Blue Sail Lithonic™ Intravascular Shock Wave System Receives CE Certification, New Tool for Complex Coronary Artery Calcification Treatment

Bluesail Medical
Producers of Medium-Low Value Consumables and High-Value Medical Devices
In January 2026, the cardiovascular division of Shandong Blue Sail Plastic and Rubber Co., Ltd., specifically its subsidiary in Singapore,Lithonic™ Coronary Intravascular Lithotripsy (IVL) Therapy System Developed by Biosensors Interventional Technologies Pte. Ltd. Officially Receives EU CE Certification. This marks the international recognition of the system in terms of clinical safety and efficacy, and also provides a new tool for the treatment of complex coronary artery calcified lesions worldwide.
1Calcified Lesions Remain a "Tough Nut" for PCI
Percutaneous Coronary Intervention (PCI) is a cornerstone treatment for coronary atherosclerotic heart disease.
However, the presence of coronary artery calcification (CAC) remains one of the main obstacles affecting both the immediate success of PCI procedures and long-term prognosis. According to the "Chinese Expert Consensus on Diagnosis and Treatment of Coronary Artery Calcified Lesions," coronary angiography shows moderate to severe calcification in about one-third of lesions, and with the aging population, its detection rate and complexity continue to rise.
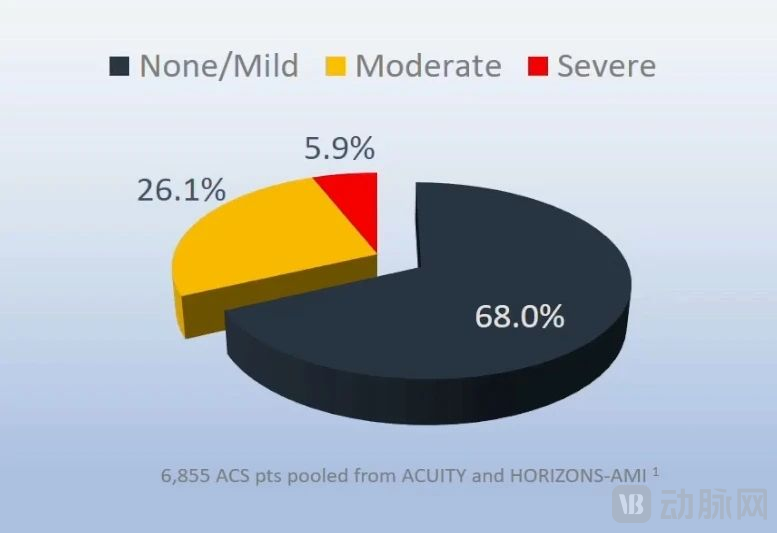
In 6,855 patients, including those in the ACUITY and HORIZONS-AMI studies, the incidence of moderate to severe coronary artery calcification was 32%, with 5.9% being severely calcified lesions.
These hard calcified plaques significantly increase the difficulty and risk of PCI. A prospective cohort study led by Fuwai Hospital in 2023 (sample size > 26,000 cases) quantified this risk: 15.33% of patients had moderate to severe calcified lesions, and the target vessel failure rate in patients with moderate to severe calcification was 5.31%, higher than that in patients with no or mild calcification (4.65%).
At the same time, stent underexpansion and malapposition caused by calcification are well-defined risk factors for in-stent restenosis and late thrombosis, significantly increasing the long-term target lesion failure rate and the incidence of major adverse cardiovascular events.
Despite the availability of existing clinical techniques such as rotational atherectomy and cutting balloons, these methods commonly suffer from limitations such as a steep learning curve, poor device deliverability, ineffectiveness against deep calcification, or higher risks of complications. How to safely, efficiently, and thoroughly address calcification, particularly severe circumferential calcification, remains a tough challenge yet to be overcome in the field of interventional cardiology.
The emergence of Intravascular Lithotripsy (IVL) technology provides a new option for treating severely calcified lesions. Its core principle differs from traditional mechanical grinding or cutting; it generates a hydroelectric effect in a liquid medium by instant discharge inside a balloon catheter, exciting short-duration, high-amplitude pulsed sound waves (shock waves).
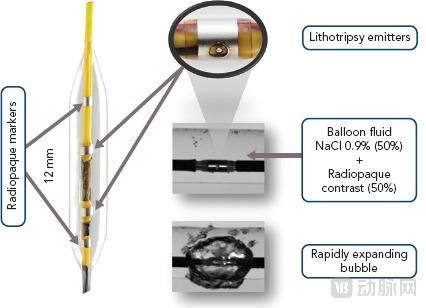
IVL System Intravascular Lithotripsy Procedure Source: Interventional Cardiology
This "shockwave lithotripsy"-style mechanism can effectively modify both superficial and deep calcifications, achieving safer and more uniform vascular pre-treatment, laying the foundation for subsequent stent implantation and optimized expansion, and is changing the procedural strategy of complex PCI globally.
2Lithonic™ System: Precise Shock Waves, Targeted Calcification
Lithonic™ Coronary IVL System is an integrated innovative solution based on IVL technology. It is not a single device but a complete system designed to address severely calcified lesions that are difficult to expand with balloons, aiming to reduce the risk of complications such as vascular perforation and no-reflow by using controlled acoustic energy at the source.
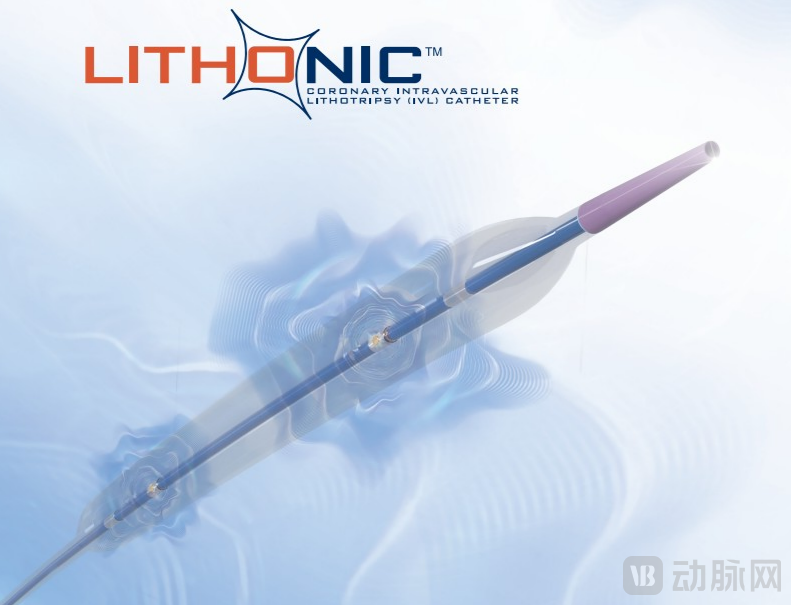
Lithonic™ System Model Diagram Source: Shandong Blue Sail Plastic and Rubber Co.,Ltd.
Core Components and Engineering Advantages of the System:
● Multi-Specification Shock Wave Balloon Catheter: Offers 18 specifications ranging from a minimum diameter of 2.0mm to a globally exclusive length of 15mm, comprehensively covering complex scenarios from small vessels and diffuse lesions to severe circumferential calcification.
● Patent Liquid Electric Pulse Generator: A lightweight and rechargeable host device that can stably generate and control the shockwave energy required for treatment. The simple interface design facilitates quick deployment and use.
● Optimized Energy Emission and Control System: Adopting an exclusive patented electrode design to ensure uniform distribution of shock wave energy within the balloon. The independent trigger design of the handle and foot pedal allows the operator to precisely control the emission timing, preventing catheter displacement and enhancing operational safety and efficiency.
● 5F Guiding Catheter Compatibility: The entire series of balloon catheters are compatible with the clinically commonly used 5F guiding catheter, reducing the requirements for surgical access and enhancing the system's versatility and convenience.
These subsystems work together to enable Lithonic™ to achieve precise, controllable, and predictable structural modification of calcified plaques.
Its core clinical value lies in significantly improving the compliance of subsequent balloon expansion through this optimized pre-treatment, creating a more ideal and regular lumen environment for the implantation of drug-eluting stents, ultimately promoting complete stent expansion and uniform apposition, which is key to reducing long-term adverse events.
3Dual Product Matrix Layout in China and Overseas
Currently, Shandong Blue Sail Plastic and Rubber Co., Ltd. is accelerating the clinical adoption and market coverage of IVL technology through a clear dual-product matrix strategy both in China and internationally.
Previously, its subsidiary, Blue Sail Boyuan's SoniCracker™-CL Coronary IVL System, as one of the first domestically produced coronary IVL products to be launched in China, received approval from the National Medical Products Administration (NMPA) in 2024. This system is currently the most comprehensive range of shockwave catheters available in China and has already been applied in the domestic market.
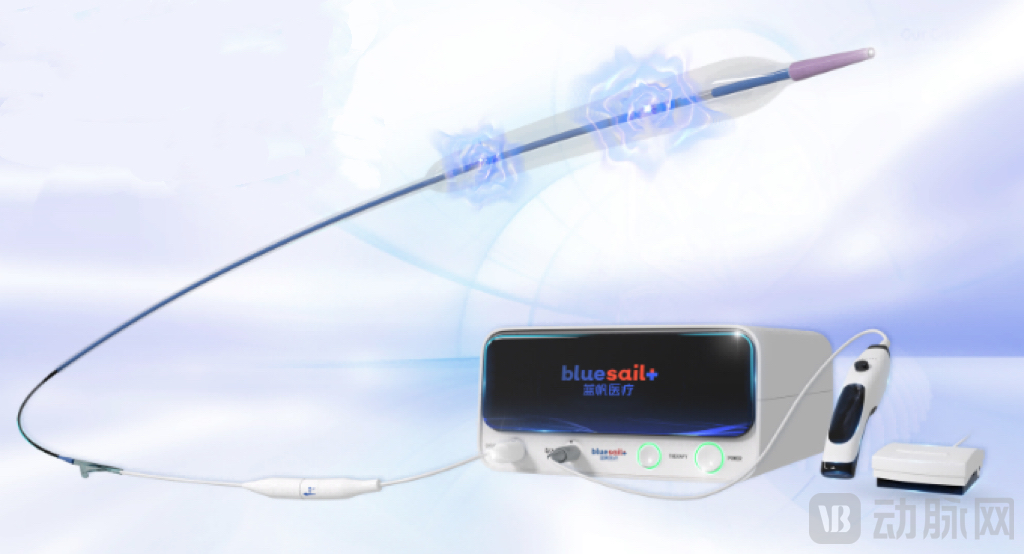
SoniCracker™-CL Coronary IVL System Image Source: Shandong Blue Sail Plastic and Rubber Co.,Ltd.
The company did not stop at going public. In 2025, it launched a clinical study named SONICURE in collaboration with top hospitals, aiming to further explore the application of IVL in extremely complex calcified lesions and continuously accumulate high-level Chinese evidence-based medical data.
The CE certification of Lithonic™ marks a key step in the company's "Innovate Overseas" strategy, signifying its official entry into the strictly regulated European and global markets.
This layout not only meets the regulatory and clinical needs of different regions but also highlights the R&D strength and market competitiveness of Shandong Blue Sail Plastic and Rubber Co., Ltd. in the global high-end interventional device field. With the increasing penetration rate of coronary artery calcification treatment and growing clinical recognition, the IVL market has broad prospects. The dual product lines of Shandong Blue Sail Plastic and Rubber Co., Ltd. are expected to share in this growth dividend.
