21-Day Long-Lasting CGM Arrives! Tangjian Medical Brings Blood Glucose Monitoring into the "Three-Week Era"
In the current global market, the wear cycle of mainstream continuous glucose monitoring systems (CGM) generally stays within the range of 14 to 16 days. However, a Chinese innovative company, SugarMed Technology (Suzhou) Co., Ltd. ("SugarMed"), has successfully extended this cycle to 21 days with its first self-developed CGM product, Instara-1, breaking through the industry’s key upper limit.
The product made its debut at the 2nd Asian Conference on Innovations in Diabetes Management (ATTD-Asia 2025) held in Singapore from December 9 to 11, 2025, drawing continuous attention from global experts and channel partners in the diabetes field. Based on this technological breakthrough, Mao Jian, founder of Tangjian Medical, stated in an exclusive interview with VCBeat, "Our goal is not merely to achieve 'China-produced alternatives,' but to comprehensively surpass through fundamental technological breakthroughs."
 The 2nd Asian Conference on Innovative Therapies for Diabetes ManagementScene Source: SugarSimple Medical
The 2nd Asian Conference on Innovative Therapies for Diabetes ManagementScene Source: SugarSimple Medical
1The Origin of Entrepreneurship: Not to Replace Domestic Products, but to Solve Global Problems
SugarMed was founded in 2021, with dual R&D centers in Shanghai and Silicon Valley, focusing on the full process of R&D, production, and commercialization of CGM systems.
Its R&D foundation comes from a technical team with over two decades of experience in biosensing, microelectronics, and materials science. The core members mostly come from multinational medical device companies, where they have long been engaged in the research and development of electrochemical sensing and Micro-Electro-Mechanical Systems (MEMS), establishing a solid foundation in underlying technologies. Currently, more than 50% of the company’s staff are R&D personnel. The team focuses on the fundamental principles of sensors, aiming to challenge established market rules.
As Mao Jian said: "Our choice of CGM is not because it is easy to do, but because many underlying technical issues have not been truly resolved for a long time. The industry technology has entered a plateau, and the majority of patients worldwide still cannot afford or access high-quality continuous glucose monitoring devices. This represents a significant structural gap."
According to 2025 data from the International Diabetes Federation (IDF), the number of adults aged 20-79 with diabetes worldwide is approximately 589 million, with Chinese patients accounting for 1/4 of the total. The majority of these individuals need to cover the cost of using CGM out-of-pocket, while the price of a single mainstream sensor typically ranges from tens to hundreds of dollars and requires replacement every 1-2 weeks. For patients who need lifelong diabetes management, such expenses create significant pressure both in terms of financial burden and long-term accessibility.
It is against this backdrop that Tangram Medical abandoned the path of "creating a Chinese version of the 14-day CGM" from the very beginning, instead focusing its technical route on the most challenging foundational problems in the industry:How to improve the battery life and signal stability of sensors simultaneously without increasing costs.
221-day battery life, 8.2% accuracy, 15-minute startup: "Super Sensor" breakthrough in long-term CGM challenges
The development of CGM technology is an engineering history of continuously finding balance among miniaturization, longevity, and precision.
From Medtronic's first FDA-approved CGM system in 1999, to Dexcom extending the wear duration to 7 days in 2004, and then Abbott’s launch of the FreeStyle Libre with 14-day wear and no calibration required in 2014, each breakthrough has relied on systematic iterations in materials science, enzymology, electrochemistry, and signal processing.
However, since Libre ushered in the 14-day era, the industry's technical curve has almost stagnated. Current mainstream products still cluster around this boundary: Abbott's Libre series has established a mature mass-production standard of 14 days; Dexcom’s G-series operates within the 10-15 day range, relying on robust algorithms to balance signal attenuation; while other competitors, including Yuwell (16 days) and Sinocare (15 days), have focused their innovations primarily on optimizing within the 14-16 day range.
This means that extending the long-term stability of sensors in the body, rather than relying solely on algorithmic compensation or material replacement, has become the core technical bottleneck faced by the entire industry. To overcome this bottleneck, it is necessary to address three major core challenges that have long constrained the industry:
1. Low Power Consumption and Miniaturization Physical Contradiction。As sensors continue to become thinner and lighter, battery space is extremely compressed, and any extension of battery life requires the redesign of circuits and signal processing strategies.
2. Long-term wear biocompatibility challenges.The longer the sensor is worn, immune cells such as macrophages around the probe continuously encapsulate the foreign object, forming a fibrous capsule, which leads to glucose data drift and reduced accuracy. Additionally, avoiding bacterial growth that may cause folliculitis, cellulitis, and other infections, while ensuring medical adhesive materials remain stable and do not fall off for over three weeks, as well as mitigating risks from sweat, humidity, and skin sensitivity reactions, all present significant challenges.
3. "Essential Bottleneck" of Long-term Stability of Enzyme Sensors.This is also the most challenging problem. Glucose oxidase gradually loses activity in the interstitial fluid environment, leading to signal drift; once the drift becomes irreversible, it is difficult to maintain long-term accuracy.
The breakthrough of SugarSimplify Healthcare starts precisely from this fundamental bottleneck. The team has reconstructed a "super sensor" system targeting long-term stability: by optimizing the underlying structure, slowing down enzyme activity attenuation, enhancing signal processing capabilities, and coupling with a low-power microelectronic system, the sensor maintains stable output over a longer period.
On this basis, its independently developed TS-1 super biosensor can continuously operate under high load for more than 30 days in experimental conditions, with extremely low signal attenuation, laying a core foundation for the performance upgrade of CGMS products.

Instara-1 Product Image Source: Sugar Medical
This breakthrough in underlying technology has ultimately coalesced into three verifiable core advantages in the first product, Instara-1:
● 21 Days: The wear cycle reaches 21 days, extending one week longer than the current mainstream 14-day products. This means that patients can reduce the number of sensor replacements by nearly one-third each year, which helps to lower the risk of skin irritation, infection, and long-term usage costs caused by frequent replacements. It provides a more stable and sustainable monitoring foundation for patients who require lifelong blood glucose management.
● 8.2%: During the 21-day wear period, its MARD value (Mean Absolute Relative Difference) remains as low as 8.2%. This level of accuracy meets international high standards. Even during extended wear, the device continues to provide highly reliable glucose data, offering credible data support for clinical medication adjustments, efficacy evaluations, and patient self-management.
●15 minutes: Sensor activation time is significantly reduced to 15 minutes, a decrease of 45 minutes compared to the traditional product's 60-minute wait time. In hospital scenarios where professionals assist with wearing, users no longer need to wait for an extended period, further enhancing the user experience.
In the industry, it has long been believed that "long cycle" and "high precision" are difficult to achieve simultaneously. However, Instara-1 has found a rare and verifiable balance between the two through fundamental innovation.
3CE certification obtained, global registration accelerating
The global CGM market has now approached nearly 20 billion US dollars and is growing at an annual rate of nearly 20%. However, regional differences in penetration remain significant — North America and Western Europe, supported by mature medical insurance systems, are in high-penetration areas, while in emerging markets such as Asia and Latin America, the popularity of CGM remains low due to limited payment capabilities and higher out-of-pocket proportions.
Under such structural differences, Sugar Medical's 21-day product has a natural market orientation. Without increasing hardware costs, the usage cycle is extended by 50%, which means users' annual spending is significantly reduced, making it particularly appealing to price-sensitive markets.
"For B-end clients such as medical institutions and chronic disease management organizations, a longer wearing cycle means lower follow-up frequency and consumables management costs," analyzed Mao Jian. "For C-end users, it directly reduces annual expenses and replacement frequency, enhancing the user experience."
Product advantages have also attracted continuous attention from capital:
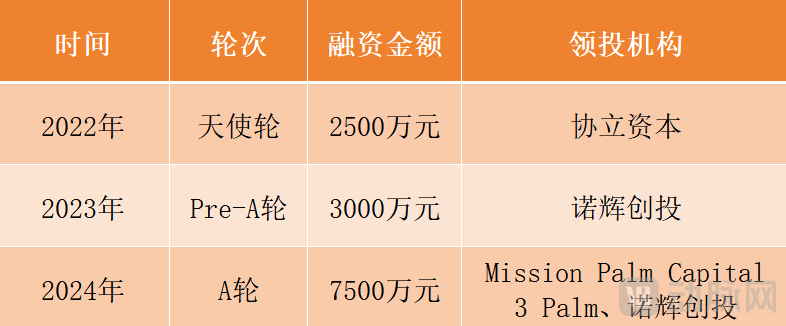
Overview of SugarJian Healthcare Financing Journey
As of now, the company's cumulative financing has reached nearly 130 million RMB. Mao Jian frankly stated, "What capital values is the underlying technical capability and a clear product roadmap."
On the technology front, the company is also continuously building its own technological barriers. As of 2025, it has applied for 34 patents, covering core components such as sensors and applicators, reflecting a comprehensive layout of systematic innovation around the CGMS.
It is reported that the first CGM product, Instara-1, developed by SugarMed, has officially obtained the CE certification issued by the British Standards Institution. The product’s entry into the European market has moved from the preparation stage to the actual implementation phase. According to the company's plan, in the first half of 2026, registration and approval will continue to be advanced in Saudi Arabia, India, Russia, Canada, and the UK, gradually completing the first round of compliance arrangements for major growth markets. Meanwhile, the domestic registration application for Instara-1 has been accepted by the National Medical Products Administration in China.
It can be foreseen that once the 21-day long-cycle, high-precision CGM achieves scale supply, it will push the entire industry to accelerate technological iteration, driving CGM competition into a new phase of "endurance battle" from the previous "accuracy battle" and "price battle."
4The Next Breakthrough: From Blood Sugar to Metabolic全景, and Then to 60-Day续航
TangJian Healthcare's layout does not stop at blood glucose monitoring.
The company is simultaneously advancing three dynamic metabolic monitoring products: Continuous Uric Acid Monitoring (CUA), Continuous Ketone Monitoring (CKM), and Continuous Lactate Monitoring (CLM), attempting to extend from a single blood glucose metric to multi-dimensional metabolic monitoring, building a more complete closed-loop for metabolic health management. Ma Jian stated, “Blood glucose is just the starting point; we will explore more metabolism-related indicators to achieve more comprehensive health management.”
This multi-dimensional product matrix not only enriches the company's revenue structure but also creates mutually reinforcing synergies in technology platforms, user education, and channel systems, further solidifying its core barriers in the metabolic monitoring field.
In addition, to meet future growth, Sugar Medical has simultaneously advanced its production capacity construction. The annual capacity of the first production line can reach 2 million sets, providing a strong guarantee for market supply after the product launch.
At the end of the interview, Mao Jian spoke about future plans with a longer time horizon: "We will continue to challenge the battery life of CGM, aiming to extend the wear cycle of a single sensor beyond 60 days." It is reported that the design and development of the next-generation product have been completed and are expected to officially launch in 2027.
From 21 days to 60 days of battery life, if this goal is achieved, it will not only represent a numerical change but may also fundamentally transform diabetes management—shifting from "frequent monitoring" to "long-term continuous monitoring," and from "disease management" to "maintaining a healthy常态."
In the current industry coordinate system, Sugar Medical has clearly marked its position. With longer endurance, lower cost, and more accurate data, it is practicing the same goal: to make better health management tools accessible and affordable for more diabetes patients.
