From caffeine giant to biotech contender: the strategic pivot of CSPC Innovation Pharmaceutical

CSPC Innovation
Biopharmaceutical, API, and Functional Food Developer

CSPC
Innovative Drug Research and Development, Manufacturer
There is a new development in the capital operations of the "CSPC Group" ecosystem.
On December 10, CSPC Innovation Pharmaceutical submitted its listing application to the Hong Kong Stock Exchange, commencing its journey for a Hong Kong IPO. Prior to this, CSPC Innovation Pharmaceutical was already listed on the A-share market as Xinnuowei (300765.SZ), which debuted on the Shenzhen Stock Exchange in March 2019. If this Hong Kong listing succeeds, CSPC Innovation Pharmaceutical will become another pharmaceutical company with dual primary listings on both the A-share and H-share markets ("A+H").
Tracing back to before 2019, Xinnuowei (the predecessor of CSPC Innovation Pharmaceutical) was actually a spin-off from the Hong Kong-listed CSPC Pharma for its return listing on the A-share market. Now, as it initiates its Hong Kong listing, the business and strategic direction of CSPC Innovation Pharmaceutical have undergone significant changes. This shift reflects a strategic repositioning by its ultimate controlling shareholder, CSPC Pharmaceutical Group. The intricate capital maneuvers within the "CSPC Group" ecosystem are ongoing.
In March 2019, Xinnuowei (300765.SZ), the predecessor of CSPC Innovation Pharmaceutical, was listed on the ChiNext Board of the Shenzhen Stock Exchange. Prior to this, Xinnuowei was a wholly-owned subsidiary of the Hong Kong-listed parent company, CSPC Pharma (1093.HK).
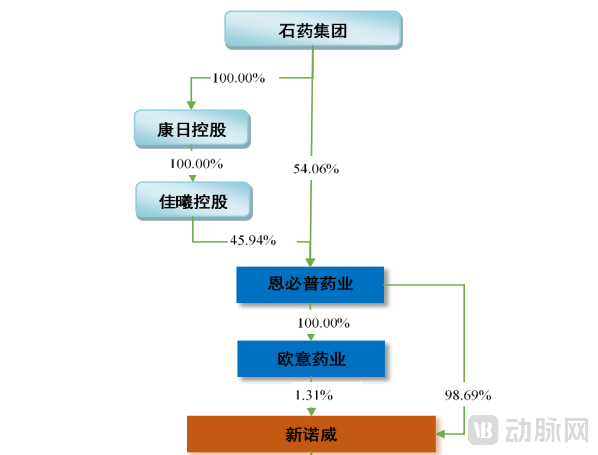
Figure 1. Equity Structure of Xinnuowei (predecessor of CSPC Innovation Pharmaceutical) Before Its A-Share Listing (Source: Xinnuowei's A-Share Listing Prospectus)
Through its A-share listing, Xinnuowei transitioned from being a wholly-owned subsidiary of CSPC Pharmaceutical Group into a publicly traded company. This move also established Xinnuowei as the first Chinese red-chip company to execute a spin-off and return listing on the A-share market.
Initially, Xinnuowei's core businesses were caffeine and health food products. Subsequently, it acquired equity in CSPC Shengxue, gaining access to the acarbose active pharmaceutical ingredient (API) and anhydrous glucose businesses, thereby expanding its operations into the functional ingredients sector.
A pivotal change in Xinnuowei's fundamental profile occurred in 2023. In September of that year, Xinnuowei announced its intention to acquire a 51% equity stake in CSPC Jushi Biologics, a subsidiary of CSPC Pharmaceutical Group, via a cash capital increase. In November 2023, the company changed its name to CSPC Innovation Pharmaceutical (abbreviated as CSPC Innovation), formally entering the innovative drug sector. This capital increase (amounting to 1.871 billion RMB) was completed in January 2024. Consequently, CSPC Innovation became a significant innovative drug platform under the CSPC Pharmaceutical Group umbrella.
On September 30, 2025, CSPC Innovation further acquired an additional 29% equity stake in CSPC Jushi Biologics in a cash transaction. Upon completion of this deal, CSPC Innovation's total ownership in CSPC Jushi Biologics increased from 51% to 80%.

Figure 2. The Development History of CSPC Innovation Pharmaceutical (Source: CSPC Innovation Pharmaceutical Prospectus)
As of the third quarter of this year, public information indicates that CSPC-NBP Pharmaceutical holds a 73.83% stake in CSPC Innovation Pharmaceutical, making it the largest shareholder. Its connected party, CSPC OUYI Pharmaceutical, holds a 0.82% stake. Consequently, CSPC Pharma maintains absolute controlling ownership over CSPC Innovation Pharmaceutical.
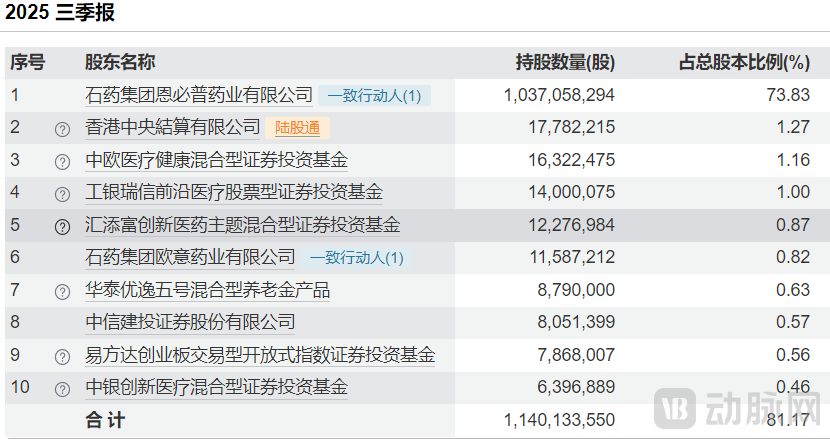
Figure 3. Top Ten Shareholders of CSPC Innovation Pharmaceutical in Q3 2025 (Source: Wind)
Following its successful Hong Kong listing, CSPC Innovation Pharmaceutical will have a cross-market presence spanning both the A-share and H-share markets. This dual-listing structure will enable CSPC Innovation Pharmaceutical to leverage the advantageous financing environments of both capital markets to better facilitate its business operations.
Currently, CSPC Innovation Pharmaceutical's traditional product portfolio is focused on two sectors: active pharmaceutical ingredients (APIs) and health supplements. The company exhibits significant economies of scale, particularly in the caffeine and acarbose segments.
As a food additive, caffeine is used in functional beverages. CSPC Innovation Pharmaceutical holds a globally leading position in this field. According to data from Frost & Sullivan, from 2020 to 2024, CSPC Innovation Pharmaceutical has consistently been the world's largest producer of chemically synthesized caffeine, based on both revenue and shipment volume.
In terms of market structure, statistics from Soochow Securities indicate that CSPC Innovation Pharmaceutical's caffeine production capacity accounts for approximately 50% of the market share in China. Furthermore, the company supplies an estimated 60% to 70% of the global demand for caffeine.
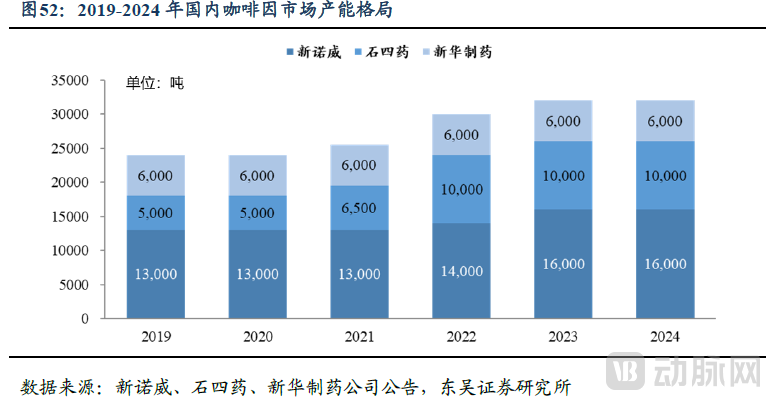
Figure 4. Caffeine Market Capacity Landscape (Source: Soochow Securities)
CSPC Innovation Pharmaceutical's caffeine products are currently exported to 65 countries and regions worldwide, primarily serving the North American and European markets. Leveraging consistent product quality, cost advantages from scaled production, and a robust supply chain system, CSPC Innovation Pharmaceutical has established stable cooperative relationships with leading international beverage industry clients. Its products serve industry giants such as Coca-Cola, PepsiCo, and Red Bull.
However, caffeine prices are cyclical. At the beginning of 2022, caffeine export prices reached a multi-year high, after which they have been in a state of continuous decline. This sustained price decline has consequently been a drag on the revenue of CSPC Innovation Pharmaceutical's caffeine business segment.
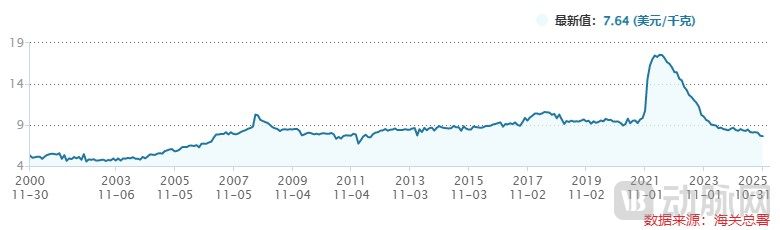
Figure 5. Unit Price of Caffeine and Its Salts Exports (Source: Wind)
While consolidating its leading position in the caffeine sector, CSPC Innovation Pharmaceutical also acquired a 100% equity stake in CSPC Shengxuein 2022. This acquisition expanded its product portfolio to include functional ingredients such as acarbose and anhydrous glucose, which are used as active pharmaceutical ingredients (APIs) or food additives. Furthermore, CSPC Innovation Pharmaceutical operates a functional food business, represented by its brand "Guoweikang" Vitamin C Effervescent Tablets.
The decline in caffeine product revenue has had a significant impact on CSPC Innovation Pharmaceutical's overall income. Financial reports show that the revenue from its functional food and ingredients segment dropped from 2.571 billion RMB in 2022 to 1.840 billion RMB in 2024, representing a decrease of 28.43%.
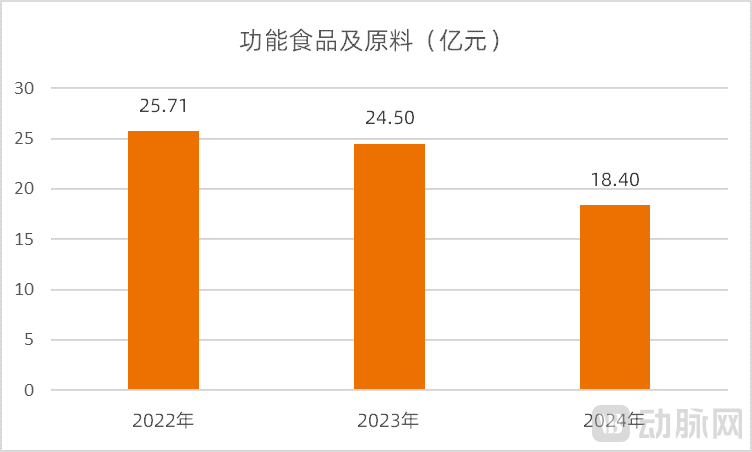
Figure 6. Revenue Changes in CSPC Innovation Pharmaceutical's Functional Foods and Ingredients Business (Source: Wind)
Perhaps recognizing the downturn in the functional food and ingredients business, and against the backdrop of China's booming innovative drug industry, CSPC Innovation Pharmaceutical made a major investment to acquire a stake in CSPC Jushi Biologics. This move propelled the company into the ranks of innovative pharmaceutical enterprises, resulting in a significant transformation of its fundamental business profile.
The capital market has responded with strong approval to this strategic shift. Following the announcement of the acquisition of CSPC Jushi Biologics, CSPC Innovation Pharmaceutical's stock price on the A-share market surged dramatically, with a maximum increase exceeding 500%. Although the share price has since retreated from its peak, it still maintains an appreciation of approximately 200%.
CSPC Innovation Pharmaceutical is strategically focused on key therapeutic areas including oncology, autoimmune diseases, and infectious diseases. The company has built technological advantages in antibody drugs, Antibody-Drug Conjugates (ADCs), and mRNA vaccines. Currently, its Class 1 innovative drug Enlonstobart Injection(Enshuxing) (an anti-PD-1 monoclonal antibody) and Omalizumab for Injection(Enyitan), China's first biosimilar of Omalizumab, have received marketing approval. In addition to these two antibody therapeutics, CSPC Innovation Pharmaceutical has two self-developed COVID-19 mRNA vaccines.
However, judging by the current commercialized products, the revenue contribution from CSPC Innovation Pharmaceutical's innovative drug business remains a small portion of its overall income. As of the end of July 2025, the revenue generated from sales of its biopharmaceutical products was only 118 million RMB.

Figure 7. Revenue from CSPC Innovation Pharmaceutical's sales of biopharmaceutical products (Source: CSPC Innovation Pharmaceutical prospectus, VCBeat)
Although CSPC Innovation Pharmaceutical's current revenue scale in the innovative drug sector remains relatively small, the company has a pipeline of 15 drug candidates in clinical or late-stage development. This pipeline includes 9 ADC candidates and 1 mRNA vaccine candidate, many of which show considerable promise.
CSPC Innovation Pharmaceutical's R&D pipeline is characterized by a focus on ADC drugs. These investigational products target indications such as lung cancer, breast cancer, gastric cancer, colorectal cancer, and esophageal squamous cell carcinoma, covering a range of targets including EGFR, Nectin-4, HER2, CLDN18.2, B7-H3, HER3, FRα, ROR1, and DLL3. Among the current pipeline, two ADC candidates, SYS6010 and SYS6002, demonstrate significant potential.
1) SYS6010: A Potential Best-in-Class EGFR ADC
SYS6010 utilizes a topoisomerase I inhibitor as its cytotoxic payload and directs it to cancer cells via EGFR targeting. This mechanism of action enables it to circumvent common resistance mechanisms associated with downregulated EGFR expression, maintaining efficacy even in resistant patients.
SYS6010 enters cells via endocytosis. Within lysosomes, it is degraded by proteases, releasing the small-molecule toxin JS-1. JS-1 forms a stable complex with DNA, inducing DNA damage and subsequently leading to apoptosis (programmed cell death).
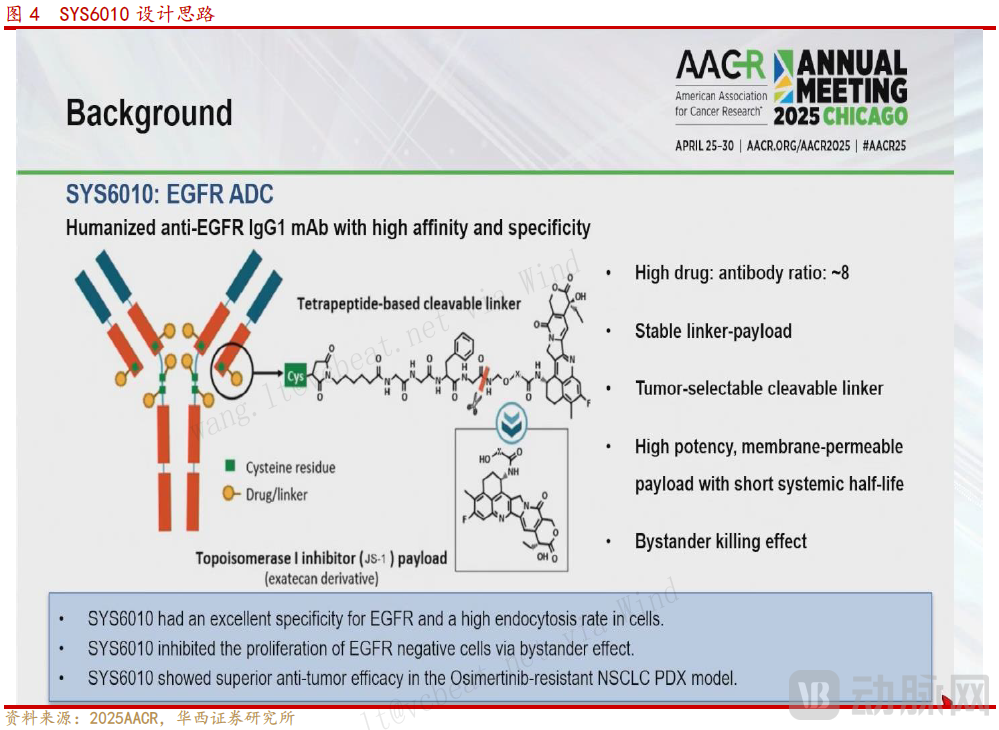
Figure 8. SYS6010 Design Concept (Source: Huaxi Securities)
In April 2025, data from the first-in-human study of SYS6010 in patients with advanced solid tumors was presented at the American Association for Cancer Research (AACR) Annual Meeting. In the 4.8 mg/kg dose cohort for EGFR-mutant non-squamous non-small cell lung cancer (nsq-NSCLC), the Objective Response Rate (ORR) and Disease Control Rate (DCR) were 46.9% and 93.9%, respectively. In the 4.2 mg/kg, 4.8 mg/kg, and 6.4 mg/kg dose cohorts for EGFR wild-type nsq-NSCLC, the ORR and DCR were 71.4% and 85.7%, respectively.
The clinical results indicate that SYS6010 has a manageable safety profile and demonstrates promising efficacy in patients with advanced solid tumors, particularly in those with EGFR TKI-resistant or EGFR wild-type nsq-NSCLC.
Currently, SYS6010 has successively received Breakthrough Therapy Designation from China's National Medical Products Administration (NMPA) and Fast Track Designation from the U.S. Food and Drug Administration (FDA).
2) SYS6002: A Promising Next-Generation Nectin-4 ADC with Potential Superiority
SYS6002 is an ADC designed to deliver the potent mitotic inhibitor MMAE precisely to cancer cells expressing Nectin-4, with a Drug-to-Antibody Ratio (DAR) of 2.
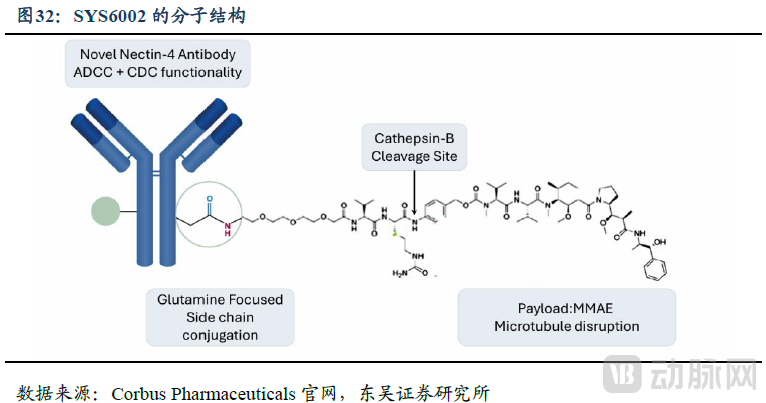
Figure 9. Molecular Structure of SYS6002 (Source: Soochow Securities)
Preliminary results from a Phase I clinical trial of SYS6002 were presented at the 2024 ASCO Annual Meeting. The data indicated that among evaluable patients with metastatic urothelial carcinoma at doses ≥1.2 mg/kg (n=9), the Objective Response Rate (ORR) was 44% and the Disease Control Rate (DCR) was 78%. Among evaluable cervical cancer patients at doses ≥1.2 mg/kg (n=7), the ORR was 43% and the DCR was 86%. Furthermore, SYS6002 demonstrated a more favorable safety profile compared to similar products in its class.
In February 2023, CSPC Jushi Biologics entered into an exclusive license agreement with Corbus Pharmaceuticals for the development and commercialization of SYS6002 in several countries, including the United States, the United Kingdom, Canada, and Australia. Under the agreement, CSPC Jushi Biologics received an upfront payment of $7.5 million and is eligible to receive up to $130 million in potential development and regulatory milestone payments, plus up to $555 million in potential sales milestone payments. Corbus Pharmaceuticals was granted the exclusive license to develop and commercialize SYS6002 in the specified territories.
Beyond these, CSPC Innovation Pharmaceutical has several other antibody-based and ADC drug candidates in its development pipeline.

Figure 10. Main R&D Products of CSPC Innovation Pharmaceutical (Source: Prospectus of CSPC Innovation Pharmaceutical)
SYSA1801 Injection is an ADC drug targeting Claudin 18.2. Its technology is similar to that of SYS6002, as both utilize MMAE as the cytotoxic payload and have a Drug-to-Antibody Ratio (DAR) of 2. SYSA1801 received Orphan Drug Designation from the U.S. FDA for gastric cancer in 2020 and for pancreatic cancer in 2021. In July 2022, the overseas rights for SYSA1801 were licensed to Elevation. The deal terms included an upfront payment of $27 million, potential milestone payments totaling up to $1.168 billion, and a share of sales royalties. Subsequently, due to clinical data not meeting expectations, the rights to this asset were returned to CSPC Innovation Pharmaceutical.
CSPC Innovation Pharmaceutical's expanding product pipeline and ongoing clinical trials have placed significant financial pressure on the company. Its traditional functional food and ingredients business can no longer adequately meet the substantial funding demands of drug R&D. In 2024, CSPC Innovation Pharmaceutical reported a net loss exceeding 300 million RMB. Following its successful Hong Kong stock market listing, CSPC Innovation Pharmaceutical will be able to leverage the advantageous financing environment of the Hong Kong market to better support its subsequent product development.
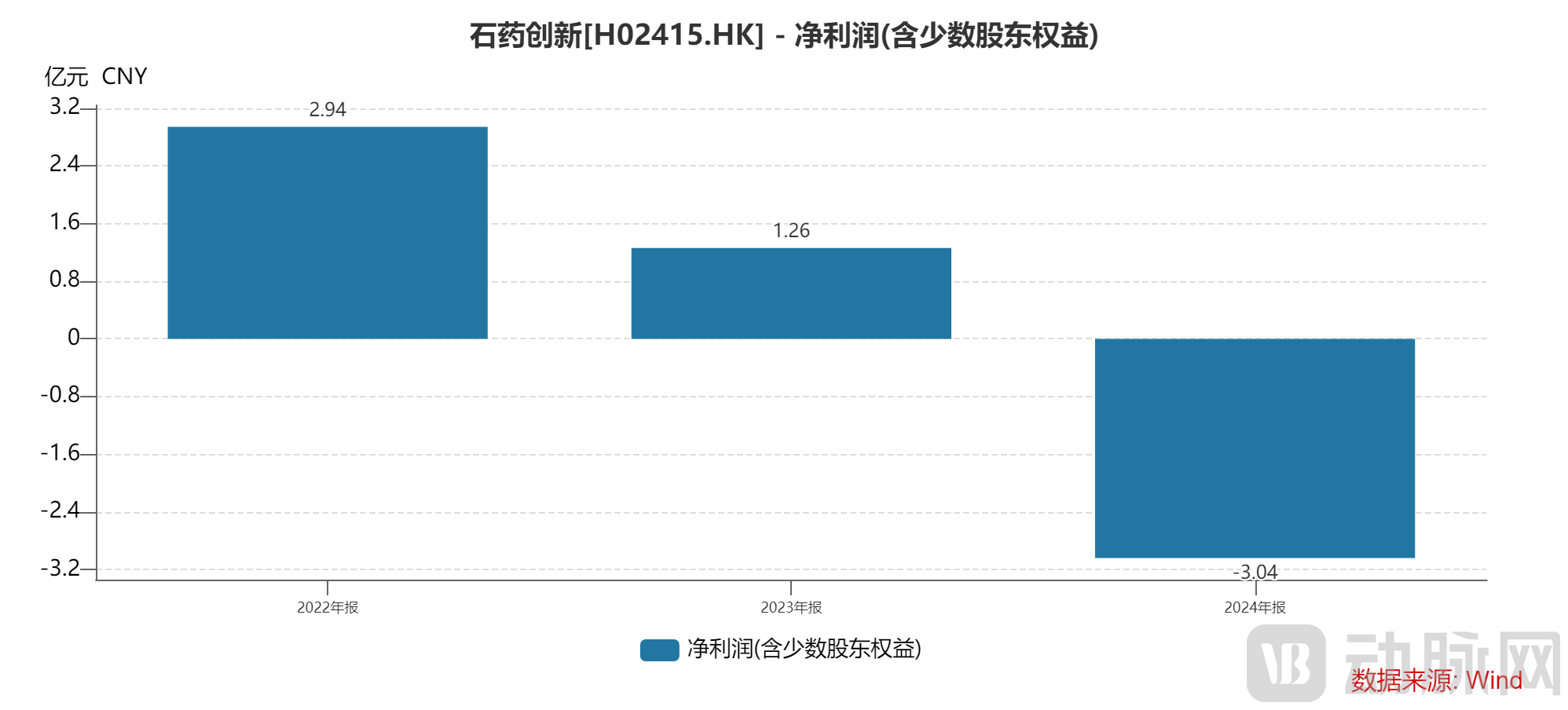
Figure 11. Net Profit of CSPC Innovation Pharmaceutical (Source: Wind)
Looking back at the development journey of CSPC Innovation Pharmaceutical, we see that its early-stage businesses primarily comprised functional ingredients such as caffeine, acarbose, and anhydrous glucose, as well as the functional foods sector. While these businesses lacked high-growth potential, they provided CSPC Innovation Pharmaceutical with stable cash flow.
Subsequently, by acquiring a controlling stake in CSPC Jushi Biologics, CSPC Innovation Pharmaceutical rapidly transformed into an innovative pharmaceutical company, marking the beginning of its accelerated development within the innovative drug sector.
If the initial investment in CSPC Jushi Biologics was CSPC Innovation Pharmaceutical's first step into the innovative drug arena, then its current push for a Hong Kong listing can be seen as the onset of an acceleration phase for its innovative drug business. This move signifies that CSPC Innovation Pharmaceutical can leverage the favorable financing environment and the international shareholder base of the Hong Kong market to better expand its operations. The resources and networks behind these international shareholders are expected to further accelerate the growth of CSPC Innovation Pharmaceutical's innovative drug business.
