At JPM 2026: 5 Chinese biotechs in the spotlight, set to shake up key markets

RemeGen
Biological New Drug Developer

Simcere Zaiming
--
From January 12 to 15, 2026, the 44th J.P. Morgan Healthcare Conference (JPM 2026) was held as scheduled in San Francisco, USA. As the preeminent global summit in the pharmaceutical and healthcare sector, the JPM week represents the most intensive period for innovative drug asset transactions, marked by frequent high-profile deals. These landmark transactions often set the tone for the year's trends in licensing deals and pipeline advancement in the innovative drug space.
For China's innovative pharmaceutical industry, JPM holds milestone significance. From peripheral participation nearly two decades ago to securing a presence in the main conference venue today, JPM has witnessed the growth of Chinese drug innovation on the global stage. At JPM 2026, 24 Chinese innovative biopharmaceutical companies presented in the main conference and Asia-Pacific sessions, while an even greater number completed significant licensing deals with multinational corporations during the event. In key focus areas at JPM 2026, such as antibody-drug conjugates (ADCs), metabolic diseases, and cell and gene therapies, Chinese innovative drugs have become an indispensable source of value.
Chinese Biotech Takes the Global Stage
Despite pre-conference analyses suggesting that the initial surge of asset collaboration between multinational corporations (MNCs) and Chinese innovative drug companies had waned, the intensive series of major licensing deals during JPM 2026 demonstrated that, driven by patent cliffs, China remains a critical destination for MNCs to acquire assets. More significantly, at JPM 2026, China's innovative drug sector no longer merely followed trends or inferred annual industry directions from MNCs' fragmented remarks, but has established itself as a proactive driver of emerging trends.
In this article, we attempt to identify Chinese innovative pharmaceutical companies likely to reshape the competitive landscape in 2026 based on developments during JPM 2026. By 2026, MNCs acquiring Chinese innovative drug assets—even through major licensing deals—has already become a routine occurrence within the global pharmaceutical ecosystem. What could now meaningfully influence competition in specific therapeutic areas are the substantive changes these Chinese assets bring to clinical practice in the new cycle. We will therefore focus on Chinese innovative drug assets that, with MNC backing, have entered a new phase of development.
Blockbuster Deals Reshape the Race in Hot Targets
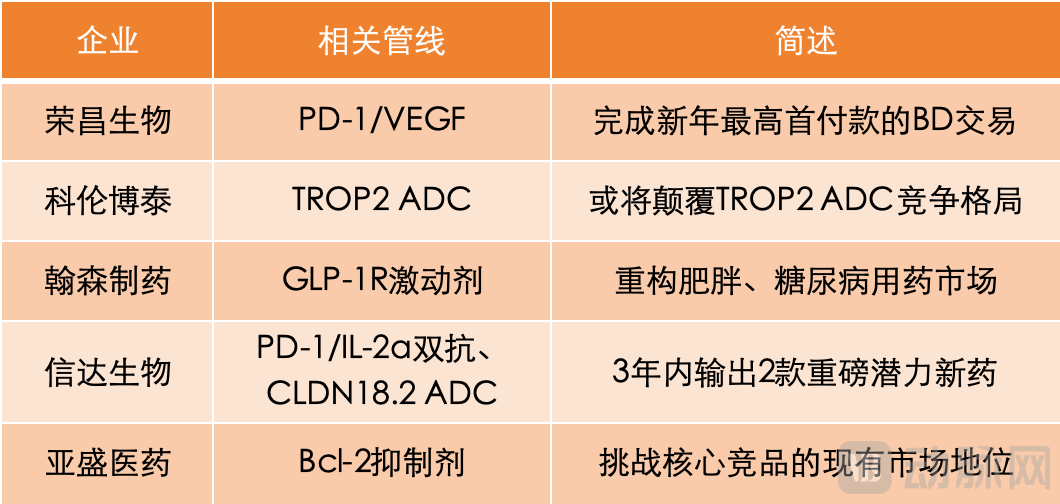
RemeGen executes the highest upfront payment licensing deal of the new year. On the first day of JPM 2026, RemeGen took the lead by announcing the signing of an exclusive licensing agreement with AbbVie, granting the latter exclusive rights for the development, production, and commercialization of its investigational product RC148 outside the Greater China region. In return, RemeGen will receive an upfront payment of USD 650 million and is eligible for up to USD 4.95 billion in development, regulatory, and sales milestone payments, as well as tiered double-digit royalties based on net sales outside Greater China. Following this, Chinese domestic innovative drug companies such as SciNeuro Pharmaceuticals and Simcere Zaiming also announced major transactions with MNCs like Novartis and AbbVie.
This was the first licensing deal finalized after the official start of the JPM 2026 agenda and the deal with the highest upfront payment during the four-day conference, setting a new record for the upfront payment received by a Chinese domestic innovative drug for a single pipeline asset.
RC148 is a PD-1/VEGF bispecific antibody currently in multiple Phase III and Phase II clinical trials both in China and globally. PD-1/VEGF bispecific antibodies represent arguably the most sought-after category in the history of cross-border licensing deals for Chinese innovative drugs, forming a classic "in China, for Global" landscape. Due to their synergistic anti-tumor mechanism combining "immune activation and anti-angiogenesis," PD-1/VEGF bispecifics are regarded as a next-generation therapy with high potential to succeed PD-1 monotherapies, making them a key focus for MNC asset acquisitions. Behind several leading PD-1/VEGF bispecific pipelines stand major MNCs.
Prior to this deal, RC148 was not considered the frontrunner in the rapidly advancing field of PD-1/VEGF bispecific development. Akeso's ivonescimab (AK112), as the world's first approved PD-1/VEGF bispecific antibody, has long been the global leader. In a head-to-head Phase III study, AK112 even demonstrated superior progression-free survival (PFS) data compared to the former "blockbuster" pembrolizumab. Behind AK112 stands Summit Therapeutics. In 2022, the two parties entered a licensing deal worth up to USD 5 billion, setting a record for out-licensing by a Chinese pharmaceutical company and igniting global interest in this therapeutic area. During JPM 2026, Summit stated it had submitted a marketing application to the U.S. FDA, marking another significant step in AK112's global development.
Furthermore, as part of the second wave of PD-1/VEGF bispecific pipelines, star assets like 3SBio's SSGJ-707 and LaNova Medicines' LM-299 have significantly accelerated their development through partnerships with MNCs. Following AK112, subsequent transactions related to PD-1/VEGF pipelines have continued to break records in deal value. For instance, in August 2025, 3SBio's SSGJ-707 was licensed to Pfizer for a record-breaking upfront payment of USD 1.25 billion, with a total deal value of up to USD 6 billion, while LaNova Medicines licensed its LM-299 to MSD. These pipelines, bolstered by substantial MNC investment and robust global clinical development capabilities, have seen their progress accelerate repeatedly.
With its clinical progress primarily focused on Phase II trials, RC148 was not the most prominent candidate within the PD-1/VEGF second tier. However, its clinical data are highly compelling. The deal with AbbVie may change this landscape. The renewed substantial cash flow will remove most obstacles in the pipeline's clinical development, while the infusion of AbbVie's pharmaceutical and clinical resources will greatly enhance the efficiency of RC148's clinical advancement. Consequently, the competitive positioning of RemeGen, as AbbVie's partner, is poised to shift accordingly.
Patent Cliffs Compel MNCs to Bet Big on Chinese Pipelines
At the annual JPM conference, MNCs outline their key R&D and commercialization strategies for the upcoming period. By 2026, addressing impending patent cliffs has become a strategic priority for MNCs. VCBeat notes that, in navigating these patent cliffs, in‑licensed pipelines from Chinese innovative drug companies have become a critical leverage for several MNCs, including MSD, Regeneron Pharmaceuticals, and Takeda.
Kelun‑Biotech may reshape the competitive landscape of TROP2 ADCs. During JPM 2026, MSD CEO Rob Davis repeatedly mentioned sacituzumab tirumotecan (SKB264/MK‑2870) in his discussion of future strategies. Rob Davis stated that in 2025, sacituzumab tirumotecan initiated multiple Phase III clinical studies, with primary endpoint data expected to be reported from 2027 onward.
Sacituzumab tirumotecan was originally developed by Kelun‑Biotech. In May 2022, Kelun‑Biotech out‑licensed the exclusive global development, production, and commercialization rights for sacituzumab tirumotecan outside Greater China, receiving an upfront payment of USD 47 million and potential milestone payments exceeding USD 1.3 billion for development, regulatory, and commercial milestones. This deal stands as a benchmark early‑stage out‑licensing transaction for Chinese ADCs. More than three years after the agreement, sacituzumab tirumotecan has become one of MSD's most important late‑stage pipeline assets.
Within MSD's oncology portfolio, as the patent cliff for pembrolizumab approaches, identifying the next growth engine has become its most critical strategic task. In this context, sacituzumab tirumotecan, as an ADC that can be combined with pembrolizumab, carries high expectations from MSD. Currently, the TROP2 ADC field, in which sacituzumab tirumotecan operates, is one of the most intensely pursued R&D arenas. Leading candidates include Gilead's Trodelvy (sacituzumab govitecan) and AstraZeneca/Daiichi Sankyo's datopotamab deruxtecan, which hold clear advantages.
As the world's first approved TROP2 ADC, sacituzumab govitecan has been approved for multiple indications, including triple‑negative breast cancer, HR+/HER2‑ breast cancer, and urothelial carcinoma, securing first‑mover advantage and a broad market presence. Datopotamab deruxtecan, leveraging the powerful DXd technology platform, has gained approval for triple‑negative breast cancer and demonstrated “best‑in‑class” potential in areas such as non‑small cell lung cancer (NSCLC). Data from its lung cancer study presented at ESMO 2023 directly challenged the foundational role of chemotherapy.
With the initiation of multiple Phase III trials for sacituzumab tirumotecan in 2025, a new contender has entered the competitive TROP2 ADC market. As a later entrant, sacituzumab tirumotecan has the potential to overtake competitors in this specialized field, supported by MSD's extensive experience in oncology drug development. In this process, Kelun‑Biotech, which retains the rights to sacituzumab tirumotecan in China, has gained the capacity to influence the market landscape.
Hansoh Pharmaceutical may reshape the obesity and diabetes drug market. At JPM, Regeneron highlighted the small‑molecule GLP‑1R agonist olatorepatide (HS‑20094) as a key component of its strategy to enter the trillion‑dollar obesity and diabetes market. In June 2025, Hansoh Pharmaceutical entered into a licensing agreement with Regeneron for olatorepatide, receiving an upfront payment of USD 80 million. The total potential value of the deal could reach USD 1.93 billion, with Hansoh retaining rights to olatorepatide in the Chinese market.
Olatorepatide is a once‑weekly, subcutaneous, long‑acting GLP‑1/GIP dual‑receptor agonist. Phase II data indicate that its efficacy in weight loss and glucose reduction is comparable to semaglutide, with potential advantages in muscle preservation and tolerability. Prior to the licensing deal, Hansoh was advancing a Phase III trial for obesity and a Phase IIb study for type 2 diabetes in China, with key obesity Phase III data expected in the first half of 2026. Following the agreement, Regeneron plans to initiate global multicenter Phase III trials in 2026, covering obesity (including patients with comorbid type 2 diabetes).
For Regeneron, entering the obesity and type 2 diabetes arena dominated by Novo Nordisk and Eli Lilly requires a differentiated approach, and olatorepatide serves as an important asset in this effort. Regeneron views olatorepatide as a core component for combination with its own antibody therapies (e.g., GPR75 antibody), aiming to explore weight‑loss efficacy "beyond GLP‑1" or improved weight‑maintenance regimens.
Currently, the global obesity and diabetes market is overwhelmingly led by peptide‑based injectable drugs from Novo Nordisk and Eli Lilly. Oral formulations represent a key avenue for later entrants seeking breakthroughs.
As an oral small‑molecule drug, olatorepatide inherently offers better patient compliance than injectables, representing its greatest potential to challenge the current market dynamics. Early clinical data have demonstrated favorable weight‑loss effects and safety, positioning it as a potential "best‑in‑class" oral GLP‑1R agonist. Its small‑molecule profile makes it an ideal candidate for combination therapies, providing Regeneron with flexibility to explore various innovative treatment regimens.
Of course, compared to competitors that are already marketed or in late‑stage Phase III development, such as oral products from Pfizer and Eli Lilly, olatorepatide remains in Phase II and is unlikely to disrupt the market landscape for such drugs in 2026. Nevertheless, its unique combination therapy approach will undoubtedly introduce a significant new variable into this market.
Innovent Biologics delivers two high‑potential blockbuster drugs within three years. At JPM 2026, Takeda explicitly stated its plan to launch at least eight breakthrough, blockbuster‑potential drugs between 2026 and 2030. Two of these key pipeline assets — TAK‑928 (IBI363) and TAK‑921 (IBI343) — originate from Innovent Biologics, landing the company in the spotlight at JPM 2026.
In October 2025, Innovent entered into a licensing agreement with Takeda valued at up to USD 11.4 billion, which included rights related to IBI363 and IBI343. Under the agreement, Takeda secured global co‑development and U.S. commercialization rights for IBI363 and exclusive rights to IBI343 outside Greater China. Upon integration into Takeda's pipeline, IBI363 and IBI343 were assigned the new codes TAK‑928 and TAK‑921, respectively. Based on presentations at JPM 2026, these two assets are set to become key products in Takeda's future oncology portfolio.
TAK‑928 is a first‑in‑class PD‑1/IL‑2α‑biased bispecific antibody targeting treatment bottlenecks such as PD‑1 resistance, "cold" tumors, and PD‑L1 low‑expression tumors, with the potential to set a new benchmark for next‑generation immunotherapy. TAK‑921 is a CLDN18.2‑targeting ADC. CLDN18.2, the antibody target selected for TAK‑921, is the most prominent "star target" in gastric cancer after HER2.
Objectively, both TAK‑928 and TAK‑921 operate in highly competitive development areas. Prior to the Takeda agreement, however, their development progress and clinical data were already competitive within their respective fields. TAK‑928 has initiated a global Phase III trial for second‑line treatment of squamous non‑small cell lung cancer, making it the world's first PD‑1/IL‑2α bispecific antibody to enter Phase III. TAK‑921 has advanced into Phase III for gastric cancer and Phase I/II for pancreatic cancer, demonstrating clear differentiated advantages in these indications.
With access to Takeda's medical and clinical resources, these two oncology drugs now possess the potential to address unmet needs in their respective therapeutic areas. In this process, Innovent, which retains certain rights to TAK‑928 and TAK‑921, will also gain renewed competitiveness.
Homegrown Disruption: Challenging Established Markets
Ascentage Pharma challenges the existing market position of a core competitor. Ascentage Pharma's inclusion differs from the previously mentioned innovative drug companies whose prominence is tied to licensing deals, primarily due to the global registration trials and potential indication breakthroughs of its self-developed product, Lisaftoclax. At JPM 2026, Ascentage Pharma's founder, Dr. Dajun Yang, highlighted that in 2026, Lisaftoclax aims to achieve two key milestones: the completion of majority enrollment in the GLORA-4 trial (for first-line treatment of high-risk MDS) and the inclusion of Lisaftoclax in China's National Reimbursement Drug List (NRDL). The achievement of these milestones could enable Lisaftoclax to challenge the current market dominance of venetoclax, which is jointly developed by Roche and AbbVie.
In July 2025, Lisaftoclax was approved in China for the treatment of chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL) following BTK inhibitor failure, making it the first China-developed and globally second-approved Bcl-2 inhibitor. Previously, venetoclax, the world's first approved Bcl-2 inhibitor, had been approved in China in December 2020 for certain types of myeloid leukemia, CLL, and SLL. Starting March 2023, venetoclax was included in China's NRDL, reducing the out-of-pocket monthly treatment cost for patients to approximately RMB 3,000.
For Lisaftoclax, establishing a differentiated advantage is crucial. The GLORA-4 trial mentioned by Dr. Yang is the world's first registrational Phase III trial of a Bcl-2 inhibitor for previously untreated high-risk myelodysplastic syndromes (HR-MDS). For nearly two decades, no targeted therapy has been approved globally for HR-MDS. Current standard treatments for HR-MDS primarily involve hypomethylating agents (HMAs), which offer limited efficacy, with objective response rates (ORR) of only 30-40% and a 5-year survival rate of just 16-24%.
Meanwhile, venetoclax, the world's first approved Bcl-2 inhibitor, saw its Phase III trial for HR-MDS (VERONA) fail, leaving Lisaftoclax as the only Bcl-2 inhibitor currently advancing in a registrational Phase III trial for this indication. If GLORA-4 achieves majority enrollment in 2026 and subsequently reports positive data, Lisaftoclax could potentially become the world's first Bcl-2 inhibitor approved for first-line HR-MDS between 2027 and 2028, capturing a global unmet market worth billions of dollars and potentially weakening venetoclax's dominance in myeloid malignancies.
Of course, during JPM 2026, the number of Chinese innovative pharmaceutical companies integrated into MNCs' annual strategies extended far beyond those mentioned above. Companies such as Zai Lab, 3SBio, and Zion Pharma also became significant components of MNCs' strategic plans for the new year through multiple collaborative pipelines. This article highlights only the most representative examples.
While JPM 2026 provided a powerful stage, whether the competitive landscape in the specific therapeutic segments of these five Chinese innovative drug companies fundamentally shifts in 2026 will ultimately be determined by clinical and commercial execution in the months ahead.