Hengrui’s NewCo Kailera files for Nasdaq IPO with 4 GLP-1 assets, charting a new paradigm for China’s “system going global”

Hengrui Pharma
Innovative and High-Quality Pharmaceutical Developer

Kailera Therapeutics
Developer of Obesity and Related Disease Therapies
On March 27, 2026, the U.S. Securities and Exchange Commission (SEC) updated the S-1 filing on its official website, revealing that Kailera Therapeutics, a biotechnology company established less than two years ago, is set to launch its Nasdaq IPO, bringing the global GLP-1 assets of Hengrui Pharma to the public market. Kailera operates without its own laboratories or early-stage R&D pipeline; its core assets are exclusively licensed from Hengrui Pharma on an overseas basis. Within 18 months of its founding, Kailera completed $1 billion in financing and has advanced a dual-target weight-loss drug into global Phase III clinical trials, aiming to compete in the global weight-loss drug market dominated by Eli Lilly and Novo Nordisk.
This marks the first time in the history of Chinese innovative drug development that a high-value clinical-stage asset, already validated in China, has been incorporated into a U.S.-based capital structure to leverage global funding for late-stage development and commercialization. Kailera's IPO represents not only its own public listing but also a critical validation of Hengrui Pharma's "NewCo out-licensing" model, as well as an important exploration in the evolution of Chinese innovative drugs—from "product going global" to a more integrated "asset, capital, and talent ecosystem going global."
1Hengrui Pharma Paves the Way with Authorization, Top Capital Accelerates Clinical Progress with $1 Billion Investment
The starting point was in May 2024, when Hengrui Pharma made a pivotal and far-reaching decision: it exclusively licensed the global development, manufacturing, and commercialization rights (outside of Greater China) for four core GLP-1 assets to a newly established company then known as Hercules CM NewCo, which is now Kailera Therapeutics. Under the agreement, Hengrui received an upfront payment of $110 million, up to $200 million in clinical development and regulatory milestone payments, and up to $5.725 billion in sales milestone payments, while retaining a 19.9% equity stake in Kailera. Following the agreement, Kailera quickly completed its capital structuring, securing $400 million in a Series A financing round in October 2024, with participation from top-tier investors such as Bain Capital and RTW Investments. In October 2025, it raised another $600 million in a Series B round, ranking as the second-largest financing in the global pharmaceutical sector that year. Backed by this capital, Kailera initiated its global Phase III clinical program, KaiNETIC, from late 2025 to early 2026, with a total planned enrollment of over 4,700 patients. Its prospectus clearly defines its core positioning—a focused approach to obesity treatment, without complex target narratives, straightforward and determined: leveraging Hengrui's assets to capture value in the global obesity market.
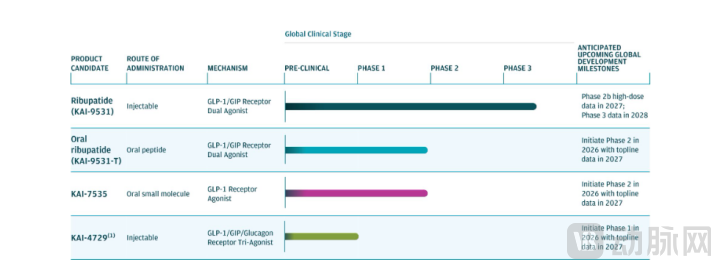
Kailera's competitive edge stems from the four GLP-1 assets licensed from Hengrui, forming a portfolio that includes a dual-target injectable, a dual-target oral, a single-target oral, and a triple-target injectable, enabling a tiered, full-course coverage of obesity treatment. Among them, Ribupatide (KAI-9531) is a once-weekly injectable GLP-1/GIP dual agonist, with three times the GLP-1 receptor affinity of Eli Lilly's tirzepatide and 0.5 times the GIP receptor affinity of tirzepatide, and a half-life of approximately seven days. According to the prospectus, the 6 mg dose group in a Phase III trial in China achieved an average weight loss of 19.2% (evaluated by efficacy assessment) and 17.7% (evaluated by treatment policy assessment). In a Phase II trial in China, the 8 mg dose resulted in a 23.6% weight loss over 12 weeks with almost no plateau observed. Currently, three global Phase III trials under the KaiNETIC program have been initiated, covering three populations: BMI ≥ 30, BMI ≥ 27 with diabetes, and BMI ≥ 35, with the highest dose at 10 mg. The third Phase III trial, KaiNETIC-3, includes a randomized, open-label arm for head-to-head comparison against semaglutide 2.4 mg. The company plans to release high-dose Phase II data in 2027 and global Phase III topline data in 2028—a pivotal milestone that will determine Kailera's future.
 Source: Kailera Therapeutics' IPO Prospectus
Source: Kailera Therapeutics' IPO Prospectus
Ribupatide (KAI-9531-T) is a once-daily oral formulation. In a Phase II trial in China, the 50 mg dose achieved a 12.1% weight loss over 26 weeks, with vomiting and nausea rates of 7.5% and 20%, respectively, demonstrating better tolerability than existing oral GLP-1 drugs. A global Phase II clinical trial is planned to initiate in 2026, with topline data expected in 2027.
KAI-7535 is a once-daily oral, single-target small molecule drug. In a Phase II trial in China, the 180 mg dose achieved a 9.5% weight loss over 36 weeks. Its U.S. IND has been approved, and a Phase II clinical trial will be conducted in 2026. The triple-target drug KAI-4729 (GLP-1/GIP/glucagon) is positioned against Eli Lilly's retatrutide. In vitro data show its GLP-1 affinity is 1.6 times that of retatrutide, with a dual focus on weight loss and reduction of liver fat. A Phase I clinical trial in the U.S. is planned for 2026.
This pipeline strategy creates a full-spectrum coverage model: injectable dual-target agents for severe obesity, oral formulations for mild obesity, and triple-target agents for treatment-resistant obesity—precisely addressing the core gaps in the global obesity market.
2Targeting the Global Obesity Market Dividend, Differentiated Positioning for Breakthrough
Behind Kailera's ambition lies the substantial opportunity presented by the global obesity market. According to its prospectus, the global obese population exceeds one billion, accounting for approximately one-eighth of the world's population. Among this group, the population with a BMI ≥ 35 is growing the fastest; it is projected that by 2030, half of obese adults in the United States will fall into this category. The global prevalence of obesity is expected to rise from 13% of the world's population in 2022 to 25% by 2035, representing 1.9 billion people. Although GLP-1 class drugs have transformed the obesity treatment landscape, unmet needs remain in the market. In Eli Lilly's SURMOUNT-1 Phase III clinical trial for tirzepatide, among subjects with a baseline BMI ≥ 35 who received the highest dose of tirzepatide for 72 weeks, 68% remained in the obese category. Greater weight loss efficacy, improved safety profiles, and more convenient dosing regimens remain core market demands.
The global weight-loss drug market is currently highly competitive, with major market share held by Eli Lilly's tirzepatide and retatrutide, Novo Nordisk's semaglutide and liraglutide, and exenatide, co-developed by Bristol-Myers Squibb and AstraZeneca. Kailera's strategic positioning is clear: achieving superior efficacy with comparable tolerability in the dual-target space, ranking among the top tier of oral agents in terms of data, and establishing a second growth trajectory through its triple-target candidate. Leveraging clinical data already validated by Hengrui, Kailera aims to break through in a market dominated by established players.
3No Revenue in Clinical Stage Yet, IPO to Support Later Development
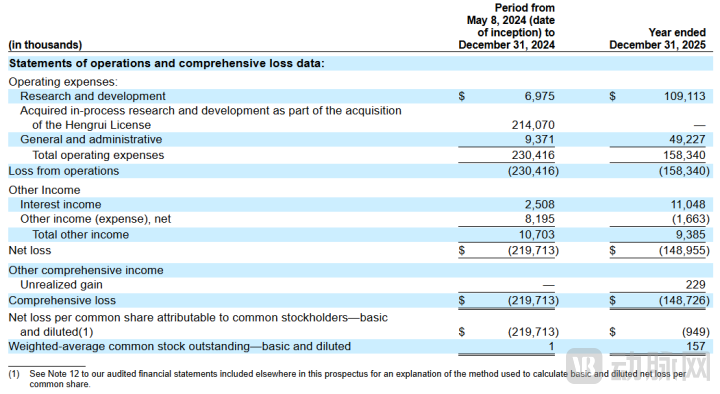
According to the financial data disclosed in its prospectus, Kailera has not yet generated revenue and remains in a pure clinical-stage development phase. From May 8, 2024, to December 31, 2024, Kailera reported research and development expenses of $6.975 million, general and administrative expenses of $9.371 million, and acquired in-process research and development expenses of $214 million, bringing total operating expenses to $230 million, with a net loss of $220 million. For the full year 2025, research and development expenses increased to $109 million, accounting for nearly 70% of total expenses, while general and administrative expenses reached $49.227 million. Total operating expenses amounted to $158 million, with a net loss of $149 million, resulting in an accumulated deficit of $367 million. As of the filing date of the prospectus, Kailera held approximately $653 million in cash and marketable securities, having completed $1 billion in private financing prior to the IPO. The primary purpose of this IPO is to provide funding support for the advancement of global Phase III clinical trials, the development of oral pipeline candidates, and the exploration of triple-target therapies. Whether the company's continued losses will improve and when profitability may be achieved remain to be seen.
 Source: Kailera Therapeutics Prospectus
Source: Kailera Therapeutics Prospectus
The rapid completion of financing and clinical advancement would not have been possible without Kailera's top-tier management team and strong lineup of institutional shareholders. Kailera's CEO, Ronald C. Renaud Jr., previously served as CEO of Cerevel, Translate Bio, and Idenix, all of which were acquired by major pharmaceutical companies, bringing an international perspective on strategic integration at asset value inflection points. CMO Scott Wasserman formerly served as Global Head of Metabolism at Amgen, possessing deep expertise in the clinical development logic of metabolic disease drugs. CCO Jamie Coleman previously led brand operations for Eli Lilly's Zepbound and Trulicity, bringing extensive commercialization experience. CFO Douglas Pagán previously served as CFO of Dicerna, with deep expertise in capital operations within the biopharmaceutical industry. On the institutional shareholder front, top-tier investors including Bain Capital Life Sciences, RTW Investments, Atlas Venture, and the Canada Pension Plan Investment Board (CPPIB) have all established positions. According to the prospectus, Hengrui Pharma held a 13.6% equity stake in Kailera prior to its listing, providing dual strategic and financial shareholder support.
Among these, Hengrui stands as the biggest beneficiary of this out-licensing strategy—in addition to the $110 million upfront payment, it is eligible for up to $200 million in clinical and regulatory milestone payments, up to $5.725 billion in sales milestone payments, and tiered royalties ranging from low single digits to low double digits. Furthermore, through its 13.6% equity stake, Hengrui will continue to share in the benefits of Kailera's global commercialization. This represents the most successful overseas asset monetization in the history of Chinese innovative drug development, breaking the traditional model of simply "exchanging rights for cash" and achieving a multi-faceted return structure combining equity, cash, and milestone payments.
4Providing a "Nasdaq Model" for China's Innovative Drugs: Moving from "Product" to "System"
Despite its well-established positioning and strong capital backing, Kailera's future development faces several key challenges. The core challenge lies in its heavy reliance on Ribupatide. Should the global Phase III clinical trial for this candidate fail to meet expectations, it would have a material impact on Kailera's corporate value. Additionally, the pivotal clinical data supporting its pipeline are derived entirely from trials conducted in China, introducing uncertainty regarding the level of acceptance by regulatory authorities such as the FDA and European regulators, which could affect the drug's approval and launch in Western markets.
On the operational front, all of Kailera's core pipeline assets are licensed from Hengrui Pharma, creating a degree of dependence on the licensor in terms of intellectual property and supply chain. At the same time, competition in the global weight-loss drug market is intensifying. The promotional and pricing strategies of established players such as Eli Lilly and Novo Nordisk may exert competitive pressure on Kailera.
Nonetheless, the significance of Kailera's IPO extends far beyond the public listing of a single biotechnology company. It represents a replicable "Nasdaq model" for the global expansion of Chinese innovative drugs: a Chinese pharmaceutical company completes clinical validation of a high-value asset domestically, establishes an overseas NewCo to license global rights in exchange for upfront cash, equity, and milestone payments; brings in top-tier Western capital and professional management to accelerate global clinical development; and leverages a Nasdaq listing to unlock global valuation and exit pathways, thereby maximizing asset monetization.
This path represents a new out-licensing model pioneered by Hengrui for the industry, marking a transition for Chinese innovative drugs from "product going global" to a more integrated "asset, capital, and talent ecosystem going global." As Kailera's IPO enters a critical phase, whether the global Phase III clinical trial for Ribupatide achieves its expected efficacy, successfully secures regulatory approval, and attains global pricing power remains the central question. The outcome of this endeavor may shape the competitive landscape of Chinese innovative drugs in the global metabolic disease space.
