth of subsequent pipelines—namely, how each company builds a moat in the field of autoimmune diseases through the IL-4Rα monoclonal antibody and provides patients with more treatment options.
Beyond the product level, commercialization capability is also an important breakthrough for domestically produced enterprises. Pharmaceutical companies in China can seek opportunities for their products in the global market. Currently, the only IL-4Rα monoclonal antibody approved for marketing in overseas markets is Dupilumab. By expanding into overseas markets, companies can break through the intense competition faced by IL-4Rα monoclonal antibodies within China and seek new growth opportunities abroad.
