The Eve of the Alpha Nuclear Medicine Boom: Novartis Takes the Lead, 2026 Nuclear Medicine Track Stages "Life and Death Race"
Entering 2026, the RDC field has seen new changes.
In the past year, lutetium-177 radiopharmaceuticals have undoubtedly become the hottest research direction in the RDC field. The continuous growth in sales of Novartis' key product Pluvicto has ignited enthusiasm for the development of lutetium-177 radiopharmaceuticals. Nuclear medicine companies, including Beijing Xiantong International Technology Co., Ltd., Yantai Lannacheng Biotechnology Co., Ltd., NuriTech, Tongrui Biologics, and Grand Pharmaceutical, are all strategically positioning themselves in this area. Even ADC companies like Baili Tianheng have joined the ranks of lutetium-177 radiopharmaceutical research and development.
In addition, a breakthrough has been made in the supply chain of lutetium-177 isotopes, resolving the production bottleneck for nuclear medicine. In 2025, the core product lutetium-177 from Hefu No. 1 will be launched. This project, relying on the commercial heavy water reactor at the Qinshan Nuclear Power Base, has achieved large-scale production of lutetium-177. Sichuan Haitong has also successfully achieved the production of lutetium-177 based on the Jiacheng reactor.
As the commercialization of Lutetium-177 has been achieved, the previous reliance on imports has been improved, and the supply of Lutetium-177 will become more stable. The research and development of Lutetium-177 radiopharmaceuticals in China has entered a period of rapid growth, with an increasing number of pipelines under development. Industry insiders predict that market competition for Lutetium-177 radiopharmaceuticals will become increasingly intense.
In this context, researchers have begun to focus on novel α-nuclear drugs. This field has become a key R&D priority for 2026.
Compared with β particles, α particles have greater mass and higher energy. This characteristic enables them to break double helix chains, destroy DNA, and cause localized cell destruction. Another key advantage of α particles is their limited penetration distance, typically only about 50 to 100 micrometers, which can be blocked by a single sheet of paper. Therefore, therapies utilizing α particles can achieve highly localized effects, destroying tumor tissues while avoiding damage to nearby healthy cells.
In the process of tumor treatment, compared with β nuclear medicine, α nuclear medicine has two main advantages: one isLow effective dose. Taking the commonly used Actinium-225 radiopharmaceutical as an example, its usage is about one-thousandth of that of beta radiopharmaceuticals to achieve the same bombardment effect; secondly,Relatively Low Toxicity, mainly due to the shorter radius of α particles combined with a smaller administered dose, resulting in lower cytotoxicity.
Overall, the differences between α and β radionuclide drugs in the field of cancer treatment reflect two distinct therapeutic approaches. β radionuclide drugs kill tumors with relatively mild, wide-ranging radiation, while α radionuclide drugs use intense, short-range energy to precisely eliminate targets. These two characteristics determine their respective strengths and limitations in fighting cancer.
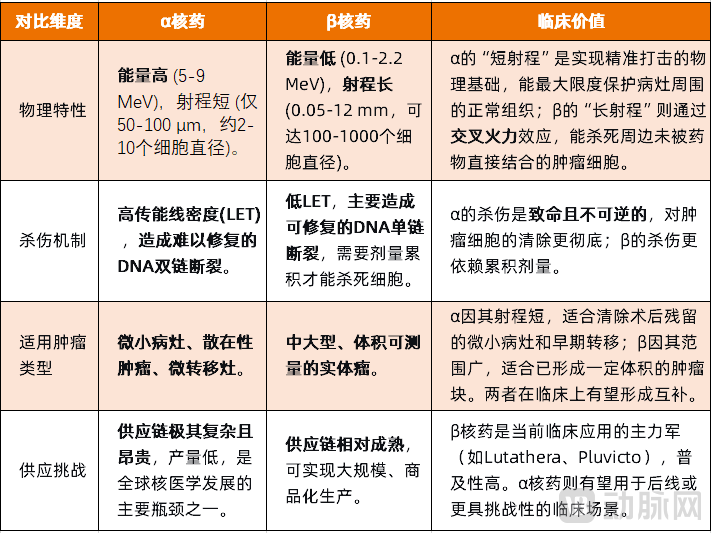
Figure 1. Differences Between α and β Radiopharmaceuticals (Compiled by VCBeat)
Located in the upstream of the nuclear medicine industry chainCROCompanies have already been the first to feel this change. According to Yuan Dawei, founder of Quark Pharmaceuticals, the changes in the nuclear medicine field are mainly reflected in several aspects:
1)Project Type Changes:Radioactive Drugs in the PastCROMore is Lutetium-177, Diagnostic radionuclides, now gradually emerging actinium-225, Lead-212、Astatine-211etc.αNuclide-related projects.
2)More complex needs:AndβRadionuclide (such as¹⁷⁷Lu、⁹⁰Y、¹³¹I) Compared with,αThe R&D chain of nuclear medicine is significant.Longer, more complex, and more specialized, involving multipleβThe link that requires almost no consideration for radionuclides, such as: higher specificity for coupling reactions, and greater difficulty in radiation dose acquisition, thereforeFor those who simultaneously possess a deep understanding of nuclear medicine and full-chain capabilities in drug developmentCROSurge in Demand。
3)Industrial Chain Advanced Layout: SomeCDMOOr radiopharmaceutical enterprises start to build specializedαThe radionuclide production and labeling platform was established due to the anticipated growth potential of future pipelines.
But from the perspective of the overall market size, the commercialization of α-nuclear medicine is still in the pre-outbreak stage, and the real large-scale outsourcing demand is expected to significantly increase only in the later clinical stages or when nearing commercialization.
The current α radionuclide drug pipeline under research mainly focuses on Actinium-225, Dr. Ning Xi, CEO of Fanencore, told VCBeat.The half-life of Actinium-225 is close to that of Lutetium-177, with similar ionic radii, complexation processes, and distribution in the human body.Similarities between Actinium-225 and Lutetium-177 in four core dimensions have becomeThe Bridge for the Field of Nuclear Medicine to Leap from the "β Era" to the "α Era"These four major similarities bring comprehensive convenience to the research and development of Actinium-225 nuclear medicine, from laboratory to clinical applications, and from technology to commercialization.
1) Similar half-life: Simplifying clinical protocols and logistics systems
The half-life of Lutetium-177 is 6.7 days, and that of Actinium-225 is 10 days. The close proximity of their half-lives is crucial in clinical translation.
In the development of radiopharmaceuticals, the biological half-life of the carrier must match the physical half-life of the radionuclide. Since lutetium-177 has successfully validated the in vivo kinetics of a large number of targeting molecules as feasible, when switching to actinium-225, these existing carriers can still work perfectly with the new radionuclide without the need to search for a needle in a haystack among a vast number of compounds.
In addition, the hospital has established a mature "appointment-administration-hospitalization observation-discharge standardized process" for lutetium-177 based drugs (such as Novartis' Pluvicto). The half-life of actinium-225 is close to that of lutetium-177, meaning that the hospital does not need to significantly modify its existing nuclear medicine ward management system. The patient’s hospital stay and medical staff radiation protection protocols can largely draw from existing experience.
2) Similar Ionic Radii: The "Plug and Play" of Chelation Chemistry
The ionic radii of lutetium-177 and actinium-225 are very close, with the ionic radii of the trivalent ions both on the order of 100 picometers.
A crucial step in radiopharmaceutical development is finding a "cage" (chelator) that can tightly hold metal ions and prevent their leakage in the body. The classic DOTA chelator has an extremely strong affinity for Lu³⁺. Since the size of Ac³⁺ is similar to that of Lu³⁺, DOTA can also efficiently chelate Ac³⁺. This means that the experience gained from using DOTA to chelate lutetium-177 (including labeling temperature, pH, reaction time, and quality control methods) can be directly applied to actinium-225, significantly reducing the difficulty of chemical development.
3) Complexation Process Approximation: Ensuring In Vivo Stability
177The stability of Lu-DOTA complexes in vivo has been clinically confirmed, with a low risk of off-target effects. Based on the similarity of the chelation process, we can reasonably infer that,225Ac-DOTA also exhibits similar stability. This provides strong support for the safety argument of the new drug.
Moreover, the method for detecting the stability of radiopharmaceuticals is also universal. Researchers can apply the same quality control standards established for lutetium-177 to evaluate actinium-225 drugs, significantly accelerating the preclinical research process.
4) Similar distribution in the body: Achieving "precise bridging" using imaging data
This is the most strategically valuable among the four similarities. Lutetium-177 is not only a therapeutic radionuclide, but it also emits gamma rays, allowing for imaging, which makes it relatively convenient to observe the distribution and metabolism of the drug in the body. On the other hand, Actinium-225 can hardly be imaged.
Although it is impossible to directly see where Actinium-225 goes, because it is linked to the same targeting molecule, doctors can use the imaging of Lutetium-177 to guide the treatment of Actinium-225. The imaging of Lutetium-177 becomes the "eyes" of Actinium-225.
The four major similarities between Actinium-225 and Lutetium-177 constitute a comprehensive technical translation pathway.
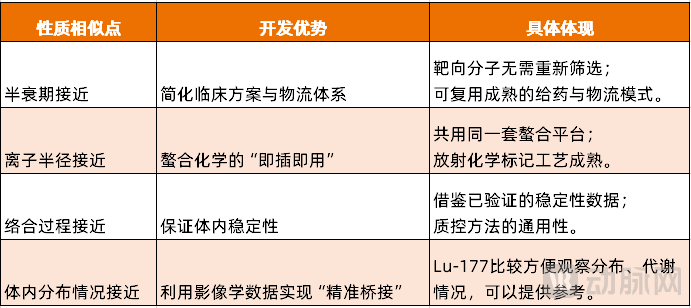
Figure 2. Actinium-225 and Lutetium-177 have similar properties, creating a development advantage (Source: VCBeat)
In addition to Actinium-225, another type of α-radiotherapeutic drug, Lead-212, is also under development. Lead-212 inSignificant advantages in supply stability and costMoreover, the observation of lead-212 is easier than that of actinium-225, as the gamma rays released during the decay of lead-212 can be used for SPECT imaging, enabling "what you see is what you treat."
The difference between lead-212 and actinium-225 lies in their half-lives. Lead-212 has a half-life of only 10.6 hours, while actinium-225 has a half-life of up to 10 days. The short half-life of lead-212 meansFast radiation effect, potentially safer. However, the short half-life of lead-212 also means that its circulation time in the body does not match with the target molecules, and the development experience accumulated with lutetium-177 is also difficult to apply to the development of lead-212-based radiopharmaceuticals.
Moreover, due to the short half-life of lead-212, its production cannot adopt a centralized approach similar to actinium-225 and lutetium-177. Lead-212 radiopharmaceuticals need to be produced in facilities close to hospitals and supplied for patient use.
Actinium-225 has a longer half-life and can release more energy, while lead-212 offers better safety and is more abundantly supplied. These two radionuclides have become the mainstream focus of current α-radiotherapeutics research and development.
Currently, the research and development of nuclear medicine companies in the α-nuclear medicine field are mainly focused on two key directions: Actinium-225 and Lead-212, with pipelines advancing rapidly. According to statistics from VCBeat, as of March 4, 2026, there are 28 active Actinium-225 nuclear medicine pipelines globally that have progressed beyond clinical Phase I, along with 11 Lead-212 pipelines.
Among them, the pipeline of Actinium-225 nuclear medicine is more extensive, and the clinical progress is faster.225Ac-PSMA-617 and RYZ101 have advanced to Phase III clinical trials and are about to be launched.
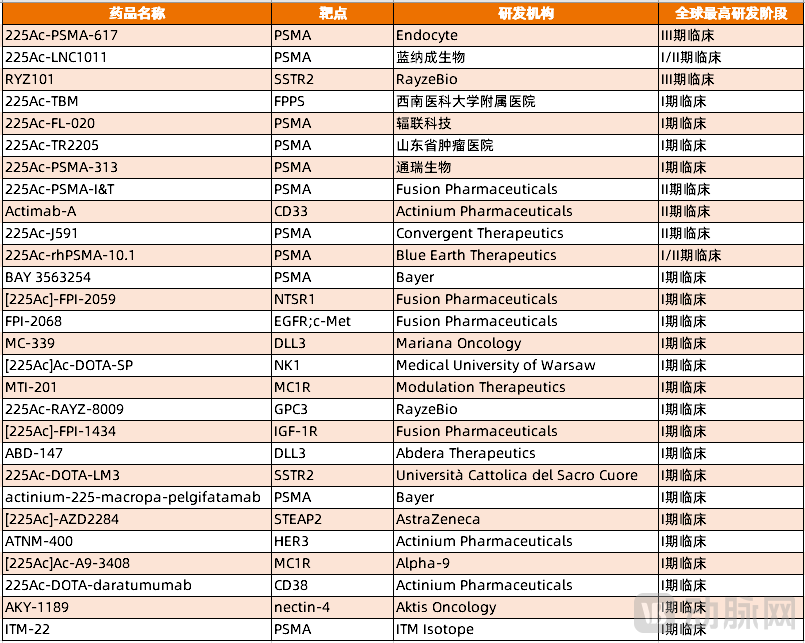
Figure 3. Actinium-225 nuclear medicine pipeline after entering clinical Phase I (Source: PharmaCube)
Novartis225Ac-PSMA-617 (AAA817) Injection is currently the most highly anticipated Actinium-225 radiopharmaceutical product. Compared to Novartis' blockbuster drug Pluvicto, AAA817 replaces the radioactive isotope Lutetium-177 with Actinium-225 and then conjugates it with PSMA-617.
AAA817, as Novartis' next-generation RDC product, utilizes the α-particles emitted by the Actinium-225 radionuclide, which possess the unique advantages of high energy and short range. This enables the induction of more severe DNA damage in cancer cells while reducing off-target toxicity to healthy tissues and tissues surrounding the diseased area. AAA817 achieves precise tumor cell killing and minimizes damage to normal tissues to the greatest extent, compared to β-particle emitting177Compared with Lu-PSMA-617, its therapeutic potential is even stronger.
Previously, in a clinical trial where patients with mCRPC who had developed resistance to standard therapies (including next-generation anti-androgen therapies and steroid-based chemotherapy) were treated with AAA817, the treatment demonstrated promising efficacy for advanced mCRPC patients. Eligible patients received AAA817 at doses of 100-150 kBq/kg. Of the initial 63 patients enrolled, a total of 56 completed at least two cycles of AAA817 treatment and were included in this study.
The results showed that 91% of patients had a decrease in PSA levels, with 67.8% experiencing a reduction of 50% or more. PSA (Prostate-Specific Antigen) is a protein produced by prostate cells and is commonly used to detect and monitor prostate cancer. A significant drop in PSA levels often indicates effective control of tumor burden. In another study targeting Novartis177In the Phase III clinical study of Lu-PSMA-617, 57.6% of patients had a PSA decline of 50% or more.AAA817 Achieved a Greater Proportion of Patients with PSA Reduction。
Fatigue is one of the most common treatment-related adverse reactions in AAA817 therapy, with 70% of patients experiencing grade 1/2 fatigue and 3.5% of patients experiencing grade 3 or higher fatigue. This fatigue is transient and will resolve before the next treatment cycle. Additionally, approximately one-third of patients experience xerostomia (grade 1/2: 32.1%).
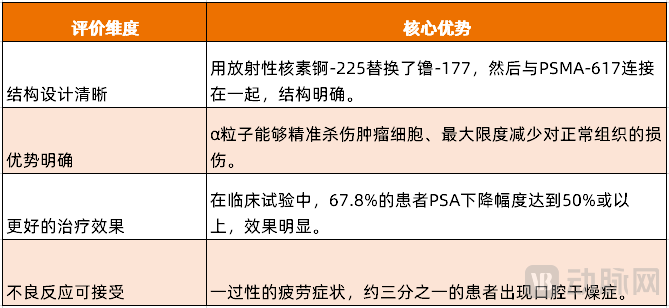
Figure 4. AAA817's Core Advantages (Source: VCBeat)
Overall, AAA817 targeted α-therapy was well-tolerated with acceptable adverse reactions and showed efficacy in treating advanced mCRPC patients. The product is currently undergoing Phase III clinical trials.Expected to become the first α radionuclide drug to be marketed。
Also targeting PSMA, the progress of the pipeline in China closely follows AAA817 and shows advantages: 1)225Ac-LNC1011 Shows Better Therapeutic Potential in Early Animal Trials, In addition177The development experience of Lu-LNC1011 will also benefit Yantai Lannacheng Biotechnology Co., Ltd.225The development of Ac-LNC1011 provides experience; 2)225Ac-PSMA-CY313 Receives High Peer RecognitionAt the European Association of Nuclear Medicine (EANM 2025) conference held in October last year, two studies supported by Tongrui Biotech on225The research on Ac-PSMA-CY313 in the mCRPC field has gained high recognition from the conference review committee for its innovativeness and significant value, and was selected as the "Top Rated Oral Presentation."FluorineLink Technology225Ac-FL-020 Injection Granted Fast Track Designation by FDA。
In the PSMA target, the domestic pipeline closely follows Novartis' AAA817 and has formed a strong catching-up trend.
Another product that has entered Phase III clinical trials for Actinium-225 nuclear medicine is RYZ101, an RDC drug targeting SSTR2. However, the progress of RYZ101's Phase III clinical trial has been affected by the supply issues of the upstream radionuclide Actinium-225.
Lead-212-based radiopharmaceuticals are another highly regarded α-therapy. Not long ago, Sanofi terminated AlphaMedix (212The development of Pb-DOTAMTATE, an α radionuclide drug targeting SSTR, has brought significant pressure to the subsequent advancement of lead-212-based radiopharmaceuticals. Currently, there are 11 lead-212 radiopharmaceutical pipelines that have reached or progressed beyond Phase I clinical trials.
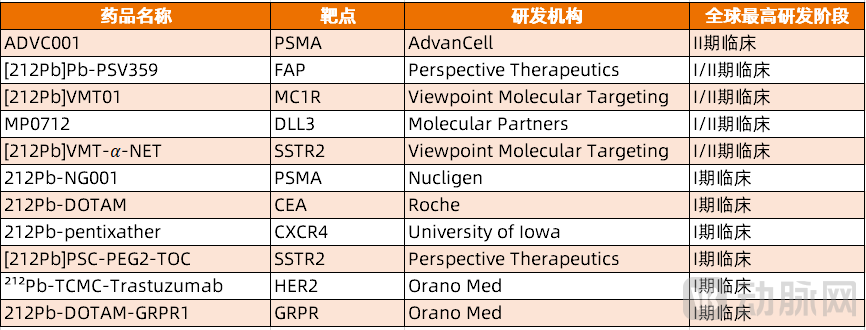
Figure 5. Lead-212 radiopharmaceutical pipelines entering Phase I clinical trials (Source: PharmaCube)
Although the development of lead-212 is lagging behind that of actinium-225, funding continues to flow into this area. In early February this year, nuclear medicine company Perspective Therapeutics raised approximately $175 million through a share issuance to accelerate its lead-212-based technology platform and clinical programs. Two of its pipeline products, VMT-α-NET and VMT01, have entered Phase I/II clinical trials, targeting MC1R and SSTR2, respectively.
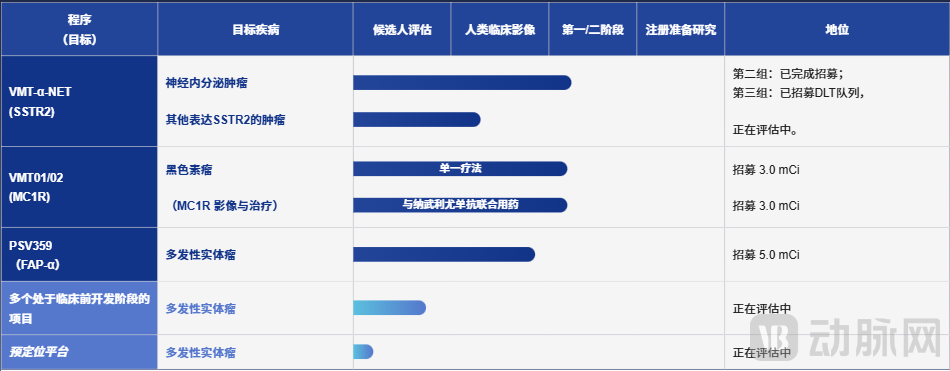
Figure 6. Pipeline of Perspective under research (Source: Company official website)
Lead-212 has a short half-life and is more abundantly supplied, which gives such nuclear medicines significant development potential.
Although α radionuclide drugs are currently developing strongly, it does not mean that α radionuclide drugs will replace β radionuclide drugs in the future. In fact, the two should be complementary and develop synergistically. Dr. Ning Xi, CEO of Fanencore, elaborated on the relationship between these two types of drugs.
On one hand, β radionuclide drugs remain the cornerstone of current nuclear medicine, and with their mature supply chain and more reliable clinical data, β radionuclide drugs will continue to play a central role in the treatment of various cancers. On the other hand, α radionuclide drugs, with their precision and stronger destructive power, are expected to demonstrate advantages in clearing micro-metastases. Additionally, for certain cancer patients who develop metastases after β radionuclide drug treatment, α radionuclide drugs are anticipated to further deliver greater clinical value.
The research and development of α radionuclide drugs are still in the early stages, and more clinical data needs to be accumulated. With further research, α radionuclide drugs are expected to demonstrate greater clinical value.
