SanegeneBio inks potential up to $1.5B collaboration with Genentech, 2026's first major RNAi outbound deal in China
On February 2, SanegeneBio announced that it has entered into a global research collaboration and licensing agreement with Genentech (a member of the Roche Group). The collaboration will advance the development of an RNAi therapeutic based on SanegeneBio's proprietary RNAi drug discovery platform. This proprietary platform encompasses novel chemical modifications and delivery technologies, which can be leveraged to develop potential breakthrough siRNA medicines across multiple disease areas.
Under the terms of the agreement, SanegeneBio will grant Genentech exclusive worldwide rights to develop and commercialize the therapeutic candidate. SanegeneBio will be responsible for early-stage research and development, while Genentech will assume responsibility for all subsequent clinical development and global commercialization activities. SanegeneBio will receive an upfront payment of USD 200 million and is eligible to receive development and sales milestone payments totaling up to USD 1.5 billion, as well as tiered royalties on future product sales.
With Proprietary Delivery Technology and a Pipeline of 17 Drug Candidates Under Development
A breakthrough in delivery technology is a prerequisite for the success of small nucleic acid drugs. The focus of this collaboration lies in the announcement by SanegeneBio and Genentech that they will jointly advance the development of an RNAi therapeutic based on SanegeneBio's proprietary RNAi drug discovery platform. Currently, SanegeneBio possesses a self-developed GalNAc liver-targeted delivery platform and a proprietary LEAD™ (Ligand and Enhancer Assisted Delivery) extrahepatic delivery platform.
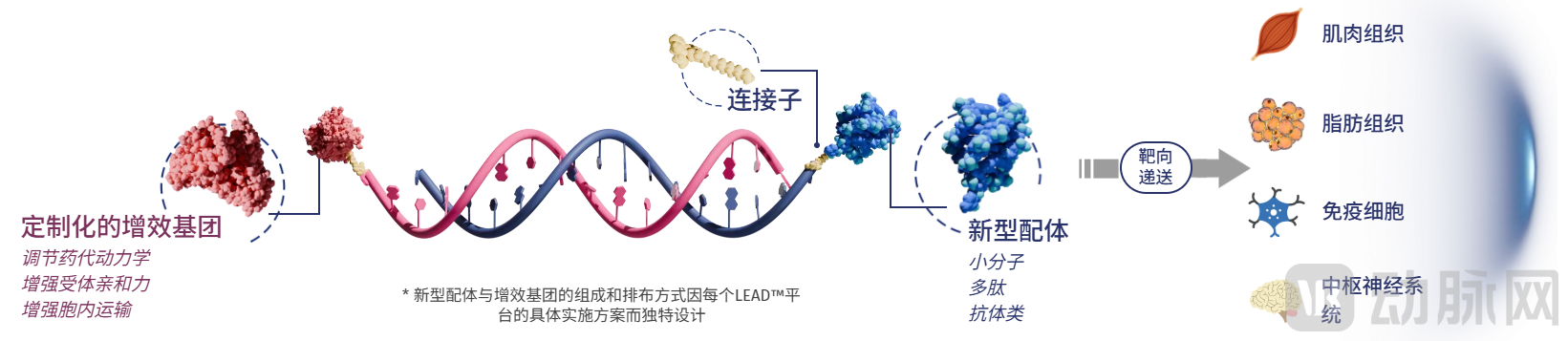
LEAD™ Extrahepatic Delivery Platform, Image Source: SanegeneBio Official Website
Among these, the LEAD™ extrahepatic delivery platform is a tissue-selective delivery technology independently developed by SanegeneBio. It enables the efficient and specific delivery of siRNA to extrahepatic tissues such as adipose, muscle, immune cells, and the central nervous system, achieving potent, safe, and durable silencing of disease-causing genes while significantly reducing dosing frequency. Based on this platform, breakthrough therapies for metabolic diseases requiring only two subcutaneous administrations per year are anticipated.
If liver-targeted delivery represents the current mainstream of nucleic acid drug delivery, then extrahepatic delivery is the future trend for small nucleic acid therapeutics. SanegeneBio's forward-looking development of the LEAD™ extrahepatic delivery platform is attributed to the extensive experience of its leadership.
Dr. Weimin Wang, Founder and Chief Executive Officer of SanegeneBio, is a pioneer in oligonucleotide medicinal chemistry with nearly three decades of research and development experience in the siRNA pharmaceutical industry. He previously held scientific leadership roles at Sirna Therapeutics (one of the earliest RNAi companies, acquired by Merck for USD 1.1 billion in 2006), Merck, and Dicerna Pharmaceuticals (acquired by Novo Nordisk for USD 3.3 billion in 2022).
Dr. Wang is a core inventor of innovative RNAi delivery technologies such as GalXC™ and GalXC-Plus™ and has led the development of multiple major RNAi therapeutics, including Rivfloza® for primary hyperoxaluria, xalnesiran for chronic HBV infection, and lepodisiran for cardiovascular disease. He has evaluated over 500 external opportunities in nucleic acid drug discovery and delivery technologies and managed multiple collaborative partnerships with leading pharmaceutical companies such as Eli Lilly, Roche, Novo Nordisk, Boehringer Ingelheim, and Alexion.
Leveraging its core team and proprietary technologies, SanegeneBio has established a pipeline of 17 programs, spanning broad areas of high unmet need including autoimmune diseases, cardiovascular diseases, metabolic disorders/obesity, and coagulation disorders. The pipeline comprises four clinical-stage candidates (one in Phase II and three in Phase I) along with multiple preclinical projects.

Drug Candidates in Development, Source: SanegeneBio Official Website
$2.81B Inflow: SanegeneBio's Financial Momentum in Three Months
In fact, this is not only the first outbound small nucleic acid collaboration from China since the beginning of 2026, but also the second partnership SanegeneBio has established with a multinational corporation within three months.
On November 8, 2025, SanegeneBio announced a collaboration with Eli Lilly to jointly advance the development of RNAi candidates targeting metabolic diseases based on SanegeneBio's proprietary LEAD™ platform. SanegeneBio will be responsible for screening and identifying optimal RNAi molecules for the collaboration using the LEAD™ platform, while Eli Lilly will oversee subsequent IND-enabling studies, clinical development, and commercialization. Under the agreement, SanegeneBio will receive near-term payments, including an upfront payment and an equity investment. Additionally, SanegeneBio is eligible for up to USD 1.2 billion in milestone payments as well as tiered royalties on commercial sales.
One month later, on December 8, 2025, SanegeneBio announced the completion of a Series B financing round exceeding USD 110 million. Investors included a leading industrial institution, an international sovereign wealth fund, Sino Biopharm, Legend Capital, Vivo Capital, Invus, SymBiosis, Suzhou Capital Group, TruMed Investment, and Lake Bleu Capital. The round also secured strategic investment from Eli Lilly. Existing shareholders, including GL Ventures, Qiming Venture Partners, K2 Bio Partners, TF Capital, Oriza Holdings, and Northern Light, continued their support.
Due to the inherently shorter development cycles and higher success rates associated with small nucleic acid drugs, this means that once the delivery platform is refined and with the financial and resource support from multinational corporations and investors, SanegeneBio's subsequent research, development, and translation efficiency will be significantly enhanced, accelerating its progress into clinical validation and commercialization stages.
Including SanegeneBio, the broader Chinese small nucleic acid sector has been highly active in the first month of 2026. On January 6, Sanofi's siRNA drug plozasiran (brand name: Redemplo®) was approved by the NMPA for use in combination with dietary control to lower triglyceride levels in adult patients with familial chylomicronemia syndrome. On January 9, Ribo Life Science, a small nucleic acid company backed by Legend Capital and Legend Star, officially listed on the Hong Kong Stock Exchange, becoming the "first small nucleic acid drug stock of 2026." On January 13, Sino Biopharm announced it would fully acquire the Chinese siRNA innovator Hygieia Pharmaceuticals for a total consideration of RMB 1.2 billion.
Internationally, in January 2025, Alnylam, a leading RNAi therapy company, disclosed at the JPM 2026 conference that its revenue in 2025 approached USD 3 billion from four independently commercialized siRNA products, an 81% year-over-year increase, achieving non-GAAP profitability. Among these, its transthyretin amyloidosis (ATTR) TTR product series performed exceptionally well, with sales doubling to approximately USD 2.5 billion. The company even raised its 2026 revenue guidance for the TTR products significantly to USD 4.4–4.7 billion, with a median growth expectation of 83%. Currently, Alnylam's market capitalization stands at USD 44.8 billion, while Ionis and Arrowhead each exceed USD 10 billion in market value.
Undoubtedly, the small nucleic acid field has become one of the hottest segments in the biopharmaceutical industry today. Within this highly competitive landscape, companies with key technological platforms such as Ribo Life Science, Argo Biopharma, SanegeneBio, Sirius Therapeutics, and Hygieia Pharmaceuticals will lead Chinese pharmaceutical firms in pioneering innovation from the source and contribute first-in-class drugs globally in the "third wave of modern drug development."
