Corxel Pharmaceuticals completes $287 million Series D1 financing to develop oral small-molecule GLP-1 RA

CORXEL
Developer of Oral Small Molecule Cardiac Myosin Inhibitors
On January 22, Corxel Pharmaceuticals announced the successful completion of its Series D1 financing, raising $287 million. The proceeds from this round of financing will be primarily used to advance the development of its investigational pipeline asset, CX11.

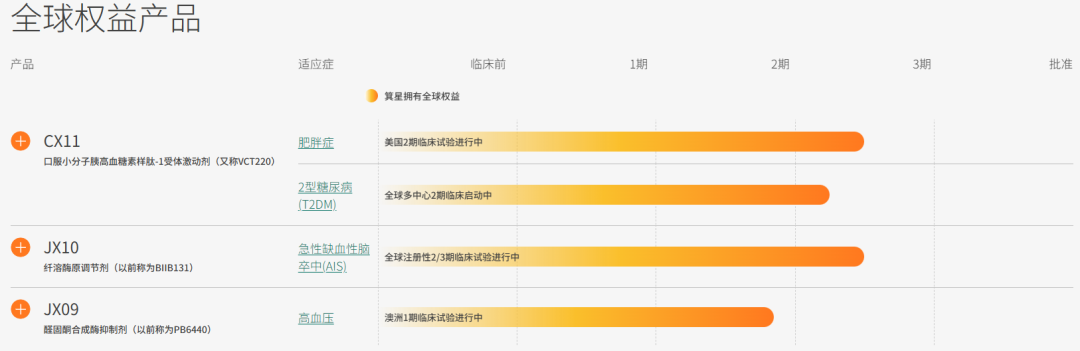
Its pipeline includes CX11, a differentiated oral small molecule GLP-1 RA for obese and overweight patients currently being evaluated in a Phase 2 trial conducted by Corxel in the United States and a Phase 3 trial conducted by Vincentage in China, and other cardiometabolic programs, including for acute ischemic stroke and hypertension.
The proceeds of this round of fundraising will be used for:
Advance the lead program, CX11, through its ongoing U.S. Phase 2 trial to treat obesity and overweight patients, its planned global Phase 2 trial to treat Type 2 Diabetes Mellitus (T2DM) and initial preparations for CX11's Phase 3 trials;
Advance the Corxel's additional programs through clinical development;
Further strengthen Corxel's global operational and development capabilities to support global multiregional clinical programs.
Corxel Pharmaceuticals was incubated by RTW in 2019 and completed a Series D financing round of $162 million in 2024. Bayer and RTW made equity investments in Corxel Pharmaceuticals (Shanghai), amounting to $35 million and $127 million, respectively.
On December 23, 2024, Corxel Pharmaceuticals in-licensed the global development and commercialization rights (excluding Greater China) for CX11 (also known as VCT220) from Vincentage.Currently, Corxel holds global rights to three products and Greater China rights to two products.