Villanelle Life secures tens of millions in Pre-A funding to accelerate AI-driven precision diagnostics deployment
Recently, Villanelle Life completed a new round of financing. The proceeds from this round will primarily be used to advance the company's AI-driven precision diagnostic products through multi-center clinical validation, regulatory submissions, and large-scale application deployment.
As a platform company focused on AI-driven diagnostics originating from real-world clinical challenges, Villanelle Life has long concentrated on addressing "unmet needs in clinical decision-making." Villanelle Life seeks to establish a sustainable pathway for translating medical research into clinical practice, bridging scientific data, algorithmic models, and real-world clinical application.
Founded in Shanghai, Villanelle Life is positioned as a platform company integrating clinical research services with the translation of research findings into practical applications. Leveraging its proprietary "integrative omics + AI" big data analysis platform, Villanelle Life collaborates extensively with numerous top-tier hospitals in China to identify diagnostic and decision-making challenges with translational potential within real-world clinical settings.
Villanelle Life's core data and algorithm team consists of members holding undergraduate degrees from China's premier universities (Tsinghua, Peking, Fudan, Shanghai Jiao Tong) and doctoral degrees from renowned overseas institutions. Team members have published multiple research findings as first authors or corresponding authors in leading international journals. Building on long-term accumulated clinical data and algorithmic expertise, Villanelle Life is progressively advancing mature findings validated through research collaborations toward product development.
Currently, Villanelle Life has established collaborative relationships with dozens of top-tier hospitals in China, including Ruijin Hospital and Zhongshan Hospital affiliated with Fudan University, forming a collaborative network spanning data analysis, clinical validation, and translational application.
In terms of its product pathway, Villanelle Life has not remained at the level of a single algorithm or tool. Instead, it is advancing the systematic development of AI-assisted diagnostic products centered around specific clinical decision-making scenarios.

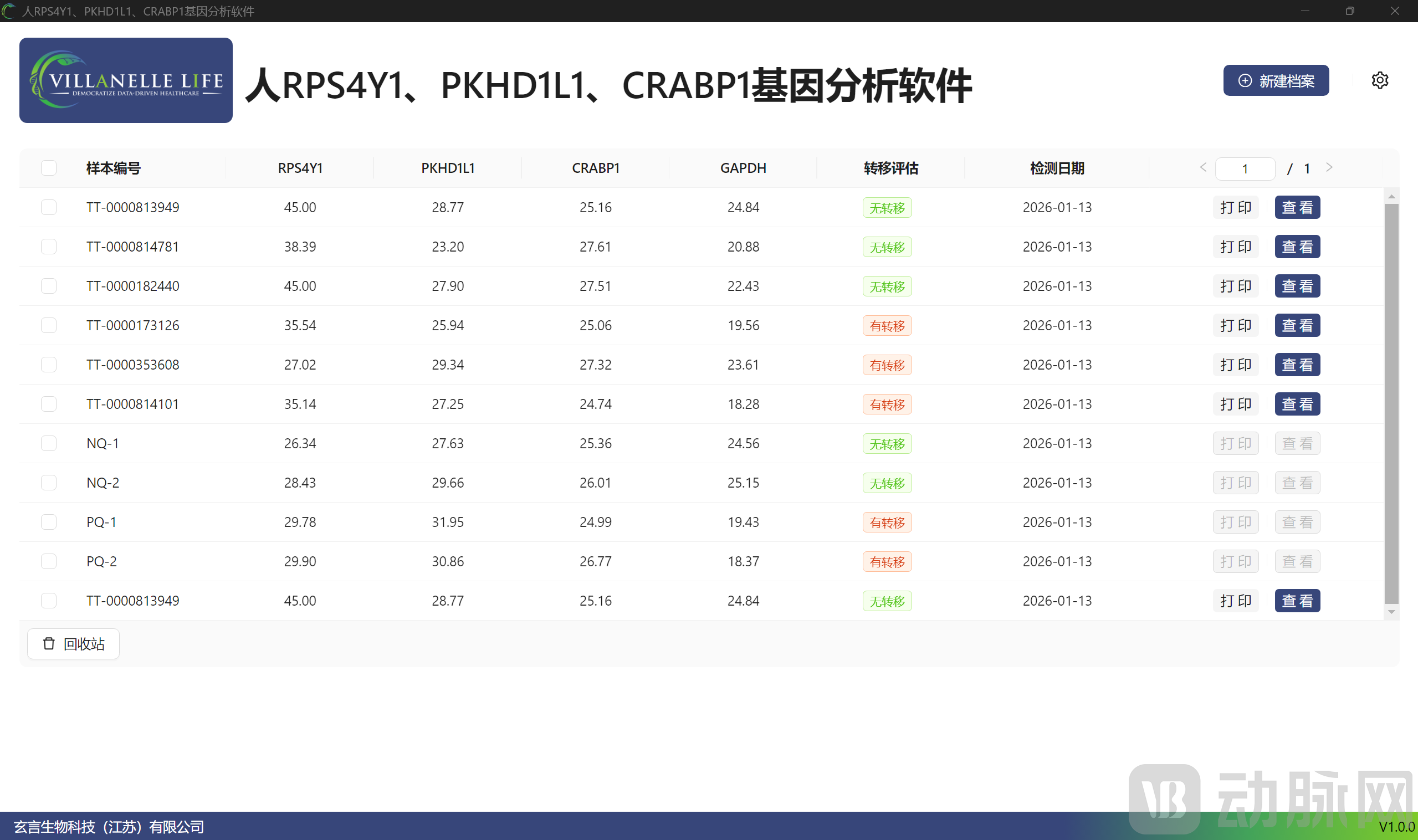
Taking thyroid cancer as its entry point, Villanelle Life has launched an AI-based lymph node metastasis detection kit, accompanied by AI software to assist in clinically assessing metastasis risk and optimizing surgical decision-making. This product originated from an official scientific research translation project conducted in collaboration with Ruijin Hospital. It has already been deployed for clinical application across multiple regions in China, including Shanghai, Jiangsu, Shandong, Hebei, and Jiangxi. Having obtained "dual" certification from the China National Intellectual Property Administration, the product has entered the application process for the Innovative Medical Device Special Review Pathway, while simultaneously pursuing CE marking and the FDA's Breakthrough Devices Program (BDD) internationally.
Building on this foundation, Villanelle Life is steadily advancing a robust pipeline, including detection of lymph node metastasis in esophageal cancer, recurrence monitoring for urothelial carcinoma, detection of lymph node metastasis and recurrence in breast cancer, and rapid triage for acute chest pain. This forms a product structure characterized by "one clinically validated product plus multiple scalable translation pathways." The underlying logic is to continuously serve more diseases and indications using the same platform capabilities, significantly reducing the repetitive research and development costs of advancing new pipelines from the ground up.
Furthermore, leveraging its independently constructed AI-driven multi-omics integration platform, Villanelle Life has identified novel targets with translational potential in advanced recurrent bladder cancer and triple-negative breast cancer. By optimizing molecules through AI algorithms and formulating combination strategies with established targets, the company has entered into a strategic collaboration with the School of Pharmaceutical Sciences Fudan University. This partnership aims to advance systematic pharmacodynamic and safety evaluations, along with related validation research, in preparation for Investigational New Drug (IND) applications.
