Regend Therapeutics completes RMB 350M Series C financing, leading innovation in organ regenerative medicine
In February 2026, Regend Therapeutics Limited, a leading enterprise in the field of organ regenerative medicine, announced the completion of its Series C financing round of approximately RMB 350 million. New investors introduced in this round include seven renowned companies and institutions such as Yuze Capital, Hefei Hi-tech VC, Hongtai Fund, Hefei Industry Investment, and Gongqingcheng Fuhui. Existing shareholders, including five investment firms such as FIRHealth Capital and Tasly Capital, continued to increase their investment.
As a globally leading company integrating the research, development, and industrialization of organ function regeneration therapies, Regend Therapeutics will leverage this financing round to further advance the clinical implementation of its core stem/progenitor cell products, laying a solid foundation for accelerating comprehensive commercialization.
Regenerative Medicine Ushers in Industry Boom, Regend Therapeutics Seizes the Opportunity
Currently, regenerative medicine therapies have become a core growth driver in the biopharmaceutical industry. It is well known that traditional treatments for chronic diseases face the bottleneck of "managing symptoms without addressing the root cause." The unique advantages of progenitor cell therapy in organ repair and functional regeneration are emerging as a critical pathway to overcome this challenge. Progenitor cells are a collective term for various cell types within adult human organs that possess regenerative and repair capabilities. They exhibit the differentiation and self-renewal characteristics of stem cells but, compared to traditional stem cells, have a more defined capacity for directed tissue regeneration, offering significant advantages in drug developability. According to Frost & Sullivan forecasts, the global market size for progenitor cell therapy is projected to grow from USD 100 million in 2024 to USD 11 billion by 2030, representing a compound annual growth rate of 111.4%.
With significant policy tailwinds—including the National Medical Products Administration (NMPA) classifying progenitor cell regenerative medicine therapies as "Advanced Therapy Medicinal Products (ATMPs)," the State Council's issuance of "Regulation No. 818" permitting stem cell and progenitor cell therapies as new biomedical technologies for clinical research and translational application, and the establishment of "green channels for clinical translation" in pilot zones like Boao Lecheng—coupled with China's vast patient population, the Chinese market for progenitor cell therapeutics is expected to reach RMB 14.1 billion by 2030.
Since its founding in 2015, Regend Therapeutics has successfully established a strategic leadership position in the regenerative medicine field. Its current focus on three major areas—chronic obstructive pulmonary disease, idiopathic pulmonary fibrosis, and chronic kidney disease—is characterized by large patient populations, significant limitations of existing treatments, and strong market growth potential. Particularly in the Chinese market, driven by an aging population and increasing demand for diagnosis and treatment, these areas are poised to become some of the fastest-growing segments in the progenitor cell therapy field.
As the fourth leading cause of death globally, the burden of chronic obstructive pulmonary disease (COPD) is particularly prominent in China. According to Frost & Sullivan predictions, the global number of patients will increase to 301 million by 2030, with China accounting for 36.88% of that figure. Current COPD treatments can alleviate airway obstruction and inflammation but cannot reverse alveolar damage or restore gas exchange function, offering minimal long-term benefits to patients. This situation highlights the urgent clinical need and provides a broad market space for innovative therapies like REGEND001 from Regend Therapeutics, which focuses on "alveolar regeneration."
Although Idiopathic Pulmonary Fibrosis (IPF) has fewer patients than COPD, its mortality rate is comparable to that of cancer. Its "high malignancy" attribute elevates the priority of clinical needs. According to Frost & Sullivan forecasts, the global incidence of IPF will increase to 932,100 cases by 2030, with a compound annual growth rate (CAGR) of 6.1% from 2024 to 2030; during the same period in China, the incidence will rise to 339,900 cases, with an annual CAGR of 10.7%, significantly higher than the global average. Currently, only three drugs have been approved globally for the treatment of idiopathic pulmonary fibrosis, all of which are small molecules that inhibit enzyme activity. These small-molecule therapies can only slow the decline in forced vital capacity (FVC) but cannot prevent alveolar damage or repair damaged alveoli. This situation gives REGEND001, with its "alveolar regeneration" capability, extremely strong market competitiveness.
Chronic Kidney Disease (CKD) has become a major challenge in the global health field. According to Frost & Sullivan forecasts, the global prevalence will rise to 1.022 billion by 2030, with China's prevalence reaching 191 million by 2030. Among these cases, Diabetic Kidney Disease (DKD), as the predominant subtype, accounts for over 30% of patients and continues to grow alongside the increasing number of diabetes patients. Current treatment options, including medication, dialysis, and kidney transplantation, are still limited to slowing disease progression or replacing lost renal function but cannot reverse the underlying course of CKD. Regend Therapeutics' REGEND003, the first renal progenitor cell therapy in China to receive Investigational New Drug (IND) approval from the National Medical Products Administration (NMPA), has now entered Phase I clinical trials. This therapy uses non-invasive urine collection to obtain renal progenitor cells, which can directly regenerate nephrons. Its clinical potential has been supported by China's National Key R&D Program, positioning it to pioneer a "regenerative treatment" pathway in the untapped market of DKD.
Core Pipeline Reaches Critical Clinical Milestones, Multi-Organ Strategy Builds Technological Moat
Since its establishment in 2015, Regend Therapeutics has been guided by the mission of "developing innovative cell and gene therapy products to achieve the repair, regeneration, and enhancement of human tissues and organs," consistently driving technological industrialization based on clinical needs. Its core pipeline progress and technology platforms have formed significant differentiated advantages:
● Lung Regeneration Pipeline: REGEND001 leads global clinical progress
As the world's first autologous lung regeneration therapy, REGEND001 has initiated a series of clinical pipelines targeting major lung injury diseases, including chronic obstructive pulmonary disease (COPD), interstitial lung disease (ILD), idiopathic pulmonary fibrosis (IPF), bronchiectasis, and bronchiolitis obliterans in children. Phase II clinical trials have been completed for both COPD and IPF indications. Clinical data demonstrate that patients treated with REGEND001 showed statistically significant improvements in key efficacy endpoints compared to the control group. In addition to improved lung function and enhanced quality of life, REGEND001 has also demonstrated unprecedented advantages in repairing structural lung damage, achieving an increase in lung volume of several hundred milliliters for patients.
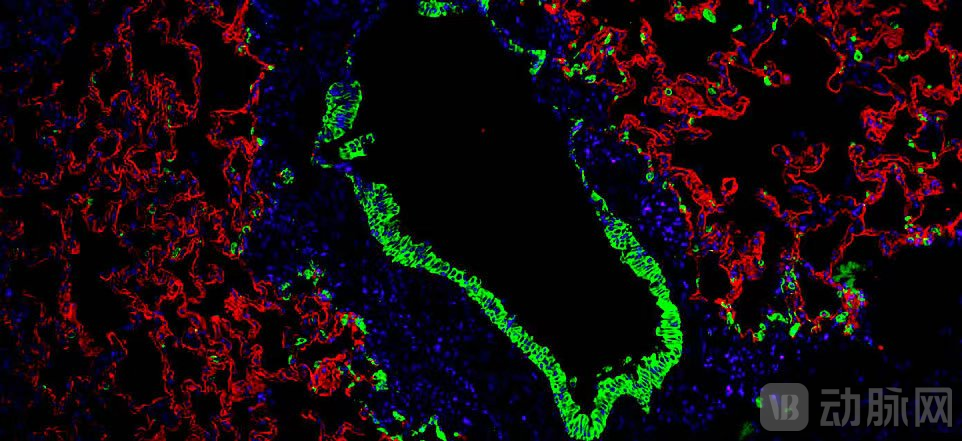
Currently, Pulmovinci, developed based on the optimized process of REGEND001, has been granted Orphan Drug Designation by the U.S. FDA for the treatment of IPF. It has also launched three "clinical translational application" projects in the Boao Lecheng Pilot Zone of International Medical Tourism in Hainan, with an approved treatment cost of RMB 150,000 per session. To date, regenerative therapy has been successfully provided to over 140 patients, preliminarily validating its commercial potential.
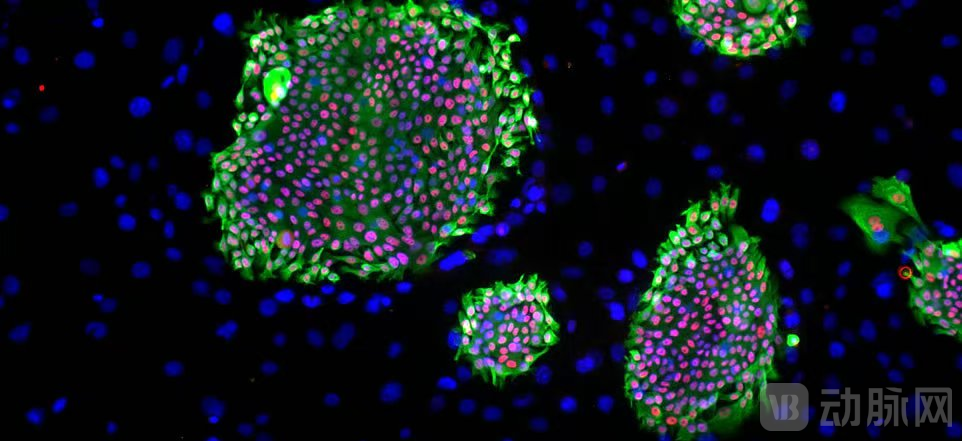
● Kidney Regeneration Pipeline: REGEND003 pioneers a new path for non-invasive regenerative treatment
Targeting chronic kidney disease (CKD), a major disease affecting over one billion people globally, Regend Therapeutics has developed REGEND003. In May 2025, it received NMPA approval to initiate Phase I clinical trials for the treatment of type 2 diabetic kidney disease (DKD) and CKD. This product utilizes a world-first "non-invasive urine-derived acquisition" technology, isolating renal progenitor cells from patient urine. These cells are expanded using the proprietary R-Clone platform and then re-infused to directly regenerate renal tubular and glomerular cells. Preclinical data demonstrate that REGEND003 exhibits potent regenerative activity, significantly improving renal tissue pathology, serum creatinine levels, and blood urea nitrogen levels.
● Technology Platform: R-Clone breaks through industry bottlenecks in cell therapy
The core driver supporting the rapid advancement of its multi-organ pipeline is Regend Therapeutics' proprietary R-Clone progenitor cell expansion platform. This platform, through its specialized systems and culture media formulations, can expand progenitor cells—obtained in only single-digit numbers from minimally invasive samples such as bronchial brushings or urine—to the tens of millions or hundreds of millions of functionally active cells required for each treatment, while maintaining the cells' stemness and organ-specific differentiation capacity. This innovative technological breakthrough overcomes the traditional cell therapy industry bottlenecks of "low expansion efficiency and loss of function" associated with primary cell culture. It has been successfully applied to the preparation of progenitor cells from various tissues and organs, including the lung, kidney, liver, uterus, testis, and nervous system, laying a critical foundation for future multi-organ regeneration initiatives.
Furthermore, independently developed systems such as the IntelCell genetic modification platform, MIX0109 cell rejuvenation technology, and the REGEN-WK AI medical decision platform collectively form a comprehensive regenerative medicine R&D ecosystem. Through a "standardized + modular" architecture, this ecosystem transforms regenerative medicine research and development into a scalable and replicable industrialization process. This enables Regend Therapeutics to rapidly develop therapies and achieve commercialization across multiple disease areas based on the same core technological framework, thereby constructing a strong technological moat and sustaining its innovation advantage.

Regend Therapeutics Product Pipeline Diagram
