The Rise of a New Track in Radiopharmaceuticals: NUCLIDIUM, with Copper at its Core, Raises $99 Million to Build a Closed-loop Diagnosis and Treatment System
Against the backdrop of the continuous deepening of precision medicine, a class of bifunctional radiopharmaceuticals that "can both illuminate lesions and precisely target them" is emerging.Integrated Diagnosis and Treatment(Theranostics, Therapy + Diagnostics). This concept has frequently appeared in the field of nuclear medicine in recent years and is gradually becoming a new direction for overcoming the bottlenecks in the diagnosis and treatment of solid tumors.
And copper (Cu) isotopes, due to their excellent imaging and therapeutic properties, are becoming a core element of great interest in this technical pathway.
Recently, Nuclidium AG, a biopharmaceutical company headquartered in Basel, Switzerland, announced the completion of a CHF 79 million (approximately EUR 84 million / USD 99 million) Series B financing round, jointly led by Kurma Growth Opportunities Fund, Angelini Ventures, Wellington Partners, and Neva SGR.
Novartis Veteran Expands Territory, Builds Team with Both Basic Research and Clinical Backgrounds
NUCLIDIUM is a biopharmaceutical company focused on copper isotope theranostics technology. Established in 2017, its headquarters is located in Basel, Switzerland, with an operations center in Germany.A diagnostic-therapeutic integrated platform covering multiple solid tumor indications has been built with "copper" as the core radionuclide., attempting to push nuclear medicine from a "niche tool" towards a more widespread and precise mainstream clinical model.
The founding team of NUCLIDIUM consists of scientists with both basic research and clinical backgrounds, ensuring that its platform achieves an organic combination of technological innovation and clinical feasibility.
Co-founder and CEO Dr. Leila Jaafar has a background in nuclear engineering and radiochemistry. She believes,Copper isotopes have obvious advantages in terms of imaging quality, safety, and production scalability. Their physical properties are considered ideal, and their production pathways are clear, holding the potential to break the current reliance on actinide elements (such as177Lu、225Ac) Highly dependent on supply bottlenecks.
Another co-founder, Professor Gustav von Schulthess, is the former Director of Nuclear Medicine at the University Hospital Zurich and has also served as a visiting professor of radiology at Stanford University. With over two decades of extensive experience in PET imaging and radiopharmaceutical development, he currently acts as the company's medical advisor, guiding clinical development processes and regulatory compliance to accelerate the platform’s clinical translation and market access.

Figure 1: Portrait of Co-founders (Left: Leila Jaafar, Right: Gustav von Schulthess)
At the same time, NUCLIDIUM continues to strengthen its commercial execution capabilities and seeks potential external BD collaborations.
On November 20, 2024, Tony Rosenberg, former Senior Vice President of Novartis and seasoned executive who once led Novartis’ international mergers and acquisitions and licensing operations, joined the company's board of directors and was appointed as chairman. Leveraging his decades of experience in M&A negotiations and business networking within global pharmaceutical enterprises, NUCLIDIUM is accelerating early-stage business discussions with potential partners while actively laying the groundwork for future pipeline internationalization, commercial licensing, and capital collaborations, propelling its copper-centric integrated diagnostics and therapeutics platform towards the global market.
From 3.3 Hours to 2.6 Days: Copper Isotope Combination Reshapes Diagnostic and Treatment Time Window
In the field of nuclear medicine, the separation between diagnosis and treatment has always been a key bottleneck restricting the development of integrated diagnosis and treatment.Traditional radionuclides often struggle to meet the dual demands of imaging and therapy.——18Although F-labeled tracers provide clear imaging, their ultra-short half-life of 1.8 hours limits the clinical application radius; while ⁸⁹Zr extends the imaging window, its prolonged metabolic period of 78 hours introduces unnecessary radiation burden. Therapeutic radionuclides such as 177Lu are constrained by complex production processes and stringent storage and transportation conditions, making large-scale application difficult to achieve.
The innovative breakthrough of NUCLIDIUM lies in the clever utilization of the property combination of copper isotopes.The diagnostic use of ⁶¹Cu has a golden half-life of 3.3 hours.This optimal time window not only ensures the effective distribution of the tracer within the body but also enables regionalized production through conventional medical cyclotrons.The therapeutic radionuclide ⁶⁷Cu, with a half-life of approximately 2.6 days, aligns with the biological requirements for tumor treatment. It ensures effective accumulation of the drug in the target tissue while facilitating cross-regional distribution.
More crucially,This pair of "twin" radionuclides can utilize identical targeting molecules and chelation technologies, enabling true "homologous matching" between diagnostic imaging and treatment plans, significantly reducing clinical translation risks.
Compared with the traditional model of "multi-nuclide patchwork," NUCLIDIUM provides a more concise, scalable, and clinically relevant integrated solution for diagnosis and treatment. NUCLIDIUM is also known in the industry as the "Copper Workshop" — not only because of its focus on copper, but also because it aims to establish copper as the foundational material for a new paradigm in nuclear medicine.
Using copper as a medium, build an "integrated diagnosis and treatment" product platform
With the "twin combination" of copper isotopes at its core, NUCLIDIUM has built a radiopharmaceutical platform that spans diagnostics and treatment, enabling seamless transition from precise imaging to personalized therapy.
The current R&D pipeline covers multiple high-demand cancer indications such as prostate cancer, neuroendocrine tumors, and breast cancer, with a focus on target designs addressing both the tumor itself and its microenvironment, gradually expanding the breadth of indications and the depth of diagnosis-treatment synergy.
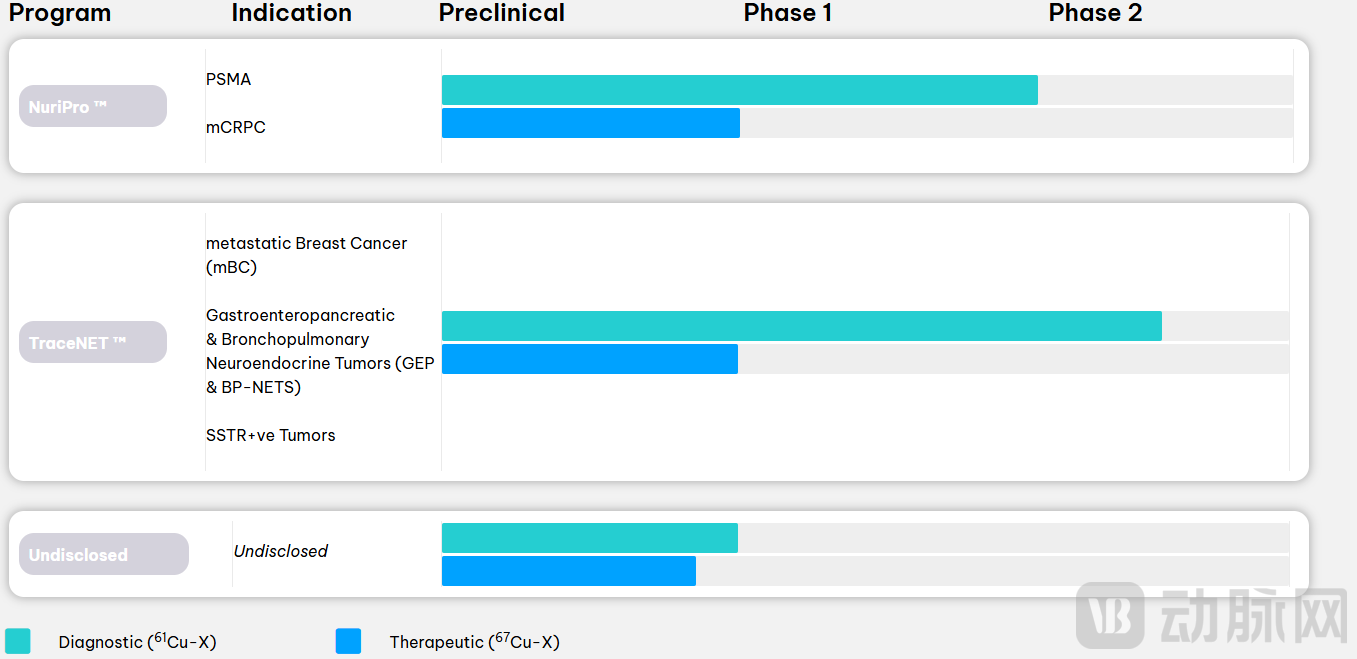
Figure 2: Overview of NUCLIDIUM Pipeline Progress
NuriPro
The first product to enter clinical trials, NuriPro, targets Prostate-Specific Membrane Antigen (PSMA) and is mainly used for metastatic castration-resistant prostate cancer (mCRPC). The expression level of PSMA in prostate cancer cells is much higher than in normal tissues, providing an ideal target for imaging and treatment. The diagnostic component of NuriPro, ⁶¹Cu, has excellent delayed imaging capabilities and can detect small lesions; its therapeutic component, ⁶⁷Cu, maintains the same ligand structure, with the potential to achieve consistency from "imaging to efficacy" at the molecular level. It has currently entered Phase I/II clinical trials.
TraceNET
Targeting Various Solid Tumors, NUCLIDIUM Develops TraceNET, Focusing on the Key Receptor Target SSTR2 (Somatostatin Receptor Subtype 2). Significant SSTR2 expression is observed in breast cancer (especially ER+/PR+/HER2- type) and multiple types of neuroendocrine tumors (GEP-NETs, BP-NETs), offering both imaging and targeting value. Compared to traditional agonists, TraceNET employs an SSTR2 antagonist structure, providing stronger binding affinity and clearer imaging information. Currently, its ⁶¹Cu diagnostic agent has entered Phase I/II clinical trials, while the ⁶⁷Cu therapeutic agent is in Phase I.
AlphaFlare
For the early detection and precise treatment of highly aggressive solid tumors, NUCLIDIUM is advancing the AlphaFlare project, a platform targeting integrins—cell adhesion molecules highly expressed in various tumors such as lung cancer and pancreatic cancer. Leveraging the imaging sensitivity of ⁶¹Cu, AlphaFlare can identify early lymphatic or distant metastases, supplemented by a therapeutic regimen using ⁶⁷Cu. It is currently in the preclinical development stage.
Kalios
In addition, NUCLIDIUM is also exploring targets related to the tumor microenvironment. The Kalios platform focuses on cancer-associated fibroblasts (CAFs), which are widely present in epithelial-derived tumors (such as breast cancer and lung cancer) and are involved in tumor progression and immune suppression. The ⁶¹Cu-labeled product from Kalios has demonstrated excellent contrast in preclinical studies and can be used to identify micrometastases in the liver and peritoneum. Further development of therapeutic components will proceed concurrently.
Overall, Nuclidium's platform-based product strategy has significant versatility and replicability.A unified copper ligand structure not only facilitates rapid target switching but also enables pathway commonality in synthesis processes, quality control, and regulatory submissions, reducing marginal development costs.At the same time, the natural synergy of copper-copper paired isotopes in physical properties, clinical behavior, and circulation standards also makes the implementation of theranostics more advantageous in terms of engineering.
Integration of Industry, Academia, and Capital, Unblocking the "Copper Chain Industry Ecosystem"
To establish a full-process closed loop from radionuclide preparation to clinical validation, NUCLIDIUM has built a closely collaborative "copper chain ecosystem," integrating key forces across the upstream and downstream industries to achieve a high level of synergy in research, production, clinical application, business, and capital. This model not only enhances the development efficiency of copper-based theranostic integrated products but also enables NUCLIDIUM to quickly gain a solid foothold in the highly competitive radiopharmaceuticals sector.
In the core production环节, Nuclidium has reached a cooperation with the United States PharmaLogic Holdings, which is responsible for61The production and distribution of Cu tracers have the capability to be manufactured without relying on cyclotrons and can be stored at room temperature, significantly reducing logistics and on-site synthesis costs.
To strengthen the industrialization of the imaging sector, NUCLIDIUM has also brought in Guerbet, a leading French contrast agent company, as a strategic partner. Guerbet not only provides imaging capabilities and a global market network but also directly participates in corporate governance by appointing an observer to the NUCLIDIUM board of directors, promoting the synergy between technology transfer and business expansion.
During the clinical validation phase, NUCLIDIUM collaborated with the Hoag Family Cancer Institute in the United States and University Hospital Basel in Switzerland to conduct clinical trials for prostate cancer (NuriPro) and neuroendocrine tumors (TraceNET), respectively.
Notably, this "copper chain ecosystem" is not driven by a single technology but is rather an integration of capital, manufacturing, and clinical strengths. Since its establishment, Nuclidium has completed multiple rounds of financing, attracting top European life science funds such as Kurma Partners, Angelini Ventures, and Eurazeo Health, with cumulative financing exceeding 100 million US dollars.
These industry capitals not only provide financial support but also deeply empower them in terms of clinical transformation resources, European pharmaceutical regulatory compliance, and manufacturing networks, assisting them in simultaneously advancing a diversified product layout in the EU and North America.

Table 1: Overview of NUCLIDIUM's Investment and Financing Situation
Through this highly collaborative "industry-academia-research-capital" chain, NUCLIDIUM not only accelerated the development pace of copper-based integrated diagnostic and therapeutic products but also established a new ecological paradigm for platform-based radiopharmaceutical companies.
The Breakthrough Path of China-Produced Radiopharmaceuticals Inspired by Copper Chains
China's radiopharmaceuticals industry is entering a critical transition period towards integrated diagnosis and treatment. With the successful breakthrough in the large-scale production technology of the 64Cu isotope by China Isotope & Radiation, a subsidiary of China National Nuclear Corporation, in 2023, China now has the confidence to compete on the same stage as international giants in this field.
This technological breakthrough quickly triggered industrial resonance — Xiantong Pharmaceutical obtained clinical trial approval for 64Cu-Dotatate Injection (a generic version of Detectnet®) in May 2024, followed by Jiangsu Huayi Technology's similar product entering the review stage in February 2025, demonstrating Chinese companies' rapid follow-up capabilities in the copper isotope field.
The development experience of Nuclidium also provides important references for China.Innovative Ambient Temperature Nuclide Generation TechnologyEffectively reducing the reliance on large equipment provides new ideas for exploring regionalized production networks in China.Modular Isotope-Ligand Combination Strategy, which has broken the traditional development limitation of "one drug, one nuclide." More notably, its industry-university-research collaborative model, through in-depth cooperation with top medical institutions, has significantly shortened the translation path from research and development to clinical application.
Currently, China's radiopharmaceuticals industry is at a critical juncture for leapfrog development. If policy support, clinical resources, and industrial foundations can be fully integrated to accelerate the construction of a localized copper isotope technology platform, China is highly expected to take the lead in the global theranostics field. With continuous breakthroughs in domestically produced accelerator technology and an increasingly enriched ligand library, a "copper era" for China's nuclear medicine is rapidly approaching.
