Rolling out the $15B bet: AstraZeneca unveils cell therapy and RDC hubs in China
On March 19, AstraZeneca announced that it will establish a cell therapy commercial manufacturing and supply base as well as an innovation center in Shanghai, becoming the first globally leading biopharmaceutical company with end-to-end cell therapy capabilities in China.
On the same day, AstraZeneca signed a memorandum of understanding with the Shanghai Municipal Commission of Science and Technology and several leading UK scientific research and financial institutions to launch the "Shanghai–UK Life Science Innovation Ecosystem Cooperation Program."
Also on that day, AstraZeneca signed a memorandum of understanding with the Guangzhou Economic and Technological Development Zone Administrative Commission, announcing its plan to build a radioconjugate drug manufacturing and supply base in Guangzhou. The base will specialize in producing radioconjugate drugs to provide next-generation cancer therapies for patients in China and the broader Asia-Pacific region.
This series of moves is undeniably significant. From the previously established global strategic R&D centers in Beijing and Shanghai, to now encompassing advanced manufacturing bases for cell therapy and radioconjugate drugs (RDCs), what is the strategic logic behind AstraZeneca's approach?
Growth Engine Slows Down
On January 29, 2026, AstraZeneca announced its plan to invest over RMB 100 billion (USD 15 billion) in China by 2030 to expand its footprint in pharmaceutical manufacturing and R&D.
Pascal Soriot, Chief Executive Officer of AstraZeneca, stated that this significant investment marks a new chapter in the company's development in China. By further expanding our capabilities in breakthrough therapies such as cell therapy and radioconjugate drugs, AstraZeneca will contribute even more to China's high-quality development, and more importantly, bring next-generation innovative treatments to more patients.
Undoubtedly, AstraZeneca's recent series of announcements reflects the fulfillment of its previously stated investment plan.
To gain a deeper understanding, however, one must ask: why now?
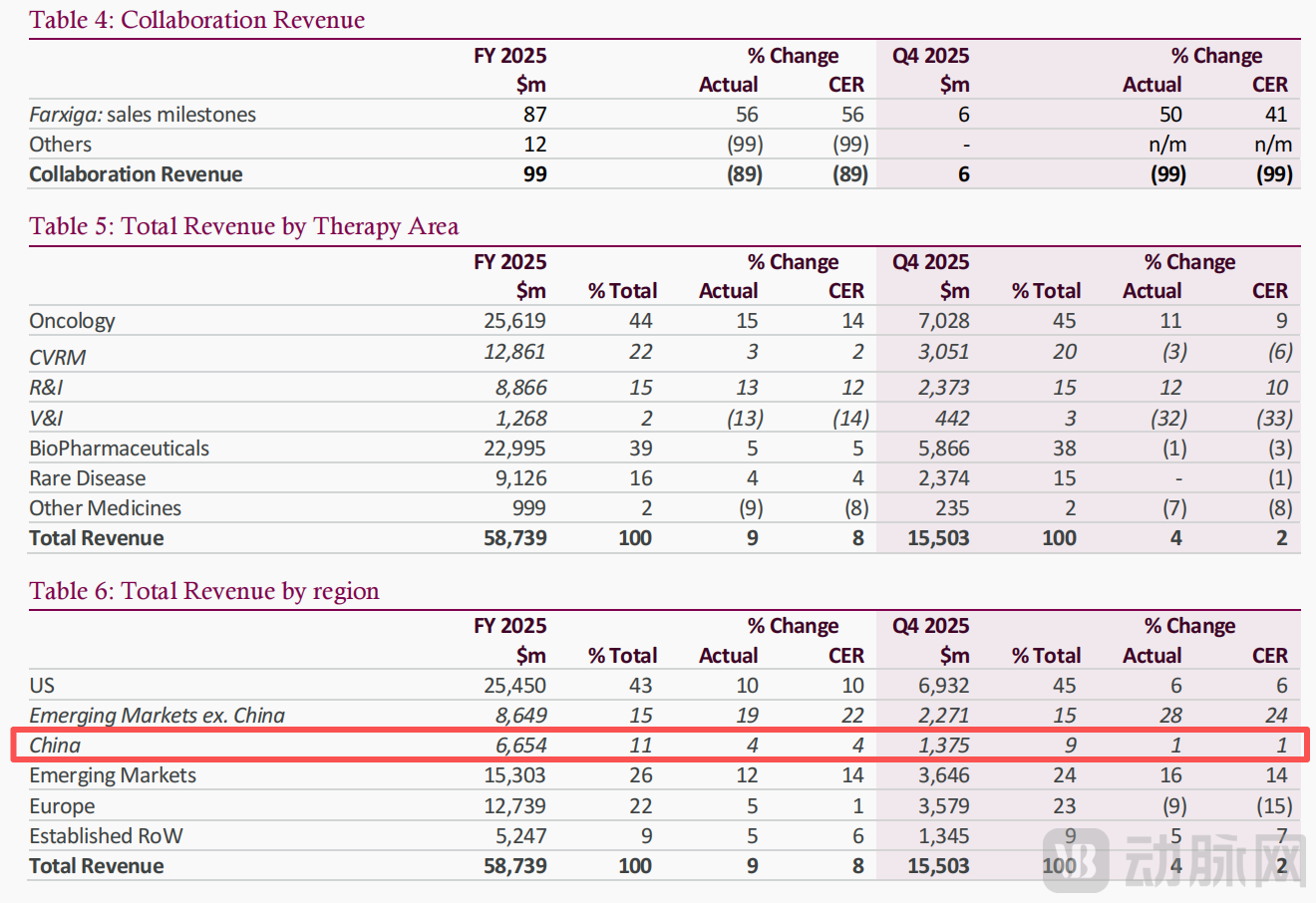
The answer lies in AstraZeneca's financial results. In February 2026, AstraZeneca delivered an annual performance report characterized as "steady but slowing." In 2025, AstraZeneca achieved total revenue of USD 58.739 billion, representing an 8% increase year-over-year (at constant exchange rates, CER, unless otherwise specified). Oncology remained the core business, generating approximately USD 25.6 billion, accounting for 44% of total revenue.
However, when viewed over a longer timeframe, the growth rate of this segment has declined from 19% in 2023 and 21% in 2024 to 14% in 2025—a clear signal of deceleration.
Meanwhile, total revenue from the China region in 2025 reached USD 6.654 billion, a 4% increase year-over-year. Although this growth rate slowed from 9% in 2024, China's revenue still accounted for 43.5% of total revenue from emerging markets, positioning it as an indispensable strategic pillar in AstraZeneca's global footprint.
Notably, in its 2025 financial report, AstraZeneca began reporting China's performance as a separate line item outside of "Emerging Markets," further underscoring its strategic importance.
Source: AstraZeneca Financial Report
Pascal Soriot stated unequivocally that China has become a major force in scientific innovation, advanced manufacturing, and global public health.
This observation helps answer the question of why AstraZeneca has chosen to increase its investment in China.
At present, the Chinese market is transforming from a pure sales market into a global hub for innovation.
In December 2025, Jacobio Pharma entered into a collaboration agreement with AstraZeneca for its proprietary pan-KRAS inhibitor JAB-23E73. AstraZeneca paid an upfront payment of USD 100 million and committed up to USD 1.9 billion in milestone payments to obtain exclusive rights to the drug outside of China.
In January 2026, AbelZeta Pharma announced that AstraZeneca had agreed to acquire its 50% stake in the development and commercialization rights for C-CAR031 in China. Upon completion of the agreement, AstraZeneca will obtain global exclusive rights to develop, manufacture, and commercialize C-CAR031, with AbelZeta eligible to receive up to USD 630 million.
These deals also demonstrate that AstraZeneca is "re-exporting" innovative outcomes from China to the global market.
According to data from BioPharma Dive, Chinese companies signed more than 60 licensing agreements with pharmaceutical manufacturers in the United States and Europe in 2025. AstraZeneca alone has entered into 16 such agreements with Chinese partners since 2023.
Targeted Cell Therapy Strategy
Why was Shanghai chosen as the base for establishing end-to-end cell therapy capabilities encompassing manufacturing and innovation?
This move may be linked to an acquisition AstraZeneca completed in 2024.
In February 2024, AstraZeneca completed the acquisition of Gracell Biotechnologies, gaining its FasTCAR rapid manufacturing platform and the BCMA/CD19 dual-target CAR-T cell therapy AZD0120, which is currently undergoing clinical research in multiple myeloma and autoimmune diseases.
AstraZeneca has announced that it will build a dedicated cell therapy commercial manufacturing and supply base in the Lingang New Area of the Shanghai Pilot Free Trade Zone to commercialize and supply autologous CAR-T cell therapies for China and other Asian markets, with AZD0120 being among them.
At the same time, AstraZeneca will establish the AstraZeneca Gracell Cell Therapy Innovation Center in the Zhangjiang Hi-Tech Parkin Shanghai, covering early-stage research, viral vector and plasmid development, analytical testing, clinical manufacturing, and regulatory support.
Notably, the collaboration program launched by AstraZeneca in partnership with the University of Glasgow, King's College London, and other institutions focuses on joint scientific research and clinical studies aimed at accelerating the development of innovative solutions to address global health challenges. Both universities have accumulated significant research achievements in areas such as CGT and immunology.
Iskra Reic, Executive Vice President, International, at AstraZeneca, stated that Shanghai has built a solid pathway from basic research, R&D translation, to advanced manufacturing, and has fostered a vibrant innovation ecosystem.
Meanwhile, the construction of the radioconjugate drug manufacturing and supply base in Guangzhou will create synergies with the cell therapy base in Shanghai, together forming AstraZeneca's localized manufacturing network in novel drug modalities.
Cell therapy represents AstraZeneca's growth engine for the next decade, following its sustained leadership in oncology.
The 2025 financial report shows that oncology revenue reached USD 25.619 billion, accounting for 44% of AstraZeneca's total revenue, representing a 14% year-over-year increase. For instance, Tagrisso (osimertinib) generated USD 3.064 billion in revenue in the U.S. market and USD 1.971 billion in emerging markets, with the Chinese market in particular benefiting from rapid volume growth driven by adjustments to the National Reimbursement Drug List.
However, as competition in the oncology space intensifies, the growth rate of the oncology business has slowed, declining from 19% in 2023 to 14% in 2025.
Cell therapy may represent a future breakthrough. In 2025, AstraZeneca acquired EsoBiotech, a Belgian company, with an upfront cash payment of USD 425 million, and a total potential consideration of up to USD 1 billion. EsoBiotech's in vivo cell therapy technology enables immune cell reprogramming within minutes via a single intravenous infusion, significantly reducing the complexity and cost associated with traditional CAR-T therapies.
Strengthen Localized Production and Innovation Capabilities
Underlying this shift is the intensifying competition in the global innovative drug market, with multinational corporations entering a more complex phase in their strategic positioning across different regions.
According to financial data, the U.S. market remains AstraZeneca's largest single market, with revenue rising from USD 19.077 billion in 2023 to USD 25.450 billion in 2025, a net increase of USD 6.4 billion over three years.
After achieving exceptionally high growth of 27% in 2024, the European market saw its growth rate moderate to 9% in 2025. AstraZeneca noted that the slowdown was related to pricing pressures in the European market and the erosion of certain products by generic competition.
Even in the Chinese market, performance has been impacted by factors such as inclusion in the National Reimbursement Drug List (NRDL) and volume-based procurement (VBP) policies.
Lynparza (a PARP inhibitor) generated USD 3.279 billion in global revenue, representing a 6% year-over-year increase. However, in emerging markets, it achieved only USD 669 million, with growth of just 2%, attributed to competitive pressures in the Chinese market and the effects of VBP.
Legacy products such as Crestor (rosuvastatin) have also faced pressure, with VBP significantly eroding their market share and pricing power.
Nevertheless, the diabetes drug Farxiga (dapagliflozin) continued to perform strongly in 2025, growing 9% year-over-year to reach USD 8.492 billion, making it the company's best-selling product for the year.
In China, the uptake of innovative drugs is heavily dependent on NRDL inclusion. For example, Enhertu (trastuzumab deruxtecan) experienced rapid volume growth after being included in the NRDL in 2025. AstraZeneca disclosed in its financial report that the product delivered strong revenue performance in the fourth quarter of fiscal year 2025.
Additionally, personnel and organizational changes may have had an impact. According to market sources, Huang Bin, a veteran executive with over twenty years at AstraZeneca and Vice President of AstraZeneca China, has officially announced his departure, with his tenure concluding on March 20, 2026.
Farxiga, mentioned earlier, achieved volume growth through NRDL negotiations. In 2019, Huang Bin led AstraZeneca's NRDL negotiations for dapagliflozin, successfully securing its inclusion in the NRDL at a global-low price of RMB 4.36 per 10 mg tablet, a 73% reduction from its original price of RMB 15.96.
As a veteran who participated in NRDL negotiations for seven consecutive years, Huang Bin took part in a total of 26 negotiation sessions, facilitating the inclusion of multiple innovative drugs such as Enhertu and capivasertib into the NRDL.
Organizational restructuring is also advancing in parallel, with an increasingly evident focus on channel synergy and integrated management.
In March 2026, AstraZeneca China established a new "BioPharmaceuticals Full-Product Channel Business Unit," consolidating retail operations, county and community operations, and the Falcon businesstogether with respiratory nebulization and digestive injectable products, which were previously under different divisions.
Thus, amid organizational, structural, and personnel changes, AstraZeneca's strategic approach in China is clear: it is not merely selling drugs in China, but also conducting R&D and manufacturing in China, and exporting Chinese innovation to the global market.
