Can an Implantable Insulin Pump with FDA Breakthrough Designation Become the "Ultimate Solution" for Type 1 Diabetes?
In the field of diabetes management, wearable insulin pumps have become a part of daily life for many people with type 1 diabetes. However, while devices such as patch pumps and tubed pumps have greatly alleviated the burden of frequent injections, they have not completely eliminated the presence of the "diabetes patient" label in everyday life—patients still need to constantly monitor whether the device is displaced, calculate the carbohydrate count for each meal, manually input doses, and remain vigilant about the risk of nocturnal hypoglycemia.
Recently, the implantable insulin pump, which has been silent for more than two decades, has once again made a breakthrough.
In February 2026, Portal Diabetes, headquartered in the United States, announced that its developed Portal Pump™ implantable insulin pump system officially received the "Breakthrough Device Designation" from the U.S. FDA.This pioneering artificial pancreas — a fully automated insulin delivery system — delivers insulin into the peritoneal cavity and integrates with modern real-time continuous glucose monitoring (iCGM) technology, aiming to achieve closed-loop control of insulin infusion to improve patient health outcomes.
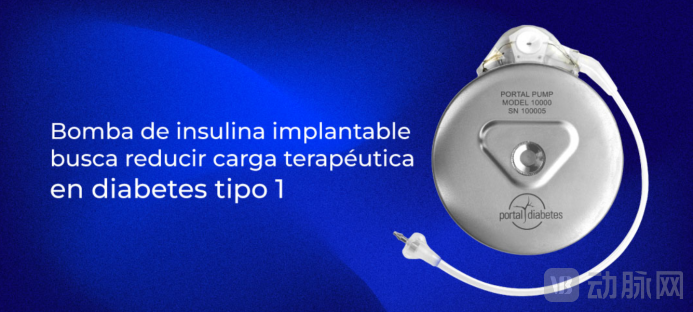
Source: portaldiabetes.com
With the continuous rise in the prevalence of diabetes, particularly Type 1 diabetes, the global insulin pump market is entering a period of rapid growth.
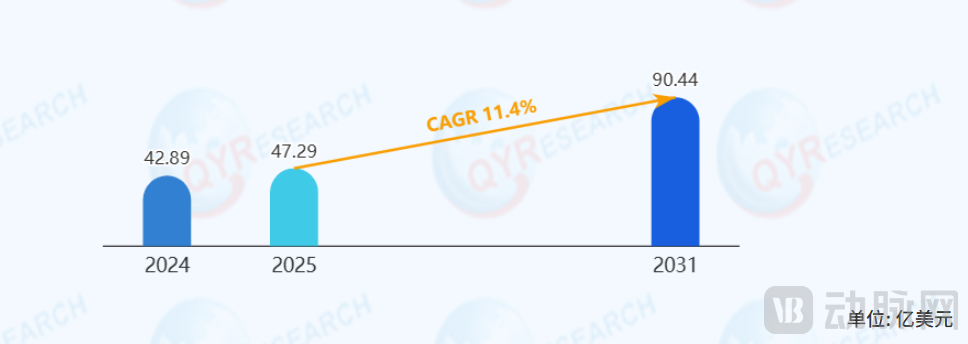
Global Insulin Pump Market Size, Source: QYR (Hengzhou Bozhi)
However, when we delve deeper into the current market landscape, we find that despite continuous technological advancements and iterations, most products on the market still fall under the "wearable" category — delivering medication through a thin tube or a patch with a needle that penetrates the skin. This not only poses risks of skin infections, foreign body sensations, and accidental device detachment, but more importantly, patients still need to inform the system in advance about meal intake (meal announcements) and physical activity (exercise announcements).
As diabetic patients increasingly demand higher quality of life and more precise physiological insulin delivery, implantable devices, with their advantages such as strong concealment and infusion pathways that better align with physiological characteristics (intraperitoneal infusion), remain a blue ocean globally and are becoming the next competitive high point in the industry.
It is worth mentioning that Portal Diabetes is not the first company to develop an implantable insulin pump — Medtronic developed a similar product, MiniMed, as early as the 1980s, which was implanted in nearly 400 patients. However, due to high costs, technical complexity, and the rapid popularity of subcutaneous insulin pumps, apart from a few patients who still use MiniMed, all other similar products have been discontinued. Recently, Medtronic has been preparing for the independent listing of its diabetes business while restoring its historical name — MiniMed.

Source: Diabetech
Insulin is the core hormone that regulates blood glucose homeostasis in the body. It is transcribed and expressed by pancreatic β-cells in the human body and then transported to the liver via the portal vein circulation, rather than being directly released into subcutaneous tissue. However, wearable insulin pumps all adopt subcutaneous infusion, where the absorption rate of insulin is influenced by blood flow, injection depth, and temperature. It takes approximately over an hour to reach its peak, which is significantly slower than the speed of physiological pancreatic secretion and has a slow metabolism. This increases the risk of blood glucose fluctuations for patients.
Source: portaldiabetes.com
In contrast, the Portal Pump™ directly infuses insulin into the peritoneal cavity.Absorption through the peritoneum, an interface with a large surface area and rich blood supply, allows insulin to take effect in a manner closer to the physiological insulin secretion pattern of the human body., which is expected to help patients achieve blood glucose targets withinIncrease by approximately 90%(Usually takes effect significantly within 15 to 20 minutes). Moreover, unlike previous implantable pumps, the Portal Pump™ enables automated insulin infusion through integration with a continuous glucose monitoring system.
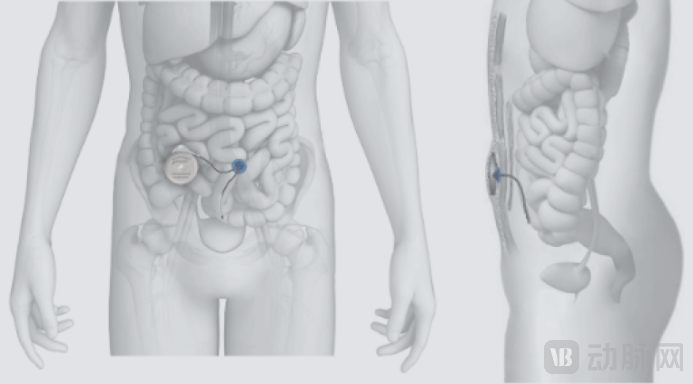
Source: portaldiabetes.com
Portal Diabetes aims to buildA Truly "Fully Closed Loop" Artificial Pancreas. Its core innovation advantages are mainly reflected in the following three dimensions:
True Physiological Infusion and Sensation-Free Treatment
Portal Pump™ is fully implanted subcutaneously in the patient's abdomen through minimally invasive surgery.(Volume is only half of MiniMed). This design eliminates the psychological and lifestyle burdens that external devices impose on patients. Moreover, since the device is fully implanted, patients are unrestricted whether they are bathing, swimming, or sleeping, greatly enhancing the concealment of treatment and quality of life.
Deep Integration of Modern CGM Technology
Portal Pump™ is not an isolated piece of hardware. Through its connection withHigh-Precision CGM Data Interconnection, the device can automatically adjust the infusion rate of insulin based on real-time blood glucose fluctuations. Combined with the pharmacokinetic advantages of intraperitoneal infusion (faster onset, smoother peaks and troughs), the system can more accurately simulate physiological insulin secretion, thereby significantly increasing the time in range (TIR) for patients' blood glucose. Users can operate it almost simultaneously with eating or even after eating, greatly reducing the risk of hypoglycemia and enhancing the freedom and flexibility of life.
"Hardware + Software + Formulation" Systematic Moat
More importantly, Portal Diabetes is building"Hardware + Software + Formulation" Three-in-One Technology Platform. Among them,Portal Insulin is a temperature-stable concentrated insulin formulation currently under development by the company.Traditional insulin formulations are prone to degradation under the high temperature (37°C) and long-term storage conditions of implantable devices, while the stability of insulin is crucial for ensuring the long-term and safe operation of the entire implantable system. Currently, Portal Insulin has entered Phase I clinical research and has completed the first patient dosing at a clinical center in the United States.
Thanks to the above-mentioned breakthrough innovations, the BDD designation granted by the FDA not only recognizes the innovative potential of Portal Pump™ in Type 1 diabetes management but also qualifies it forEmerging Technology Transition Coverage (TCET) PathwayAccelerate the future medical insurance coverage, thereby accelerating the benefits of this disruptive technology for patients.
Despite the promising prospects, the product still faces challenges before it hits the market:
Implantable devices require surgery;
Insulin needs to be replenished every 6 to 12 weeks at medical institutions.
The cost of the device and the cost-performance ratio compared with existing insulin pumps remain to be clarified.
According to the company's plan, clinical trials for this combination systemIt is expected to officially launch in the fourth quarter of 2027.. Currently, Portal Diabetes is preparing for independent production,The goal is to bring this technology to the U.S. market within the next five years or so.。
