Beyond the deal: CSPC scores $1.2B upfront from AstraZeneca for monthly weight-loss peptide pact
On January 30, CSPC Pharma announced a strategic collaboration and licensing agreement with AstraZeneca, with a total potential value of up to USD 18.5 billion. The partnership will utilize CSPC's proprietary sustained-release drug delivery (long-acting delivery) technology platform and its AI-powered peptide discovery platform to develop innovative long-acting peptide drugs.
Regarding the pipeline licensing, AstraZeneca has secured the global exclusive rights (excluding China's Mainland, Hong Kong SAR, Macao SAR, and Taiwan) to CSPC's portfolio of once-monthly injectable weight management products, including an exclusive license for their development, production, and commercialization. The assets involved include one clinical-ready program, SYH2082 (a long-acting GLP-1R/GIPR agonist advancing into Phase I clinical trials), as well as three preclinical-stage programs with distinct mechanisms of action. These programs aim to provide more durable treatment benefits for individuals with obesity and weight-related concerns.
Regarding the strategic collaboration, the two parties will engage in a comprehensive partnership in the discovery of innovative peptide molecules and the development of long-acting delivery products. In addition to advancing the existing preclinical long-acting peptide pipeline, CSPC will also discover and develop other innovative long-acting peptide products for AstraZeneca. The collaboration will also involve four additional projects based on these platforms, bringing the total number of licensed pipeline assets to eight.
Under the agreement, for AstraZeneca's access to the eight projects and the use of these platforms, CSPC will receive an upfront payment of USD 1.2 billion. CSPC is also eligible to receive up to USD 3.5 billion in potential development milestone payments and up to USD 13.8 billion in potential sales milestone payments, as well as sales royalties on the annual net sales of the relevant licensed products that could reach up to double-digit percentages.
CSPC Pharma will retain the rights to the aforementioned licensed projects in China's Mainland, Hong Kong SAR, Macao SAR, and Taiwan.
1CSPC Megalith Biopharmaceutical and CSPC Zhongqi Pharmaceutical as the Core Transaction Parties
According to an announcement by CSPC Innovation Pharmaceutical, the controlling subsidiary CSPC Megalith Biopharmaceutical, the affiliated party CSPC Pharma, and CSPC Zhongqi Pharmaceutical are collectively acting as the licensors in this transaction. CSPC Megalith Biopharmaceutical will receive 35% of the total upfront payment.
Among the transaction's projects, the product involved in the collaboration and licensing between CSPC Megalith Biopharmaceutical and AstraZeneca primarily relates to an innovative peptide molecule along with related technologies and products, currently in the preclinical research stage. It aims to provide a longer-lasting treatment solution for individuals with obesity and weight-related concerns. Furthermore, there exists the possibility for CSPC Megalith Biopharmaceutical and AstraZeneca to engage in strategic collaboration on other related projects in the fields of innovative peptide molecule discovery and long-acting delivery product development.
As a company separately listed on the ChiNext market under CSPC Pharma, CSPC Innovation Pharmaceutical gained control of CSPC Megalith Biopharmaceutical in September 2023 through a capital increase of RMB 1.871 billion, completing an internal transfer within the "CSPC group." Following this acquisition, CSPC Innovation Pharmaceutical expanded its business from functional ingredients and health foods into the field of innovative biologic drugs.
CSPC Megalith Biopharmaceutical is an innovative biopharmaceutical enterprise focused on cutting-edge areas such as antibody-based drugs, antibody-drug conjugates (ADCs), and mRNA vaccines. Its therapeutic areas include breast cancer, cervical cancer, gastric cancer, and psoriasis, among others.
It is worth noting that in 2024, CSPC Innovation Pharmaceutical acquired 100% equity of CSPC Baike Bio-Pharmaceutical through share issuance and cash payment. Established in January 1994, CSPC Baike Bio-Pharmaceutical is dedicated to the R&D and commercialization of long-acting protein drugs. Its main products include Jinyouli® (PEGylated Recombinant Human Granulocyte Colony), launched in 2021, as well as the Semaglutide Injection for which a weight loss indication has been applied, and TG103 Injection (a GLP-1 Fc fusion protein), a potential "blockbuster" weekly weight loss therapy. Additionally, CSPC Baike Bio-Pharmaceutical has a preclinical pipeline including SYH9017 (a long-acting Semaglutide Injection in fluid crystal formulation), oral semaglutide tablets, and dual- and triple-target GLP-1 products.
The other party in the transaction, CSPC Zhongqi Pharmaceutical, also known as the CSPC Central Institute of Pharmaceutical Research, undertakes the mission of strategic product R&D. It is reported that the institute has initially established a proprietary technology system in research areas such as computer-aided drug design, liposome targeted formulations, oral sustained- and controlled-release formulations, novel pharmaceutical excipients, protein modification, and ADC technology. It is the only enterprise in China holding national-level qualifications in all seven of these R&D domains. Its main research areas include the development of innovative drugs for cardiovascular and cerebrovascular diseases, oncology, endocrine, neurological and psychiatric disorders, as well as high-end antibiotics.
2Weight Loss: Core Compliance Challenges Remain, Focusing on Long-term Medication Management
The only publicly disclosed asset this time, the SYH2082 injection, has been accepted for review by the Center for Drug Evaluation (CDE), NMPA on January 6, 2026, according to CDE data. The applicant is Beijing Kangchuanglian Biopharmaceutical Technology Research Co., Ltd, with the acceptance number CXHL2600019. Public information on SYH2082 is limited, but insights into its long-acting delivery technology platform can be gleaned from other products in CSPC's portfolio.
Specifically highlighted as developed from CSPC's long-acting delivery technology platform, the once-monthly semaglutide long-acting injection SYH9017 is based on excipients with good biocompatibility. Upon subcutaneous injection, it forms a gel depot, enabling sustained drug delivery.
Another product, the weekly formulation TG103, is a recombinant human GLP-1 Fc fusion protein developed based on the hyFc long-acting fusion protein platform technology. Its molecular design extends the in vivo half-life, allowing for once-weekly or once-every-two-weeks subcutaneous administration.
Industry analysis indicates that CSPC's long-acting peptide formulation platform utilizes an in-situ gel technology. Their proprietary intellectual property, termed LiquidGel, is a depot-type long-acting injection delivery system where the loaded drug forms a gel in situ after injection. The principle involves dissolving or dispersing the peptide drug together with biodegradable polymers in a biocompatible lipophilic solvent. After injection via a fine needle into subcutaneous or muscle tissue, upon contact with tissue fluid, the lipid solution rapidly diffuses, transforming into a nanostructured gel, forming a drug-loaded depot in situ. This achieves slow and relatively stable drug release over a predetermined period, providing a sustained-release effect.
Furthermore, among the assets this time, there are three R&D projects in the preclinical stage with different mechanisms of action. This demonstrates CSPC's quite rich and diversified pipeline in weight loss and muscle building. Weight loss targets include GLP-1, GLP-1/GIP, GLP-1/GIP/GCG, Amylin, ALK7, INHBE, OX7, among others, while muscle-building targets include ActRII, GDF8, etc. Modality types encompass peptides, long-acting peptides, antibodies, and small nucleic acids.
In July 2025, CSPC entered into a global licensing agreement with Madrigal Pharmaceuticals for its self-developed novel oral small molecule GLP-1 receptor agonist SYH2086. The total potential deal value could reach up to $2.075 billion, setting a new record for out-licensing of early-stage innovative drug R&D achievements from China. This will also accelerate CSPC's global expansion in metabolic diseases. Through combination therapy development with Madrigal's Rezdiffra, they aim to explore the synergistic effects of GLP-1 in treating metabolic dysfunction-associated steatohepatitis (MASH).
Trend-wise, the weight loss indication remains underexplored, particularly for disease-oriented (e.g., MASH, cardiovascular diseases) and overall metabolic benefit-oriented (e.g., cardiovascular benefits, renal benefits) approaches. Notably, weight loss drugs possess a dual attribute: they serve as "lifestyle drugs" meeting socio-psychological needs and improving body image, while also being important long-term therapeutic agents for chronic diseases.
Current GLP-1-based drugs face challenges such as poor maintenance of efficacy, adverse reactions, poor patient adherence, lack of long-term safety and efficacy data, and insufficient data in elderly populations. From a clinical implementation perspective, "long-term adherence" represents a current urgent and prioritized need—the average patient currently uses these medications for only 2-3 months. However, obesity, as a chronic disease, requires continuous medication to maintain efficacy, creating a fundamental conflict with patients' expectation of "no weight regain after stopping medication."
Therefore, both the combination therapy development of the oral small molecule GLP-1 with Madrigal and the long-acting peptide pipeline and underlying technology platform showcased this time reflect the trend towards addressing long-term adherence and differentiated medication regimens within the context of "long-term medication management."
Looking further, the weight loss market is shifting from traditional pharmaceutical logic to a new consumer goods logic. Traditional GLP-1-based glucose-lowering drugs followed a physician-led decision-making model with passive patient acceptance. In contrast, GLP-1-based weight loss drugs have achieved unexpected market education in the public discourse, directly reaching consumers through e-commerce platforms. This enabled Eli Lilly to surpass Novo Nordisk within two years, becoming the world's first pharmaceutical company with a trillion-dollar market capitalization. Consequently, the weight loss drug market now demands products that can satisfy diverse needs, various therapeutic mechanisms and administration regimens, multiple specific sub-indication populations, and potentially multi-tiered pricing.
3AI+: First Bet on Peptide Macromolecular Design
In 2023, CSPC successively established partnerships with XtalPi, Insilico Medicine, and Huawei Cloud to build an AI-driven R&D platform targeting high-demand areas such as cancer and neurological diseases, covering key stages including drug design, target discovery, and molecular screening.
Moreover, CSPC and AstraZeneca are already long-standing partners in AI R&D platform collaborations, with this being the first implementation of a macromolecule (peptide) AI platform partnership between them:
In October 2024, CSPC and AstraZeneca entered into a global collaboration for the Lp(a) inhibitor YS2302018. This compound was discovered through an AI-driven small molecule drug design platform, with a total deal value of $2 billion (including a $100 million upfront payment).
In June 2025, the two parties signed another strategic R&D collaboration agreement worth up to $5.33 billion (including a $110 million upfront payment). AstraZeneca will leverage CSPC's AI drug discovery platform to identify and develop novel oral small molecule drug candidates.
The focus on "AI + Macromolecules" also aligns with the internal strategy of AstraZeneca, an "AI enthusiast." A previous DPI report showed that AstraZeneca leads all multinational corporations (MNCs) in AI-driven drug discovery partnerships, with 25 collaborations, the earliest of which dates back to 2017.
According to its official website, AstraZeneca currently uses AI to help infer the most suitable molecules in the shortest possible time, covering 70% of its small molecule chemistry projects. Over 90% of its small molecule discovery processes rely on AI assistance. AstraZeneca is also rapidly integrating artificial intelligence and machine learning methods into the discovery and design of next-generation therapeutics, such as biologics (peptide or protein therapies, nucleotide therapies, and cell therapies), to increase the likelihood of clinical success.
4"Sale of Green Seedlings" Still a Cause for Concern
On the same day the agreement was signed (January 29), AstraZeneca announced plans to invest $15 billion in China by 2030 to expand pharmaceutical manufacturing and research and development. This investment will fully leverage China's outstanding scientific capabilities, advanced manufacturing capacity, and the collaborative advantages of the healthcare ecosystems between China and the UK, providing cutting-edge treatment options for patients both in China and globally.
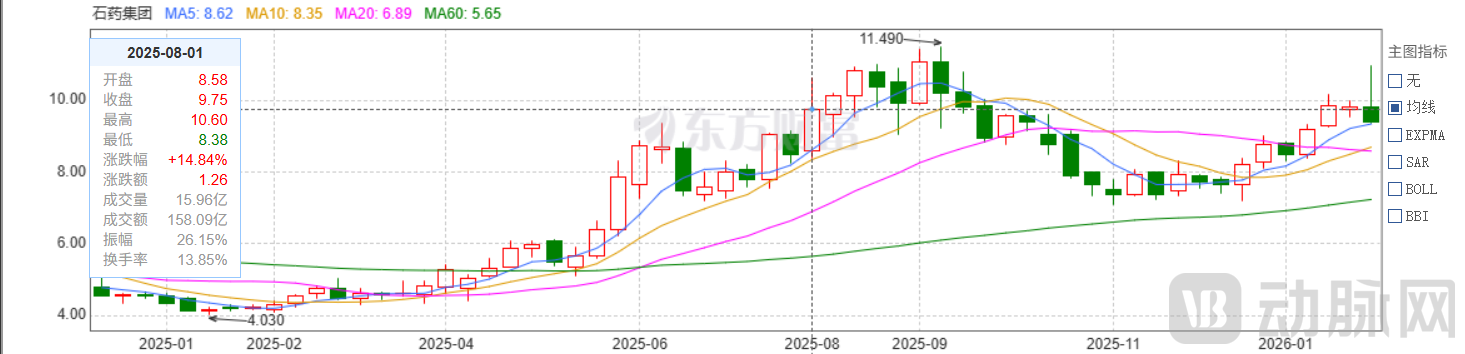
Following the market opening, the stock prices of CSPC and CSPC Innovation Pharmaceutical declined. However, CSPC's stock surged at the opening to reach its highest level in 2026 at 10.940.
In contrast, the single-asset deal with Madrigal in July 2025, which did not retain Chinese rights for SYH2086 and only preserved rights to develop and commercialize other oral small-molecule GLP-1 receptor agonist products in China, still triggered a rally in CSPC's stock. According to Haitong International's pharmaceutical analyst, the transaction with Madrigal exceeded market expectations, reflecting the value of CSPC's small-molecule platform and opening up new possibilities in the weight loss, diabetes, and MASH markets.
In the current collaboration, CSPC not only secured an impressive deal with a $1.2 billion upfront payment and a total potential value of $18.5 billion but also retained rights to the licensed assets in China's Mainland, the Hong Kong Special Administrative Region, the Macao Special Administrative Region, and the Taiwan region.
As for the out-licensing of macromolecule platforms, does the formula of "licensing deals + stock rally" no longer apply? Perhaps a longer observation period is needed.
