Simcere lands €1B+ deal with Boehringer for potentially first-in-class IBD bispecific antibody

Simcere
Innovative Drug Developer

Boehringer Ingelheim
Developer of Innovative Drugs and Therapies
On January 27, Simcere Pharmaceutical Group Ltd. (“Simcere”) (HKEX: 2096) and Boehringer Ingelheim, announced a license and collaboration agreement to develop SIM0709, a pre-clinical TL1A/IL23p19 bispecific antibody from Simcere, for the treatment of inflammatory bowel disease (IBD).
Under the terms of the agreement, Boehringer Ingelheim receives global rights to the asset, excluding Greater China. Simcere is eligible to receive an upfront payment, as well as success-based development, regulatory, and commercial milestone payments totaling up to EUR 1.058 billion. Additionally, Simcere will receive tiered royalties on net sales generated outside the Greater China territory.
This collaboration marks Simcere's second out-licensing transaction in the autoimmune disease field and its fifth out-licensing deal for internally discovered innovative drug candidates. The total potential transaction value of these out-licensing agreements has now exceeded USD 4.6 billion.
1Bispecific Antibodies Targeting Two Major IBD Targets
SIM0709 is a novel long-acting humanized bispecific antibody developed from Simcere's proprietary multi-specific antibody platform. It simultaneously targets Tumor Necrosis Factor-like Ligand 1A (TL1A) and Interleukin-23 (IL-23), blocking two core pathways that drive the onset and progression of Inflammatory Bowel Disease (IBD). This positions SIM0709 with first-in-class potential in the treatment of IBD.
Poster presentations at the 2025 American Association of Immunologists (AAI) Annual Meeting revealed that SIM0709 demonstrated excellent synergistic efficacy in both in vitro primary cell studies and in vivo animal models. In multiple in vitro and in vivo IBD animal studies, it exhibited superior functional and efficacy outcomes compared to other global investigational anti-TL1A or anti-IL-23 monoclonal antibodies, with its synergistic effect significantly surpassing even the combination of the two corresponding monoclonal antibodies.
This molecule possesses favorable drug-like properties, is suitable for subcutaneous administration, and exhibited an extended half-life in cynomolgus monkey pharmacokinetic studies. These attributes hold promise for reducing dosing frequency and improving clinical compliance. SIM0709 is anticipated to enter initiate first-in-human studies in the first half of 2026.
Globally, over three million people are affected by IBD. This lifelong, progressive condition often leads to frequent hospitalizations, surgeries, and significantly impacts patients' quality of life. Current medical interventions cannot fully prevent or reverse the disease and its complications, highlighting a substantial unmet clinical need in this field.
Both targets involved in this transaction are highly validated, with approved drugs already on the market for autoimmune diseases.
IL-23 p40 is a common subunit shared by IL-12 and IL-23. p40 antibodies block both the IL-12 and IL-23 immune pathways and represent a classic immunotherapy approach. In recent years, IL-23 p19 antibodies have demonstrated superior efficacy and a better safety profile, showing promise in treating various autoimmune diseases. Beyond forming the classic inflammatory cytokine IL-23 with p40, p19 also combines with EBI3 to form IL-39, which activates IL-23R and gp130—an important inflammatory pathway mediated by vascular endothelial cells, among others. Both IL-23 and IL-39 can activate the intracellular JAK-STAT signaling pathway, stimulating the secretion of IL-17A/F.
Several monoclonal antibodies targeting IL-23 p19 are already approved: Eli Lilly's Omvoh, AbbVie's risankizumab (approved in China), and Johnson & Johnson's guselkumab (approved in China) have been approved for treating Ulcerative Colitis and Crohn's Disease within the IBD field. China Medical System's tildrakizumab injection and Innovent Biologics' picankibart are also approved for treating moderate-to-severe plaque psoriasis.
TL1A acts as an amplifier for multiple pathways and possesses dual anti-inflammatory and anti-fibrotic effects, making it closely associated with inflammatory diseases like IBD. Globally, no TL1A-targeting drug has yet been approved, but the target is highly regarded. J.P. Morgan's 2025 Healthcare Conference noted that MSD views autoimmune therapies based on the TL1A target as having multi-billion-dollar potential. MSD's TL1A monoclonal antibody, tulisokibart, is in Phase III clinical trials and originated from a USD 10.8 billion acquisition (Prometheus Biosciences) in 2023. Roche's TL1A monoclonal antibody RG6631 and Sanofi/Teva's TL1A monoclonal antibody duvakitug also resulted from licensing deals in 2023, with total potential values exceeding USD 7 billion and USD 1.5 billion, respectively. Notably, all three TL1A monoclonal antibodies acquired by these multinational corporations at significant cost are being developed for IBD.
Furthermore, in June 2024, AbbVie entered into a licensing deal with the Beijing-based biotech company FutureGen for its internally discovered preclinical TL1A monoclonal antibody FG-M701, involving an upfront payment of USD 150 million and potential milestone payments of up to USD 1.56 billion.
Regarding bispecific antibodies, Pfizer and Roche are collaborating globally on the development of PF-07261271 (an anti-p40/TL1A bispecific antibody) as part of a global collaboration linked to the aforementioned USD 7 billion acquisition. Public information from 2025 indicates it is currently in Phase I clinical trials.
2Simcere's First Bispecific Antibody Out-Licensed, Second Autoimmune Drug for Global Partnership
Since its listing on the Hong Kong Stock Exchange in 2020, Simcere has consistently intensified its focus on the innovative drug sector. Over the past five years, the company has invested approximately RMB 9 billion in research and development. The proportion of revenue contributed by innovative drugs has increased significantly, rising from 45% in 2020 to 77% in the first half of 2025.
By therapeutic area, oncology represents the core of its transformation from generic to innovative drugs. Its dedicated oncology innovation arm, Simcere Zaiming, already has five commercialized products, generating annual revenue exceeding RMB 1 billion. Four of these products — Endostar (Recombinant Human Endostatin Injection, for lung cancer), COSELA (Trilaciclib Hydrochloride for Injection, for lung cancer), Enlituo (Cetuximab Beta Injection, for colorectal cancer), and ENZESHU (Suvemcitug for Injection, for ovarian cancer) — have been included in China's National Reimbursement Drug List. Additionally, ENWEIDA (Envafolimab Injection) has been approved for the treatment of gastrointestinal cancers and has received Orphan Drug Designation from the U.S. FDA for advanced biliary tract cancer and soft tissue sarcoma.
The latest prospectus indicates that innovative oncology drugs accounted for over 90% of the oncology segment's revenue during the reporting period, establishing them as the core growth driver for the company's oncology business. For the periods covering 2023, 2024, and the first three quarters of 2025, revenue from the aforementioned innovative drugs amounted to RMB 1.426 billion, RMB 1.185 billion, and RMB 1.036 billion, respectively, constituting 93.7%, 91.5%, and 83.7% of the total revenue for the corresponding periods.
Simultaneously, 2025 has marked a period of significant overseas licensing breakthroughs for its oncology portfolio:
In December 2025, Simcere Zaiming entered into an exclusive licensing agreement with Ipsen to jointly develop SIM0613, a novel ADC drug targeting LRRC15. Ipsen obtained global rights to the drug outside Greater China. The total potential transaction value could reach up to USD 1.060 billion.
In June 2025, Simcere Zaiming entered into a licensing and strategic collaboration agreement with NextCure to jointly develop SIM0505, a novel ADC drug targeting CDH6 for the treatment of solid tumors. The total potential payments could amount to up to USD 745 million.
In January 2025, Simcere Zaiming entered into an option license agreement with AbbVie for SIM0500, a clinical-stage candidate drug for multiple myeloma—a trispecific antibody targeting GPRC5D/BCMA/CD3. The total potential payments could reach up to USD 1.055 billion.
This month, Simcere Zaiming formally submitted its prospectus to the Hong Kong Stock Exchange, applying for an independent listing on the main board. Concurrently, Zhou Yunshu, former chairman of Hengrui Pharmaceuticals, made his public appearance in the role of Group President of Simcere Pharmaceutical Group.
The completion of this out-licensing deal for a bispecific antibody in the autoimmune field can be seen as Simcere's proactive response to market concerns that "the independent financing of its core oncology assets may impact Simcere's profitability and long-term development."
Looking back, Simcere has been strategically building its presence in autoimmune diseases for many years, with marketed products including innovative drugs such as Aidexin (Iguratimod Tablets). As early as September 2022, Simcere out-licensed the global rights (ex-China) for SIM0278, its internally discovered, preclinical-stage Treg-preferential IL-2 mutant fusion protein (IL-2 mu Fc), to the Spanish company Almirall. Simcere is eligible to receive up to USD 492 million in milestone payments. Currently, SIM0278 has advanced to Phase II clinical trials globally, with its Phase II study in China investigating its use for the treatment of moderate-to-severe atopic dermatitis.
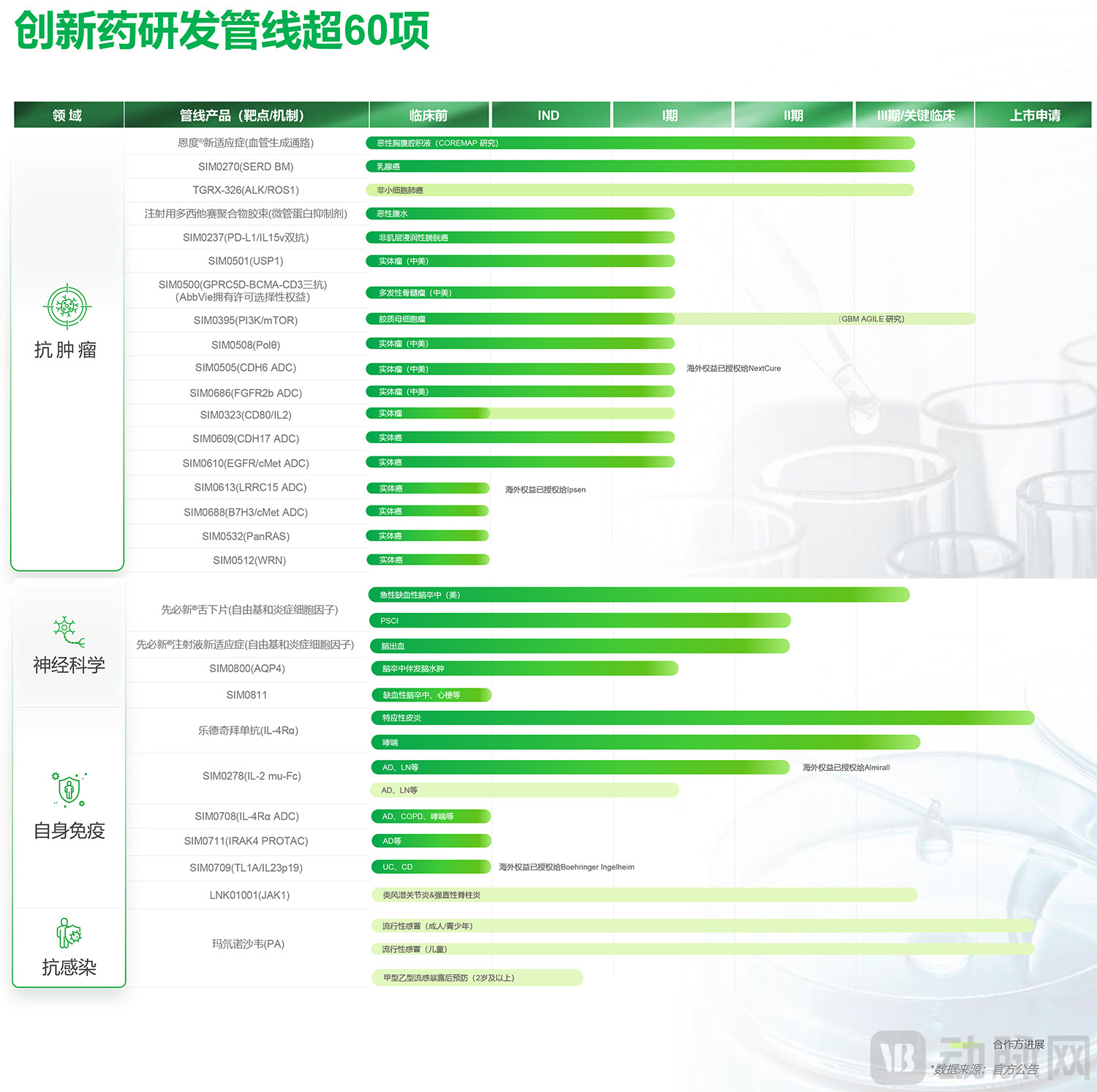 Simcere Pharmaceutical Innovative Drug Pipeline Layout
Simcere Pharmaceutical Innovative Drug Pipeline Layout
In the field of autoimmune diseases, Simcere has developed a diversified portfolio of assets. The marketing application for Rademikibart (an anti-IL-4Rα monoclonal antibody) was accepted in July 2025, with its indication presumed to be moderate-to-severe atopic dermatitis. Both the IL-4Rα-targeting ADC SIM0708 and the IRAK4-targeting PROTAC (oral proteolysis-targeting chimera) SIM0711 are in the pre-IND stage, with related preclinical studies conducted in mouse models of skin inflammation.
Furthermore, the novel B- and T-lymphocyte attenuator (BTLA) agonist monoclonal antibody SIM0710 significantly prolonged the overall survival of humanized graft-versus-host disease (GVHD) mouse models in vivo. It also markedly reduced immunologic markers such as serum anti-dsDNA IgG levels in humanized systemic lupus erythematosus (SLE) models, demonstrating potential as an innovative therapy for a range of autoimmune diseases.
3Autoimmunity, the Next Bispecifics High Ground for MNCs
This is not the first time Boehringer Ingelheim has actively pursued autoimmune bispecific antibodies.
In April 2025, Boehringer Ingelheim entered a collaboration totaling USD 357 million with Cue Biopharma to co-develop and commercialize its autoimmune bispecific antibody CUE-501, a differentiated B-cell depleting therapy. Notably, Boehringer highlighted that partnering with Cue Biopharma would not only bring a promising autoimmune bispecific asset but also strengthen its autoimmune R&D capabilities through access to Cue Biopharma's proprietary T-cell engager platform.
While bispecific drug development in oncology is relatively mature, the autoimmune field remains less explored. Autoimmune diseases are numerous, affect a large patient population, and present significant unmet clinical needs, continuously contributing new growth potential to the pharmaceutical industry. For multinational corporations (MNCs) facing patent cliffs and growth challenges, autoimmune disorders undoubtedly represent a fertile ground for strategic investment.
On the R&D front, rapid progress in autoimmune research has led to a continuous emergence of novel targets. Pharmaceutical giants such as Johnson & Johnson, Sanofi, and Roche are expanding and iterating their product portfolios by developing bispecific pipelines. In particular, bispecifics targeting well-validated targets like CD3, TSLP, and interleukin family members have become a focus of MNCs' novel drug development.
Among these, Sanofi, MSD, and Roche have chosen to advance CD3-based bispecifics. CD3 is closely involved in T-cell activation. By binding both CD3 and a tumor-associated antigen, CD3 bispecifics can redirect and activate T cells to approach and kill tumor cells. Consequently, most globally approved bispecific drugs are T-cell engagers (TCEs) based on the CD3 target, indicated for both hematologic malignancies and solid tumors. If successfully validated in autoimmune diseases, CD3 bispecifics could become blockbuster therapies spanning solid tumors, hematologic cancers, and autoimmune disorders.
Meanwhile, Sanofi and Pfizer are developing TSLP-targeting bispecifics. Thymic stromal lymphopoietin (TSLP) is a novel cytokine structurally similar to IL-7 and has been validated as relevant to several autoimmune diseases such as atopic dermatitis, psoriasis, and rheumatoid arthritis. In 2021, Tezepelumab, jointly developed by Amgen and AstraZeneca, became the first globally approved TSLP monoclonal antibody for the treatment of severe asthma. Its sales grew rapidly post-launch, reaching USD 170 million globally in 2022 and surpassing USD 1 billion for the first time in 2024 with sales of USD 1.219 billion. In the first half of 2025, its sales grew by 73%, contributing to an 11% increase in AstraZeneca's overall performance. Based on Waypoint results, Leerink projects that Tezspire could achieve sales of USD 4.3 billion by 2030.
Johnson & Johnson, which holds five leading autoimmune products, is building a differentiated and complementary portfolio of bispecific antibodies to address multiple pathogenic pathways. In 2024, the company acquired Proteologix for USD 850 million, gaining its core assets PX128 and PX130. The former is an IL-13/TSLP bispecific antibody for atopic dermatitis and asthma, currently in Phase I clinical studies; the latter is an IL-13/IL-22 bispecific antibody for atopic dermatitis, currently in preclinical development.
