Small nucleic acids' global race heats up with China in the spotlight

SANEGENEBIO
Small Nucleic Acid Drug Developer

Rona Therapeutics
Nucleic Acid Drug Developer
At the beginning of 2026, the small nucleic acid therapeutics sector has experienced a full-scale surge.
Breakthroughs in technology and commercialization progress have jointly propelled the field of small nucleic acid therapeutics into a phase of commercialization scaling from 1 to 10. The successive release of clinical data for small nucleic acid drugs targeting weight loss has boosted market sentiment. Additionally, China National Biotec Group's blockbuster multi-billion acquisition and the Hong Kong listing of small nucleic acid company Ribo Life Science signal that capital exit channels have been opened. An innovative drug investor told VCBeat that the small nucleic acid therapeutics sector has entered a phase of accelerated commercialization.
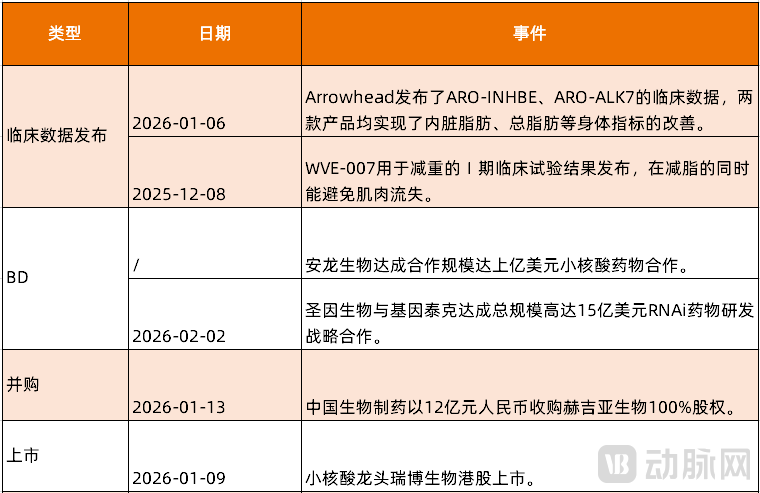
Figure 1. Recent Major Events in the Small Nucleic Acid Field (Source: VCBeat)
With leading Chinese innovative drug companies and multinational corporations (MNCs) continuing to expand their presence in the small nucleic acid therapeutics sector, this field has now entered the core track of innovative drug R&D. In the future, major developments in this area are expected to emerge successively.
The small nucleic acid therapeutics sector, which has gained significant momentum, experienced a full-scale surge in 2026, with major developments emerging continuously.
On January 6, Arrowhead Pharmaceuticals disclosed clinical data for two siRNA obesity candidate drugs. In obese patients with comorbid type 2 diabetes, both ARO-INHBE and ARO-ALK7 demonstrated improvements in body metrics such as visceral fat and total fat. Following the announcement, Arrowhead's stock price rose by 10.9%. However, market sentiment moderated due to the absence of the key metric "weight reduction" in the clinical outcomes. Nevertheless, the combination of ARO-INHBE with tirzepatide achieved a 9.4% reduction in body weight by week 16 in obese patients with type 2 diabetes, approximately doubling the effect compared to tirzepatide monotherapy (-4.8%). This data still highlights the potential application of small nucleic acid therapeutics for investors.
Just one month earlier, on December 8, 2025, Wave Life Sciences announced Phase I clinical trial results for its siRNA drug WVE-007 for weight reduction. At 12 weeks after a single dose, the drug achieved a 4.0% reduction in body weight and a 0.9% increase in muscle mass. WVE-007 demonstrated lipid-lowering effects comparable to semaglutide while mitigating the common risk of muscle loss associated with the latter. Moreover, WVE-007 was shown to be safe and well-tolerated, with no subjects discontinuing treatment and no severe or serious treatment-emergent adverse events (TEAEs) reported. On that day, Wave's stock price soared by 147.3%.
The successive release of clinical data for multiple promising candidates in the small nucleic acid weight loss sector indicates that small nucleic acid drugs have the potential to be used either as monotherapies or in combination with GLP-1 agonists to reduce fat while minimizing muscle loss. The clinical prospects for small nucleic acid therapeutics in the field of weight reduction remain highly promising.
In addition, the small nucleic acid therapeutics sector witnessed active licensing deals in 2025, with companies such as SanegeneBio, Argo Biopharma, and Sirius Therapeutics securing significant licensing-out agreements, a trend that continued into 2026. At the beginning of 2026, Anlong entered into an international strategic collaboration agreement with a Nasdaq-listed U.S. company in the field of small nucleic acid drugs, with a transaction valued at potentially over USD 100 million, marking a significant breakthrough for Anlong in international licensing partnerships.
Anlong has been steadily expanding its presence in the small nucleic acid field, and its extrahepatic targeted delivery technology platform has gained recognition from overseas companies, highlighting its commercial capabilities. Beyond licensing deals, innovative pharmaceutical companies are also strengthening their position in the small nucleic acid sector through mergers and acquisitions.
On the evening of January 13, 2026, Sino Biopharm announced that it would acquire 100% equity in Hygieia Pharmaceuticals for RMB 1.2 billion, further solidifying its presence in the siRNA therapeutics sector. Hygieia possesses unique advantages in long-acting drug delivery and extrahepatic delivery technologies. Hygieia's MVIP intrahepatic delivery platform is the world's first and currently the only clinically validated siRNA delivery system capable of achieving "once-yearly" dosing.
Extrahepatic delivery represents a key development trend for small nucleic acid drugs. Hygieia has already overcome technical barriers for delivery to multiple tissues, achieving precise targeting of organs such as the liver, nervous system, and adipose tissue. Through the acquisition of Hygieia, Sino Biopharm gains both advanced delivery platforms and a robust product pipeline.
The acquisition of Hygieia marks another significant move by Sino Biopharm in the small nucleic acid sector following its investment in SanegeneBio. It also represents the first major acquisition in the small nucleic acid field by a leading domestic innovative pharmaceutical company in China.
At the start of the new year, the succession of high-profile events clearly indicates that the small nucleic acid therapeutics sector has entered a period of rapid growth.
The current development trend of small nucleic acid drugs lies in expanding into extrahepatic indications. Previously approved small nucleic acid drugs have predominantly targeted liver-related conditions, where differences in delivery efficiency are minimal and competition is intense. As small nucleic acid drugs achieve breakthroughs in major indications, their application scope is broadening, and the commercial value of these products is being amplified.
From the current vantage point, VCBeat believes that weight reduction is the area most likely to witness significant licensing deals in the near future. This is not only due to the substantial market potential of weight loss drugs, which enables large-scale transactions, but more importantly, as clinical data from small nucleic acid candidates in the weight loss field continue to be reported, their clinical efficacy is gaining increasing recognition.
While GLP-1 receptor agonists, represented by semaglutide, are currently popular in the fields of glycemic control and weight loss, these products often face issues such as significant side effects, weight regain after discontinuation, and muscle loss due to appetite suppression. As an emerging therapeutic modality, small nucleic acid drugs hold the potential to achieve more precise and durable metabolic regulation.
Clinical data previously disclosed by Arrowhead and Wave Life Sciences indicate that small nucleic acid drugs have the potential to be used in combination with GLP-1 agonists to achieve synergistic effects and reduce muscle loss; or as maintenance therapy after discontinuation of GLP-1 drugs to suppress weight rebound; or even as monotherapy for weight loss and muscle gain in obese patients.
Currently, the most prominent targets in the small nucleic acid weight loss field are INHBE and ALK7. Both INHBE and ALK7 belong to the TGF-β superfamily and play key roles in the mechanisms of obesity and metabolic diseases: INHBE is primarily expressed in liver cells, where it encodes and secretes Activin E. The latter, along with other ligands, acts by binding to ALK7 to regulate lipid metabolism. ALK7 is mainly expressed in adipose tissue. When Activin E binds to ALK7, it exerts its effects by inhibiting the process of lipolysis. Chinese companies have active programs targeting both of these hot targets, INHBE and ALK7.
1) Rona Therapeutics' INHBE-targeting drug RN3161 ranks second globally in clinical progress
Regarding the INHBE target, which leads in R&D progress, China has a rich pipeline of candidates. The most advanced in clinical development are Rona Therapeutics' RN3161, Beisike's BC-006, and SanegeneBio's SGB-7342.
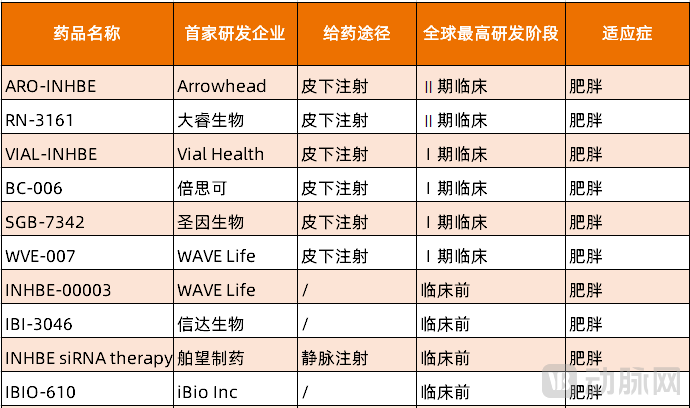
Figure 2. Global INHBE Target Small Nucleic Acid R&D Pipeline (Source: PharnexCloud)
RN3161 is developed based on Rona Therapeutics' proprietary GAIA™ platform and ranks second globally in clinical progress. Its optimized GalNAc delivery design combined with chemical modifications reduces off-target risks. Early data indicate that RN3161 exhibits favorable therapeutic effects. In primate models, a single low dose of RN3161 achieved over 90% downregulation of INHBE mRNA expression, demonstrating leading efficacy and durability. Additionally, RN3161 enables long-lasting effects. Preclinical studies show that RN3161 can achieve gene silencing for over six months, with the potential to reduce dosing frequency to just 1–2 times per year, significantly improving patient compliance and long-term management value.
Based on the GAIA™ platform, Rona Therapeutics has developed four products that have entered clinical stages: RN0361 for hypertriglyceridemia, RN0191 for hypercholesterolemia, RN5681 for atherosclerosis, and RN3161 for obesity.
At the 2025 American Heart Association Annual Meeting, Rona Therapeutics presented Phase I clinical data for RN0361. RN0361 exhibits two key characteristics: 1) Long-lasting effect: RN0361 achieves significant and sustained reduction in ApoC3 and triglyceride levels, with efficacy lasting at least six months. 2) Pronounced clinical outcomes: RN0361 can be used to prevent pancreatitis associated with severe hypertriglyceridemia and reduce cardiovascular disease risk caused by atherosclerotic lipoproteins, demonstrating potential best-in-class (BIC) characteristics.
The favorable clinical data for RN0361 further validate the superiority of Rona Therapeutics' proprietary GAIA™ platform. As the fourth clinical candidate molecule developed on this technological platform, RN3161's potential in treating obesity is further corroborated.
SanegeneBio's SGB-7342 employs next-generation GalNAc conjugation technology and utilizes RNAi to specifically silence the expression of the INHBE gene, thereby reducing the levels of its encoded protein, Activin E. This effectively inhibits the activity of downstream signaling pathways related to lipid metabolism and energy expenditure. Preclinical studies show that SGB-7342 potently and durably silences INHBE expression, significantly reducing body weight and fat mass while preserving muscle mass, and demonstrates favorable safety and tolerability.
The first subject for SGB-7342 was dosed on January 13, 2026, at the First Hospital of Jilin University, with preliminary data expected to be released in 2026.
2) SanegeneBio's SGB-ALK7 shows promising early clinical data
For the ALK7 target, domestic pipelines in China are still in early stages, with SanegeneBio's SGB-ALK7 being particularly noteworthy.
SGB-ALK7 is a drug developed by SanegeneBio based on its proprietary LEAD platform. This product exhibits common characteristics of small nucleic acid drugs targeting obesity: 1) Long-lasting effect: SGB-ALK7 has the potential to achieve ultra-long-acting treatment with subcutaneous injections every six months. 2) Favorable therapeutic outcomes: In non-human primates, a single subcutaneous dose was shown to achieve mRNA inhibition. In DIO mouse models, SGB-ALK7 demonstrated weight reduction effects comparable to semaglutide, reducing fat mass and promoting weight loss while preserving lean body mass.
Currently, this product is still in the preclinical research stage. As more clinical data become available, the therapeutic efficacy of SGB-ALK7 will be further validated.
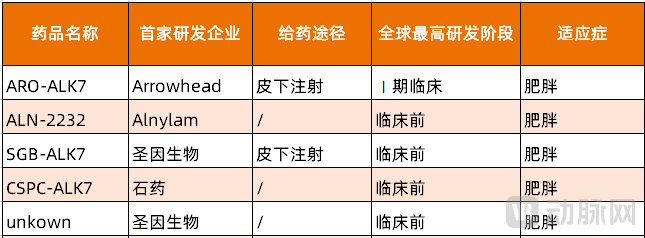
Figure 3. Global ALK7 Target Small Nucleic Acid R&D Pipeline (Source: PharnexCloud)
Beyond the aforementioned products, numerous early-stage small nucleic acid pipelines are advancing rapidly. According to VCBeat, Sunshine Nohe, a publicly listed company that began as a CRO, is transitioning toward innovative drug development, with small nucleic acid therapeutics being one of its three core strategic directions. Professor He Junlin, who has nearly three decades of experience in small nucleic acid R&D, has joined Sunshine Nohe. Under his leadership, the core team dedicated to small nucleic acid research and development has rapidly expanded from around 10 members in 2024 to 30–40 by the end of 2025, with the number of pipelines in development also growing steadily. Unlike most small nucleic acid biotech companies, Sunshine Nohe possesses end-to-end expertise spanning molecular discovery, drug screening, clinical trials, and regulatory approval. Its extensive CRO project experience can significantly accelerate the advancement of small nucleic acid programs. It is reported that Sunshine Nohe is actively promoting the development of small nucleic acid products for fat reduction and muscle preservation, with early clinical data expected to be released by the end of this year.
From an enterprise perspective, VCBeat holds an optimistic outlook on SanegeneBio's potential in the weight loss field. As a leader in next-generation RNAi technology, SanegeneBio focuses on developing "Best-in-Class" RNAi therapeutics for obesity, cardiovascular metabolism, and autoimmune diseases. Currently, SanegeneBio has 17 pipelines in development, seven of which target obesity. This rich pipeline portfolio not only provides a solid foundation for SanegeneBio's growth but also offers greater potential for licensing deals.

Figure 4. SanegeneBio's R&D Pipeline for Obesity Indications (Source: SanegeneBio Official Website)
Additionally, SanegeneBio has secured multiple licensing deals. On February 2, 2026, SanegeneBio entered into a strategic collaboration with Genentech, a member of the Roche Group, to jointly advance the development of an RNAi therapeutic, with a deal value of $200 million upfront and a total potential consideration of up to $1.5 billion. On November 8, 2025, SanegeneBio and Eli Lilly established a $1.2 billion global partnership for RNAi drugs targeting metabolic diseases. Earlier, SanegeneBio licensed its siRNA drug IBI3016 to Innovent Biologics for the treatment of hypertension.
The continuous out-licensing deals secured by SanegeneBio not only demonstrate the development potential of its proprietary LEAD technology platform but also highlight its commercialization capabilities and the recognition it has gained from leading innovative pharmaceutical companies. RNAi drugs developed based on this platform continue to hold significant promise for the future.
Currently, leading pharmaceutical companies are actively engaging in the small nucleic acid therapeutics sector. Sanofi has collaborated with Visirna, a subsidiary of Arrowhead Pharmaceuticals, in the cardiovascular field; CRISPR Therapeutics has partnered with Sirius Therapeutics to develop siRNA products for cardiovascular diseases; and Novartis has been particularly proactive, forming alliances with Regulus Therapeutics, Arrowhead Pharmaceuticals, Argo Biopharma, and Avidity Biosciences to expand indications and improve delivery systems. Eli Lilly's collaboration with SanegeneBio represents just one example of MNCs' strategic moves in the small nucleic acid space.
Specifically in the weight loss field, the ability of small nucleic acid drugs to enhance synergistic effects and reduce muscle loss when combined with GLP-1 agonists has gained industry recognition. Therefore, VCBeat believes that innovative pharmaceutical companies with blockbuster GLP-1 products are likely to supplement their pipelines with small nucleic acid therapies to achieve product synergy.
Top innovative pharmaceutical companies that have successfully commercialized GLP-1 products have all invested in small nucleic acid programs: Novo Nordisk has entered a collaboration with Replicate Bioscience on a self-replicating RNA (srRNA) platform to jointly develop novel candidates for metabolic diseases such as obesity and type 2 diabetes; Eli Lilly has established a $1.2 billion global RNAi partnership with SanegeneBio targeting metabolic diseases; and Innovent Biologics is rapidly advancing its small nucleic acid pipeline through collaborations with SanegeneBio combined with in-house R&D.
In the increasingly competitive GLP-1 weight loss space, MNCs are actively developing oral small molecule drugs: AstraZeneca has partnered with Eccogene on the small molecule GLP-1 agonist ECC5004 and, more recently, with CSPC Pharmaceutical Group on a GLP-1/GIP weight loss candidate; Roche acquired Carmot Therapeutics to gain access to the oral small molecule GLP-1 drug CT-996; MSD licensed the oral small molecule GLP-1 drug HS-10535 from Hansoh Pharma; and Pfizer recently re-entered the race for oral small molecule GLP-1 therapies by licensing YP05002 from YaoPharma, a subsidiary of Fosun Pharma, in a deal worth up to $2.085 billion, following three previous setbacks.
Among these companies, MSD and Pfizer have adopted a wait-and-see approach in the small nucleic acid field over the past two years and could emerge as potential buyers in the future. Although AstraZeneca already has a presence in small nucleic acids, it may further invest in small nucleic acid weight loss drugs to create combination therapies with its GLP-1 pipeline for enhanced efficacy. The future moves of these MNCs in this area merit close attention.
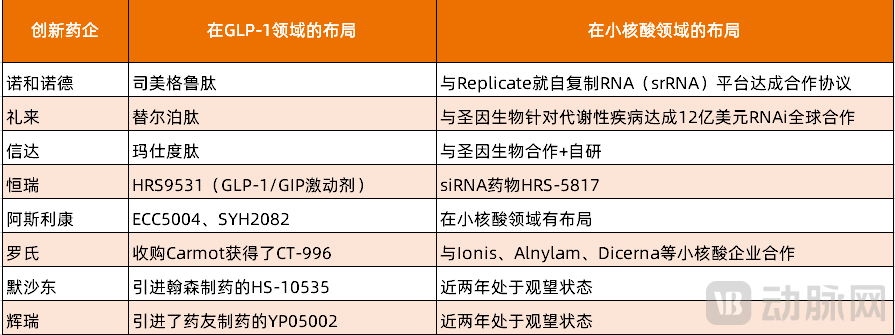
Figure 5. Layout of Leading GLP-1 R&D Companies in the Small Nucleic Acid Field
(Source: Incomplete collation by VCBeat)
The sustained momentum in the small nucleic acid therapeutics sector since the beginning of the year has clearly signaled that its commercialization is accelerating. Some commentators believe that 2026 marks the first year of commercialization for small nucleic acids. Currently, the biggest highlight in this field remains weight loss. Observing the strategic approaches of two leading Chinese innovative pharmaceutical companies, Hengrui and Innovent, in the weight loss space, we can see that the prevailing development strategy is combining small nucleic acid drugs with GLP-1 agonists to achieve better fat reduction and muscle preservation effects.
Guided by this approach, more collaborations are expected between Chinese small nucleic acid R&D companies and multinational corporations (MNCs), and significant future deals are highly anticipated.
