Top 10 medical device growth leaders: product segments beating the industry average

Medtronic
Medical Device Manufacturer
In mid-2025, EY-Parthenon predicted that the global medical device market would grow at an annual rate of approximately 6%, indicating steady overall market expansion. The medical device industry is undergoing a shift in growth drivers, with multinational corporations (MNCs) intensively divesting or selling off underperforming or slower-growth businesses to focus on higher-growth segments. While certain areas of the industry are losing momentum, which emerging segments are rising to become the new engines of growth?
VCBeat has identified ten high-growth product segments by analyzing the performance of key players in the global medical device industry. These segments are growing at a year-over-year rate exceeding 15%, significantly outpacing the industry average. Leading companies in these fields, driven by innovative products, are achieving rapid growth and outperforming their peers.
Cardiac electrophysiology pulsed-field ablation has taken off at a staggering pace. As an innovative product, PFA achieved a penetration rate of over 20% in the U.S. electrophysiology market within less than two years after approval, propelling the market size beyond $2 billion. Investors believe it has the potential to become the fastest-growing segment in terms of revenue growth and market scale expansion in China and globally.

Image Source: Boston Scientific
The swift increase in PFA's penetration rate is primarily due to its outstanding clinical value. Unlike radiofrequency ablation and cryoablation, pulsed-field ablation offers high tissue selectivity. It effectively ablates diseased myocardial cells while minimizing damage to adjacent critical structures such as the esophagus, phrenic nerve, and coronary arteries. The initial ablation effect is well-established, with studies showing an immediate pulmonary vein isolation success rate of 98–100%. Results from multiple one-year follow-up studies indicate a single-procedure success rate of 70–80% for paroxysmal atrial fibrillation, which is non-inferior to traditional thermal ablation and, in some studies, demonstrates better long-term efficacy. PFA also reduces procedure time and enhances surgical efficiency.
In terms of commercialization, the strategic focus and strong promotional efforts of major industry players have significantly driven the rapid adoption of PFA. Boston Scientific and Medtronic are currently the dominant forces in the global PFA market. These two cardiovascular giants have invested substantial resources to advance PFA commercialization. Boston Scientific's Farapulse PFA system has surpassed 500,000 procedures worldwide. In fiscal year 2025, Medtronic's PFA ablation revenue reached approximately $1 billion, with an expected additional $1 billion increase in 2026. Moreover, the subsequent entry of Johnson & Johnson and Abbott—longstanding leaders in radiofrequency ablation for cardiac electrophysiology—into the PFA space provides ongoing validation and further momentum for this sector.
The PFA market is far from reaching saturation. During the J.P. Morgan Healthcare Conference at the beginning of 2026, Medtronic positioned PFA as a market exceeding $13 billion in size with an annual growth rate of over 25%, projecting that PFA penetration will rise to 80% by 2028. The strategic consensus among major players is to build a comprehensive ecosystem by enriching supporting mapping systems and catheter varieties, while leveraging synergies with other cardiovascular interventional businesses.
In contrast, China's domestic PFA market remains in the introductory phase. Over five China-developed PFA products have already received approval, driving a rapid increase in clinical application. Chinese products also demonstrate a relatively fast iteration pace, having taken the lead in multiple innovative technologies within the PFA field. It is anticipated that through industry consolidation, gradual improvement of product support, continuous optimization of stability, and enhanced imaging precision, Chinese cardiac electrophysiology companies are poised to achieve significant growth leveraging PFA technology.
The rapid rise of single-use endoscopes in the global medical device landscape is the result of a convergence between clinical needs and industry transformation.
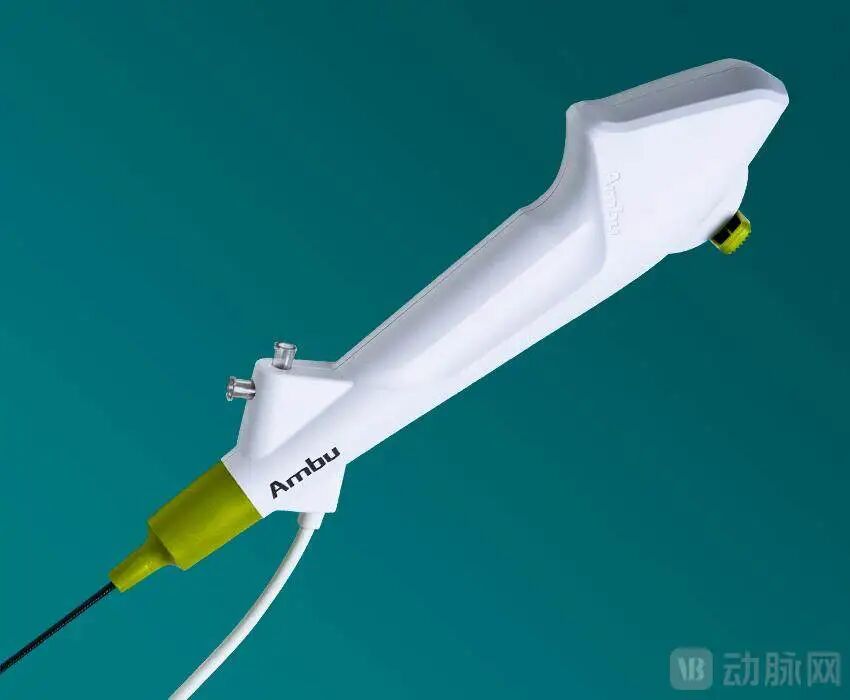
Single-use Endoscope
Image Source: Ambu
From a clinical value perspective, single-use endoscopes address the issue of cross-infection associated with reusable endoscopes, while significantly reducing hospitals' procurement and sterilization costs. In recent years, the penetration rate of single-use endoscopes has risen rapidly worldwide. After achieving high penetration in urology, their application is quickly expanding across multiple clinical departments.
From an industrial transformation standpoint, single-use endoscopes are a product born from technological revolution and industrial innovation. The rapid advancement of CMOS image sensor chip technology, coupled with the localization and controllability of domestic chips in China, alongside large-scale automated manufacturing, has substantially lowered the cost of single-use endoscopes. This has laid the foundation for their application across diverse medical specialties.
The performance of global leaders provides a clear signal of continued growth. In the first three quarters of 2025, Ambu, a pioneer in single-use endoscopy, reported revenue growth exceeding 25% year-over-year in its urology, ENT (ear, nose, and throat), and gastrointestinal divisions. Despite a slowdown in the respiratory segment, the overall business maintained strong momentum. CEO Britt Meelby Jensen projects that the market will continue to sustain a compound annual growth rate exceeding 20%. Companies are not merely following demand but actively driving a shift in usage habits across departments. Looking ahead, Ambu plans to focus primarily on urology and respiratory applications, followed by ENT, gradually transitioning these specialties from reusable to single-use endoscopic practices.
The single-use endoscopy market still holds vast growth potential. While the global endoscopy market grows at approximately 5% annually, the single-use endoscopy segment has maintained a growth rate of over 20% for two consecutive years. As aging populations, chronic diseases, and demand for minimally invasive surgeries continue to rise, hospitals' reliance on safe, efficient, and low-cost solutions will further intensify. In the future, as single-use endoscopes achieve image clarity comparable to—or even surpassing—traditional scopes, they hold the potential to replace reusable devices in areas like gastroscopy and colonoscopy.
Leveraging supply chain advantages and rapid response capabilities, Chinese companies have emerged as a core global supply force. Through partnerships with industry giants such as Olympus, Cook Medical, and Hologic, they are bringing "Made in China" solutions to the global stage. In 2026, the inclusion of single-use flexible ureteroscopes in China's national procurement program is expected to drive prices down significantly. This will further accelerate domestic penetration in China, creating a dual momentum of surging demand and declining costs.
Over the past two decades since their introduction, robotic-assisted laparoscopic systems have achieved a market size exceeding $10 billion with an average annual growth rate of over 20%, despite a global penetration rate of less than 15% in minimally invasive surgeries. This longevity surpasses the typical lifecycle of innovative medical devices.
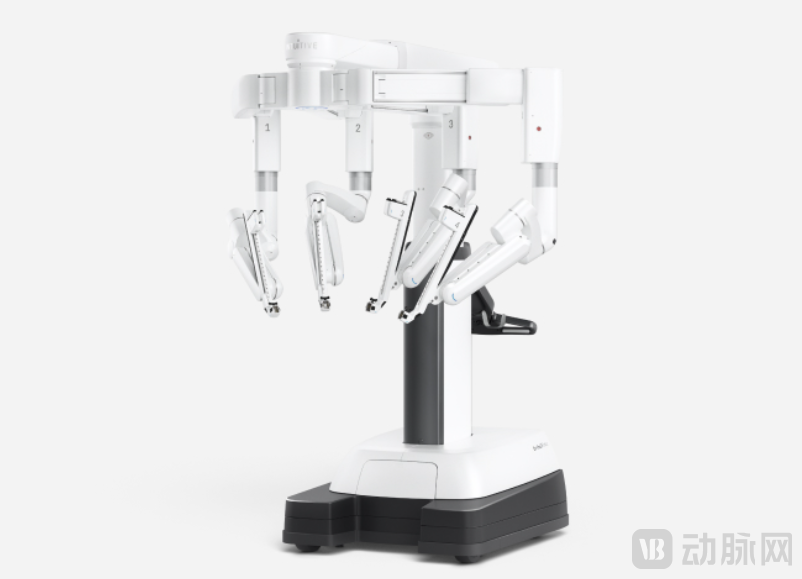
Da Vinci Surgical System Robotic Arm
Image Source: Intuitive Surgical
The enduring success of robotic-assisted laparoscopic systems is fundamentally rooted in their widely recognized clinical value. This value extends across multiple surgical specialties, primarily enhancing high-risk oncological procedures by significantly reducing the technical complexity of laparoscopic surgeries. A typical laparoscopic surgical robot system consists of a surgeon console, a patient-side cart, and an imaging system. The surgeon remotely controls surgical instruments attached to robotic arms via the patient-side cart to perform procedures such as compression, cutting, coagulation, dissection, suturing, and tissue manipulation. Compared to manual operation, this approach offers greater precision and facilitates surgery within confined spaces.
New products from Intuitive Surgical continue to inject sustained growth momentum into the market. The global robotic-assisted laparoscopic surgery market is dominated by Intuitive Surgical's Da Vinci Surgical System. Over more than two decades since its introduction, the Da Vinci platform has undergone five major iterations, each bringing significant leaps in imaging display, advanced instrument integration, and operability. The latest generation, the Da Vinci 5 (approved by the FDA in 2024), features enhanced computational power and force feedback capabilities. This new system has achieved a cumulative installation of 1,232 units over two years—870 in 2025 and 362 in 2024. Continuous innovation has been a key driver of ongoing market expansion.
This sustained innovative capability has also delivered substantial financial returns. In 2025, Intuitive Surgical, the parent company of the Da Vinci Surgical System, reported revenue exceeding $10 billion, a year-over-year increase of 21%. The annual procedure volume growth for the Da Vinci system in 2025 is projected to be between 13% and 15%. Throughout 2025, a total of 1,721 Da Vinci surgical systems were installed, surpassing the 1,526 installations recorded in 2024.
Competition in the surgical robot market is expected to intensify in 2026. At the JPM Healthcare Conference, both Johnson & Johnson and Medtronic highlighted laparoscopic surgical robots as key strategic initiatives. A major point of differentiation for these two industry giants compared to the Da Vinci system lies in the integration of surgical robots with advanced minimally invasive surgical instruments.
The Chinese domestic surgical robot market is also entering a period of growth opportunity. According to data from Zhongcheng Big Data Technology, 119 units of domestic laparoscopic surgical robots were sold in China from January to November 2025. Concurrently, Chinese-made laparoscopic surgical robots achieved stronger-than-expected growth in overseas markets in 2025. Having established channels and brand presence in emerging markets, Chinese laparoscopic surgical robots are projected to sustain this growth. The anticipated further relaxation of large medical equipment configuration permits in China in 2026 is expected to drive the expansion of the domestic laparoscopic surgical robot market.
While robotic-assisted laparoscopic systems represent a "heavyweight innovation" in medical technology with their vast and intricate engineering architecture, Continuous Glucose Monitors (CGM) demonstrate that a coin-sized product can equally carve out a rapidly growing multibillion-dollar market through its miniaturized and intelligent design.

CGM
Image Source: Yuwell Medical
CGM addresses a persistent challenge in chronic disease management by leveraging innovations in biosensors and algorithmic technology to resolve key difficulties in diabetes care. CGM technology uses a glucose sensor to continuously monitor the concentration of glucose in subcutaneous interstitial fluid, indirectly reflecting blood glucose levels and providing comprehensive, real-time data throughout the day.
From an industry promotion perspective, rapid coverage by U.S. health insurance has accelerated the widespread adoption of CGM. The global CGM market is dominated by Abbott and Dexcom. U.S. health insurance began covering CGM as early as 2017 and has continuously expanded its coverage scope, significantly reducing the financial burden on patients adopting this innovative technology. Abbott's CGM sales are projected to exceed $8 billion in 2025, representing a year-over-year increase of approximately 23%. Dexcom expects its 2025 revenue to reach between $4.63 billion and $4.65 billion, reflecting growth of about 15%.
CGM still holds substantial room for market growth. Abbott estimates that current CGM penetration is around 20% in the United States and approximately 5% globally, indicating significant room for further adoption. Abbott anticipates continued growth in the CGM market through 2026.
In China, although CGM has not yet been included in the national medical insurance coverage, leveraging the cost advantages of local enterprises in supply chain and manufacturing, CGM has successfully penetrated the self-pay market. The performance of industry leaders has been particularly outstanding: MicroTech Medical reported CGM revenue of RMB 143 million in the first half of 2025, a year-over-year increase of 91.5%. Yuwell Medical noted significant growth in its CGM business, with its market share rising rapidly. It is evident that, driven by both cost reductions and expanding demand, the domestic CGM market in China is entering an accelerated penetration phase fueled primarily by out-of-pocket spending.
Mechanical thrombectomy kicked off 2026 with a multibillion-dollar acquisition, with major players' strategic bets lending long-term value endorsement to this field.
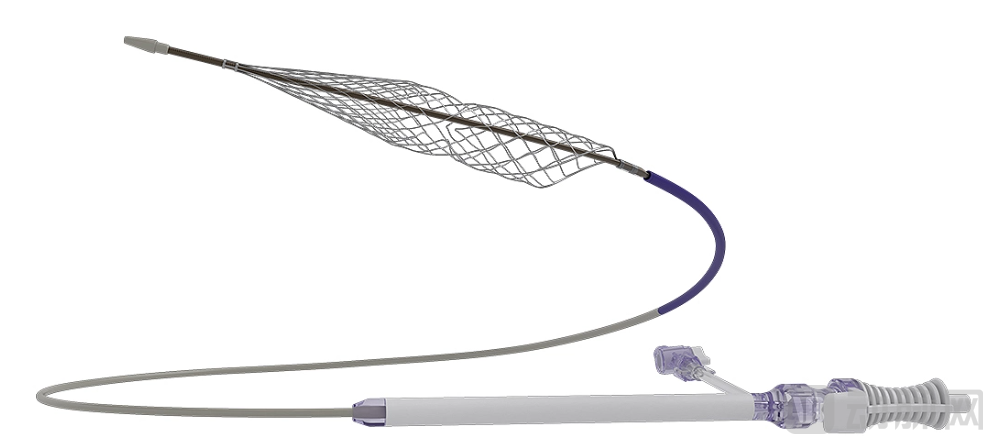
InThrill Thrombectomy System Image
Source: Stryker
From a clinical perspective, within the vast and complex field of vascular intervention, mechanical thrombectomy has rapidly distinguished itself due to its broad indication coverage, rapid vessel recanalization, and reduced procedure time. This minimally invasive, percutaneous approach delivers specialized devices directly to the thrombus site for mechanical removal, proving particularly effective for patients with acute, subacute, central, or mixed deep vein thrombosis. Compared to catheter-directed thrombolysis, it offers greater time efficiency, reduced pharmacological load, and shorter hospital stays, with advantages being especially pronounced in patients with lower bleeding risk and longer life expectancy.
Significant activity on the industry side has further propelled this segment into the spotlight. The global market is led by Inari Medical and Penumbra, both of which have been acquired by industry giants for substantial sums.
Inari Medical was acquired by Stryker for $4.9 billion in 2025. Following the acquisition, Inari contributed $590 million in revenue within ten months, achieving a remarkable year-over-year growth of 52.3% in Q2 2025 and becoming Stryker's fastest-growing business segment.
Penumbra, with its core thrombus aspiration systems, was acquired by Boston Scientific for $14.5 billion in 2026. Penumbra projects a Q4 2025 revenue growth between 21.4% and 22.0%, with full-year 2025 revenue expected to reach approximately $1.4 billion, representing year-over-year growth of 17.3% to 17.5%.
The Chinese domestic market also shows strong momentum. A large patient base with vascular diseases, combined with advancing technological maturity, supports a sustained high growth rate exceeding 20% for mechanical thrombectomy in China. Multiple domestically developed products have already received regulatory approval. As more vascular intervention companies expand their complementary product portfolios, future market penetration is expected to increase further.
Structural heart disease represents one of the fastest-growing fields in cardiology in recent years, with transcatheter mitral and tricuspid therapies (TMTT) emerging as one of the most rapidly advancing segments within it.
The rise of TMTT as one of the fastest-growing sectors in the medical device industry is no coincidence—it is the result of converging clinical value, industrial momentum, and future potential.
From a clinical value perspective, transcatheter mitral and tricuspid therapies are reshaping the landscape of cardiovascular care. Traditional open-heart surgery poses high risks for elderly patients, whereas TMTT offers a minimally invasive approach that effectively addresses the most challenging treatment gaps for valvular diseases in an aging population, driving a structural shift in global treatment paradigms.
From an industry momentum perspective, the performance of multinational leaders provides the most direct evidence. Abbott's mitral and tricuspid business continues to grow steadily, while Edwards Lifesciences reported TMTT revenue of $1.452 billion in Q3 2025, soaring 59.3% year-over-year, with the PASCAL and EVOQUE systems serving as dual growth engines. Sustained corporate investment and rapid product iteration are fueling a growth trajectory characterized by simultaneous technological breakthroughs and accelerated commercialization.
In the Chinese domestic market, catch-up is also accelerating: the number of approved TMTT devices expanded from three in 2024 to six in 2025. This leap in supply is igniting latent demand.
Looking ahead, the story of this market is far from its peak. Industry players widely recognize that TMTT penetration remains in its early stages, with the true growth cycle just beginning. In China, the combination of a vast patient population and rapidly expanding innovative supply is poised to give this segment long-term explosive potential.
Transbronchial surgical robots emerged as one of the fastest-growing segments in the medical device field in 2025, with this product category achieving large-scale commercial adoption during the year.
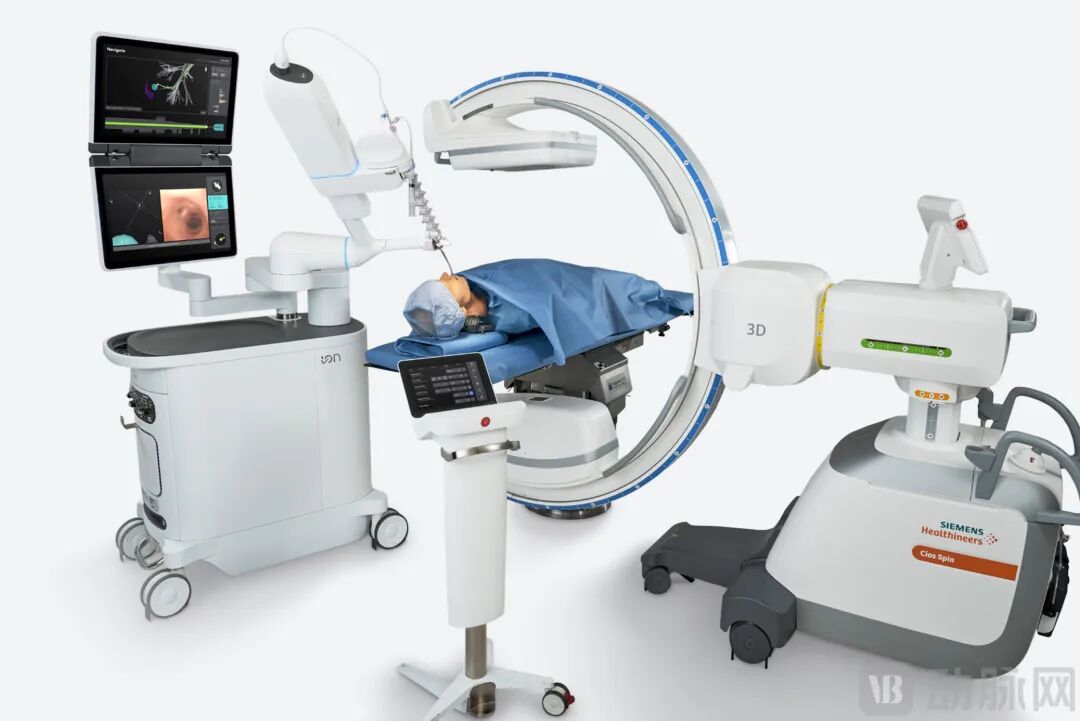
Schematic Diagram of Transbronchial Ion Application
Transbronchial surgical robots address the core challenges in early lung cancer screening and diagnosis. By integrating robotic navigation and remote control technologies, these systems enable pre-operative 3D reconstruction of the bronchial tree and trajectory planning, combined with intraoperative fusion of virtual imaging and real-time navigation. Surgeons can remotely and precisely control the distal movement of flexible bronchoscopes via an operator console, reaching over 90% of lung segments—including sixth-order and higher subsegmental bronchi—while performing real-time, accurate biopsy or ablation procedures. This significantly enhances the diagnostic accuracy for lung lesions and improves early-stage therapeutic outcomes.
The year 2025 marked the true beginning of large-scale commercial adoption for transbronchial surgical robots. Globally, three major robotic bronchoscopy systems received FDA approval: the single-arm Intuitive Surgical Ion platform, Johnson & Johnson's dual-arm Monarch platform, and Noah Medical's Galaxy system.
The adoption momentum is clearly reflected in clinical data. The Intuitive Surgical Ion platform has achieved installations of 905 units and a cumulative procedure volume of 38,000 cases, with procedure growth exceeding 50% in 2025. This rapidly rising utilization rate signals that robotic bronchoscopy has entered a phase of scaled commercialization—moving beyond the initial installation-validation period into a stage of demand-driven, large-scale growth.
Positive developments have also emerged in the Chinese domestic market. The National Healthcare Security Administration formally issued the Guidelines for the Establishment of Medical Services Price Items for Surgical and Therapeutic Auxiliary Operations (Trial), establishing a national pricing framework for this field. Looking ahead, products capable of integrating both diagnostic and therapeutic functions are expected to capture a larger market share.
In 2025, the medical aesthetics market faced headwinds, with growth under pressure globally. Yet certain products achieved rapid growth and defied the downturn. The "Baby Face" filler notably saw high-speed expansion in the Chinese domestic market.
Poly-L-lactic acid (PLLA), commonly known as the "Baby Face" filler, emerged as a high-growth segment within the medical device sector in 2025. This is primarily because its mechanism of action—"stimulating collagen regeneration"—aligns with the preference of Chinese beauty seekers for natural, long-lasting, and low-risk anti-aging solutions. As aesthetic consumption shifts from "filling-based" to "regeneration-based," PLLA, with its longer-lasting tissue-improving capabilities, has quickly become one of the key treatment focuses for medical institutions. It is also driving the expansion trend of regenerative medicine-based aesthetic products.
The growth drivers in the Chinese domestic market mainly stem from three aspects: First, consumer acceptance of "non-invasive or minimally invasive anti-aging treatments" continues to rise, characterized by high per-customer spending and repeat purchase rates. Second, increasingly stringent regulatory oversight on compliant medical aesthetic devices has made PLLA products with clinical evidence and regulatory qualifications more competitive. Third, ongoing innovation by Chinese companies in production processes, particle stability, and injection experience has lowered barriers to use and accelerated import substitution.
The current market landscape shows a pattern of "leading international brands, with Chinese brands catching up rapidly." International manufacturers dominate the high-end market relying on established clinical data and brand recognition, while Chinese enterprises are rapidly expanding their market share through competitive pricing and extensive channel penetration. Looking forward, the PLLA segment is expected to develop toward product differentiation, expanded indications, and combined treatment regimens. It is projected to maintain a high growth rate from 2025 to 2027, positioning itself as one of the most promising sub-categories in the regenerative aesthetics field.
The home medical device sector has also given rise to a hit product: non-invasive ventilators. The rapid growth of non-invasive ventilators is closely linked to rising consumer health awareness, the reshaping of market dynamics, and the global expansion strategies of domestic Chinese manufacturers.

ResMed Non-Invasive Ventilator
Image Source: ResMed Official Website
In 2025, the non-invasive ventilator market maintained strong growth momentum, with a notable rebound particularly in the U.S. market. Non-invasive ventilators are mechanical ventilation devices that continuously deliver positive airflow to the user through the mouth and/or nose in a non-invasive manner, aimed at controlling or assisting breathing. They are primarily used to treat Obstructive Sleep Apnea (OSA).
The recall of Philips ventilators reshaped the global ventilator market landscape, with domestic Chinese companies gaining a first-mover advantage through their global expansion strategies. BMC Medical reported revenue exceeding 800 million RMB for the first three quarters, a year-over-year increase of 34.24%, with net profit reaching 180 million RMB, up 43.87% year-over-year. Of this, overseas revenue accounted for 558 million RMB, growing by 52.96% year-over-year. Yuwell Medical's respiratory therapy products, serving as pioneers in its global expansion, maintained rapid growth in ventilator and mask sales, achieving breakthroughs in key countries and regions.
Against the backdrop of rising consumer health awareness, the non-invasive ventilator market is expected to continue its robust growth. At the JPM Healthcare Conference, ResMed noted that the treatment penetration rate for Obstructive Sleep Apnea Hypopnea Syndrome (OSA) remains low, with penetration below 20% in the U.S. and even lower than 10% in other global regions. Factors driving market expansion include increased consumer health awareness, the growing focus on sleep apnea associated with the widespread use of GLP-1 drugs, continuous sleep quality monitoring via wearable devices, and increased emphasis from payers and providers on preventive treatment amid rising healthcare system pressures.
The vascular intervention field has witnessed the emergence of another blockbuster product exceeding $1 billion in revenue: the Shockwave Intravascular Lithotripsy (IVL) Balloon. It has become Johnson & Johnson's 13th medical device product to surpass the $1 billion sales milestone.

Shockwave E8 Hero
Image Source: Johnson & Johnson Official Website
The core clinical value driving IVL's breakthrough to the $1 billion scale and high growth lies in its ability to effectively address the challenge of vascular calcification. This technology uses sonic pressure waves to precisely target calcified lesions within the coronary intima and media. It effectively fractures or loosens calcified tissue without damaging the vascular endothelium, restoring vessel compliance and creating more favorable conditions for subsequent stent implantation.
According to Johnson & Johnson, IVL's penetration rate in U.S. coronary intervention procedures is approximately 10%. Beyond Johnson & Johnson, companies such as Boston Scientific and Abbott are also actively developing in this field. Considering that about 30% of patients with coronary atherosclerosis present with calcified lesions, IVL still holds significant potential for further clinical adoption. With more products expected to enter the market, the sector is projected to expand further.
From the review of the above major fields, it is evident that innovative products are creating new opportunities for growth in the medical device industry and driving high-speed market expansion. The ability to strategically position in high-growth segments and to sustain innovation after product launch has become a key competitive factor. In China's domestic market, competition in innovation is generally more intense. Companies must navigate fierce competition to secure market share before achieving steady growth.
