Will a New $54 Billion Diabetes Leader Soon Emerge?
Medtronic Diabetes Business to Be Spun Off and Listed Independently!
Recently, MiniMed, which became independent from Medtronic's diabetes business, submitted a prospectus to the U.S. Securities and Exchange Commission, planning to issue 28 million shares at a price of $25-$28 per share, raising up to $784 million. The lineup of institutions supporting its IPO is impressive, including Goldman Sachs, Bank of America, Citi, Morgan Stanley, Barclays, and Deutsche Bank.
A medical device giant with a potential valuation of up to $7.86 billion (approximately over 54 billion RMB) is about to emerge.
This time, Medtronic's diabetes business became independent under its original name. MiniMed, founded by Alfred E. Mann in 1983, has a history of over 40 years and is highly renowned in the diabetes field. It launched many industry-first products, including the first pager-sized portable insulin pump, the first CGM (Continuous Glucose Monitor), and the first wearable insulin pump integrated with CGM.
In 1999, the first CGM system introduced by MiniMed became a significant breakthrough in diabetes technology. Due to this milestone innovation, MiniMed was acquired by Medtronic for approximately $3.7 billion in 2001, becoming Medtronic's diabetes business division.
After joining Medtronic, MiniMed continued to achieve breakthroughs in diabetes technology, launching several innovative products. These accomplishments have also established Medtronic's leading position in the diabetes field.
Until the middle of last year, Medtronic announced that it would spin off its diabetes business. After more than 20 years of being part of Medtronic, MiniMed will become independent again.
On the one hand, MiniMed is the smallest business unit under Medtronic. According to Medtronic's fiscal year 2025 financial report, the diabetes business accounts for only 8% of its revenue, with an even lower profit share of 4%. Against the backdrop of Medtronic's recent financial performance pressures, this is considered to severely drag down Medtronic's profitability.
In terms of business model, MiniMed also has significant differences from other Medtronic business lines. Other business segments of Medtronic, whether cardiovascular, neuroscience, or medical surgical and patient monitoring, are mainly B2B models focused on hospitals. However, diabetes-related businesses such as CGM and insulin pumps need to directly face patients, which belong to the B2C model.
This difference in operational model makes it difficult for MiniMed to have synergies with other Medtronic business lines in terms of supply chain and market strategy, and it cannot leverage the channel advantages of other business lines. Instead, a separate system needs to be built specifically for it.
However, such a small performance share has destined MiniMed to be completely incomparable with other business units in the internal competition for resources. For MiniMed, which faces many strong competitors in the diabetes technology field and needs to respond quickly, this is obviously disadvantageous.
On the other hand, the huge market potential of diabetes technology is well known to all. Past financial reports also showed that MiniMed's growth rate had indeed exceeded double digits for multiple consecutive quarters, making it the department with the highest growth rate under Medtronic.
Against this backdrop, spinning off MiniMed has become a reasonable choice. Medtronic can rid itself of the drag on performance caused by MiniMed; meanwhile, MiniMed can better respond to the rapidly evolving diabetes technology market through independence.
It is worth mentioning that this spin-off listing is another manifestation of the "spin-off listing trend" among the world's leading medical device companies in recent years. Prior to this, major healthcare giants such as 3M, GE, Johnson & Johnson, Baxter, and BD have all made corresponding spin-off moves. Johnson & Johnson, which has already split its pharmaceuticals and devices divisions, will even further spin off its orthopedics business for a separate listing.
The deep-rooted reason behind these spin-off actions is the capital market's recent preference for "specialized and refined" enterprises. Compared to large-scale, diversified conglomerates, companies that focus on a specific niche business are now more likely to gain favor with investors.
Based on past situations, after the split, the combined market value of the two companies is highly likely to exceed the previous standalone market value of Medtronic.
Of course, mergers and acquisitions are just capital maneuvers; a strong product capability is the key to ensuring competitiveness. In this regard, MiniMed, with decades of experience in diabetes technology, has deep expertise.
Diabetes management devices mainly consist of three core components: sensors (such as CGM), insulin pumps, and dosing algorithms. Most players in this field specialize in a specific core component; for instance, Abbott focuses more on CGM. MiniMed is the only company with commercialized product solutions across all aspects, naturally enabling a better closed-loop solution.
It is precisely because of this foundation that MiniMed has gradually established an advantage in the AID (Automated Insulin Delivery) system.
Currently, even though CGM sensors capable of continuous blood glucose monitoring are available, manual drug administration remains the mainstream solution for diabetes management. However, the integration of CGM with AID systems has been widely recognized as one of the important trends in the development of diabetes technology.
This integrated treatment solution combines sensors, insulin pumps, and dosage algorithms to automatically adjust and administer insulin pump doses based on real-time blood glucose data provided by the CGM system. This achieves more precise and personalized blood glucose control, effectively reducing fluctuations in blood glucose levels, and moves closer to the ultimate goal of an ideal fully automated artificial pancreas.
AID, with numerous advantages, has been increasingly recommended for first-line treatment. The American Diabetes Association has suggested that AID systems should be provided to adolescents and adults as early as at the time of diagnosis. Moreover, the consensus report from the International Conference on Advanced Technologies & Treatments for Diabetes also recommends that all patients with type 1 diabetes should consider using AID systems, especially those with suboptimal blood glucose levels, problematic hypoglycemia, and significant glucose variability, and it suggests that all payers should cover AID systems.
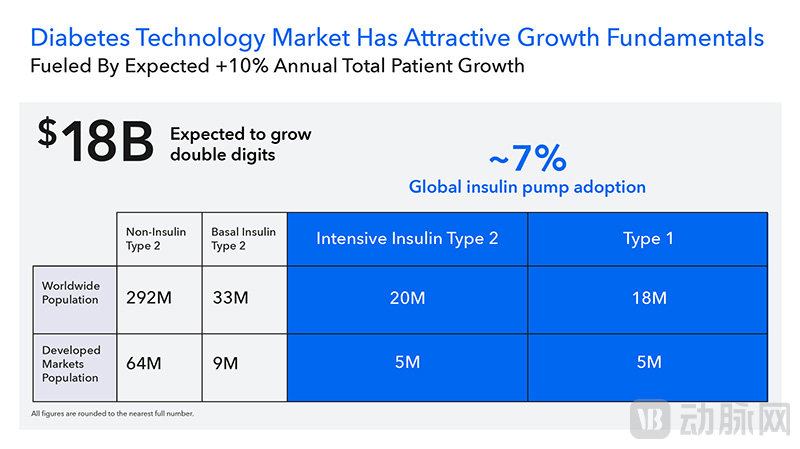
The diabetes technology market where MiniMed is located has huge potential (screenshot from the prospectus).
However, the penetration rate of AID globally needs improvement. According to the prospectus, only 26% of Type 1 diabetes patients and 6% of insulin-requiring Type 2 diabetes patients in developed markets worldwide have adopted AID systems; in developing countries, the penetration rates are as low as 3% and less than 1%, respectively. In the future, the market size of AID systems is expected to exceed $18 billion, with a compound annual growth rate of over 10% between 2025 and 2030.
Over the past decade, Medtronic MiniMed has been the driving force behind the development of AID systems, evolving from the LGS (Low Glucose Suspend) to PLGS (Predictive Low Glucose Suspend), then to HCL (Hybrid Closed Loop), and iterating to the current FCL (Full Closed Loop) system.
MiniMed 780G is the latest generation of Medtronic MiniMed's AID system and also its star product. The included 780G insulin pump is claimed to be the world's first insulin pump with meal detection technology, capable of automatically adjusting and correcting blood glucose levels every 5 minutes. Its hypoglycemia target setting can be as low as 100 mg/dL, highly similar to the average blood glucose level of non-diabetic individuals.
In the sensor part, there are two options: MiniMed's self-developed Simplera Sync CGM sensor, which has a usage duration of only 7 days, but can be combined with its proprietary algorithm to become the preferred choice; additionally, the system is also compatible with Abbott's widely-used Instinct CGM sensor—both parties established a cooperation agreement as early as August 2024, allowing the 780G system to be compatible with Abbott's sensors, thereby providing patients with a 15-day CGM sensor usage period.
The SmartGuard dosage algorithm is the key to the system's ability to automatically adjust every 5 minutes. The algorithm updates every night to adapt to the continuous changes in user behavior patterns. At the same time, it can automatically inject more insulin when the sensor detects an initial rising trend in blood glucose, reducing fluctuations while ensuring safety.
Thanks to this intelligent algorithm, the use of the 780G is almost seamless. Patients only need to estimate carbohydrates a few times a day; even if they forget to input, the patented meal detection technology will automatically correct blood glucose levels.
At the same time, the tubing of the 780G system, through advanced materials and design, can maintain insulin flow and stability, reducing the risk of infusion set blockage, with a usage duration of up to 7 days. This is more than double the typical 3-day usage time offered by competitors.
Interestingly, the launch of the 780G has been full of twists and turns. Initially, things went relatively smoothly, with the core component, the 780G insulin pump, receiving CE certification in June 2020. Medtronic then submitted an application for market approval to the FDA at the beginning of 2021.
However, due to the large-scale recall of nearly 500,000 units of Medtronic's MiniMed products due to quality issues between 2018 and 2021, the FDA issued a stern warning and suspended the approval of subsequent products. The launch of the 780G in the United States had to be temporarily shelved, allowing competitors' new products to dominate the market. This significantly impacted the performance of Medtronic's MiniMed in the United States.
Until April 2023, these issues were resolved, and the 780G insulin pump received FDA approval for use in type 1 diabetes patients aged 7 years and older, as well as type 2 diabetes patients aged 18 years and older who require insulin. As of October 2025, the 780G has gained over 640,000 users across approximately 80 countries.
After receiving approval for market launch, the 780G has gained market recognition. According to a 2024 meta-analysis of competitive systems for the 780G, the system significantly outperformed its competitors in the TIR (Time in Range, the percentage of time patients spend within the ideal blood glucose level range of 3.9-10.0 mmol/L) metric. The results of the pump satisfaction survey from the Q2 2025 "Voice of the American Diabetes Patient" report showed that since Q2 2024, the 780G has consistently ranked first in insulin pump satisfaction in the U.S. market.
This is closely related to the solid clinical trial and real-world evidence base of 780G. MiniMed has completed 8 randomized clinical trials, 9 health economics analyses, over 200 peer-reviewed papers, and real-world evidence based on approximately 400,000 disabled individuals, demonstrating a significant reduction in burden and improved outcomes.
In addition to continuously expanding the addressable market of existing products by broadening the range of indications, MiniMed's future portfolio plan is also presented in the prospectus.
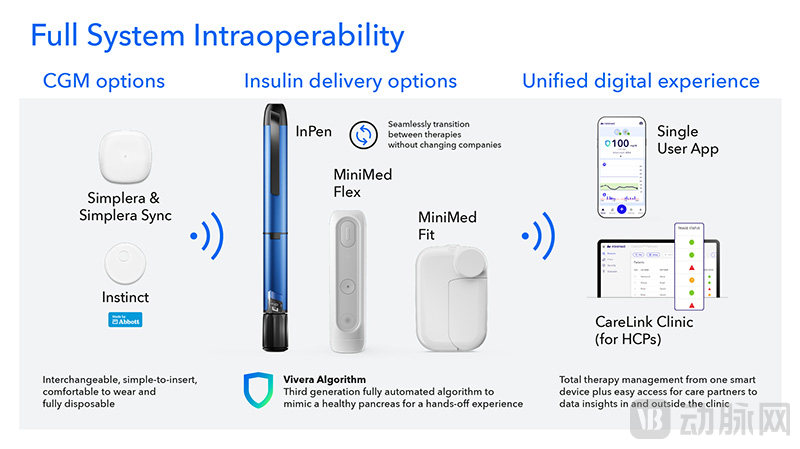
MiniMed's Future Product Layout (Screenshot from the Prospectus)
The most anticipated product is undoubtedly the next-generation insulin pump. The pen-designed MiniMed Flex insulin pump is only half the size of the 780G and has already been submitted for FDA approval, with plans to complete CE certification in the first quarter of 2026. The MiniMed Fit, on the other hand, is a patch pump, boasting a single wear time of up to 7 days and a reservoir capacity of up to 300 units, making it highly anticipated.
On the software side, the next-generation Vivera dosing algorithm is also quite promising. It will enable a "hands-free" AID system that, after just one daily total dose setting, can safely and reliably deliver the correct dose of insulin at the right time without requiring regular user input. The blood glucose target can be flexibly set between 90-140 mg/dL. This algorithm is expected to enter critical trials in the first quarter of 2026.
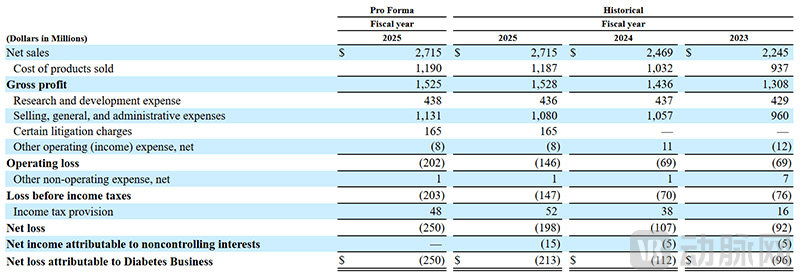
MiniMed's Recent Performance (Screenshot from the Prospectus)
The prospectus also revealed the recent performance of Medtronic's MiniMed. According to the prospectus, in the fiscal year 2025, MiniMed's revenue reached 2.7 billion U.S. dollars, increasing by 10% compared to 2.47 billion U.S. dollars in the fiscal year 2024. Looking further back, the fiscal year 2025 also saw a 10% growth compared to the fiscal year 2024.
The fact that it can achieve double-digit growth rates consecutively in the current unfavorable macroeconomic environment indeed reflects the enormous potential of this market.
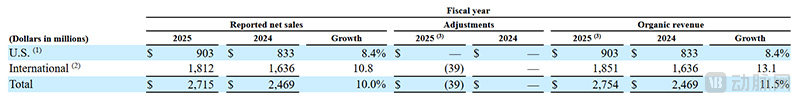
MiniMed's Revenue in Domestic and International Markets (Screenshot from the Prospectus)
MiniMed's internationalization level is relatively high, with revenue from markets outside the U.S. reaching $1.81 billion, accounting for 66.7%. Meanwhile, the growth rate of revenue from markets outside the U.S. is also significantly higher, with a 10.8% increase in fiscal year 2025 compared to fiscal year 2024.
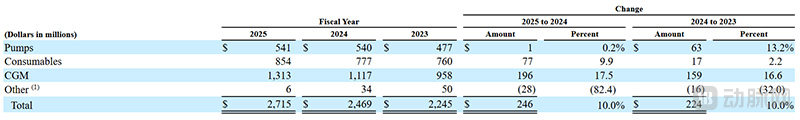
MiniMed Revenue by Product Type (Screenshot from the Prospectus)
By product type, revenue from the CGM segment was the highest, exceeding $1.3 billion, and its share of total revenue increased from 52% to 59%, with the highest growth rate at 17.5% year-over-year. Specifically, the U.S. market grew by 22% year-over-year, while the international market grew by 15%.
Since the intelligent algorithm of the 780G system only supports MiniMed's self-developed CGM sensor, the growth of the CGM segment also indirectly reflects the promising growth potential of the AID system.
Revenue from product consumables and insulin pump segments reached $8.54 billion and $5.41 billion, respectively, representing year-over-year growth of 9.9% and 8.4%, respectively.
As the AID system requires frequent replacement of consumables, the increase in revenue from the product's consumable parts also suggests from another perspective that the AID system indeed offers a great experience and has a good patient retention rate.
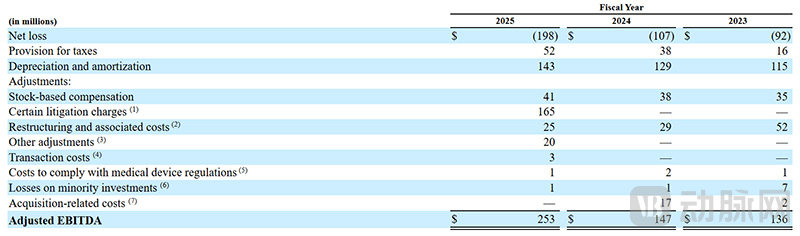
MiniMed's Financial Performance in Recent Years (Screenshot from the Prospectus)
Compared to its revenue, MiniMed's profit performance has been relatively average. In the fiscal year 2025, it reported a net loss of $198 million, with adjusted EBITDA profits returning to $253 million. From the perspective of a startup, this performance is not bad, but compared to Medtronic’s other businesses, it does fall short. However, after the spin-off, MiniMed will be able to focus its resources and adopt more flexible strategies, and its future profitability is expected to improve.
| 2025 Diabetes Revenue | Accounting for the group's total revenue | 2025 Diabetes Profit | |
| Abbott | $7.998 billion | 18% | N/A |
| Dexcom | 4$6.62 billion | 100% | $836 million |
| MiniMed (Medtronic) | $2.755 billion | 8% | -$198 million |
| Insulet | $2.708 billion | 100% | $247 million |
| Tandem Diabetes | $1.015 billion | 100% | -$205 million |
Compared with its main competitors, MiniMed's revenue lags significantly behind Abbott and Dexcom but slightly surpasses Insulet, ranking third. However, its profit is far behind Insulet, which is at a similar revenue level, and is basically only comparable to Tandem Diabetes, whose revenue is less than half of MiniMed’s. Clearly, with MiniMed's independence, barring any unforeseen circumstances, there is considerable room for profit growth. With greater autonomy and solid product strength, it is not impossible for MiniMed to challenge the two dominant players in the diabetes market moving forward.
Medtronic's Spin-off of Diabetes Business into MiniMed and Independent IPO to Have Significant Impact on Global Diabetes Market, Reflecting Growing Preference for Specialized Companies
So, will the subsequent developments really unfold as both parties expect? Will Medtronic, the medical giant, further split its businesses? VCBeat will continue to keep an eye on this.
