Beyond the deal: Lyell inks $860M deal to acquire Chinese-developed solid tumor CAR-T asset from ICT
On November 10, Lyell Immunopharma, a late-stage clinical company advancing next-generation CAR-T cell therapies, announced a collaboration with Innovative Cellular Therapeutics (ICT) to acquire exclusive global rights—excluding mainland China, Hong Kong, Macau, and Taiwan—to research, develop, manufacture, and commercialize LYL273 (formerly known as GCC19CART).
Under the agreement, ICT will receive an upfront payment of $40 million and 1.9 million shares of Lyell common stock. ICT is also eligible for additional cash and equity considerations, as well as royalties on future net sales. The additional cash considerations include a potential $30 million clinical milestone payment, up to $115 million in late-stage regulatory milestones, and up to $675 million in commercial sales milestones. Upon achieving certain clinical and late-stage regulatory milestones, ICT may receive up to 1.85 million additional shares of Lyell common stock. The total potential value of the transaction is approximately $860 million.
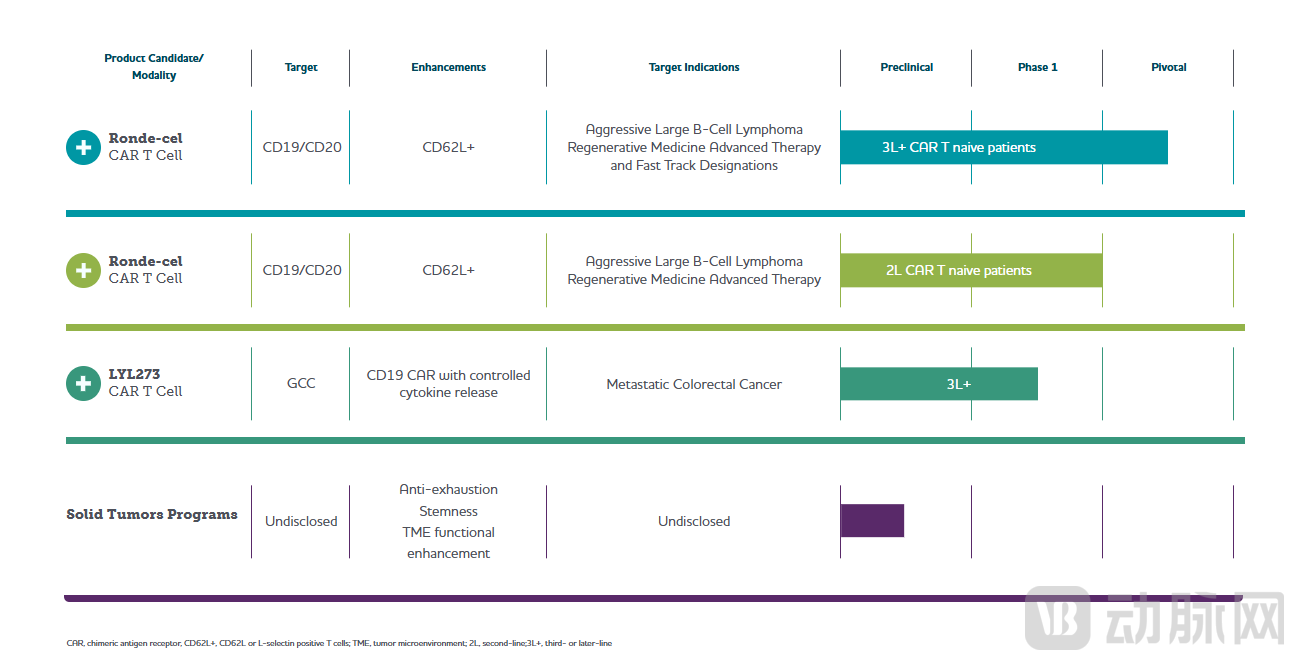
LYL273, which targets colorectal cancer—an area with significant unmet medical need—has received Fast Track designation from the FDA for the treatment of metastatic colorectal cancer (mCRC). The Phase 1 clinical trial is currently enrolling patients with refractory mCRC to determine the recommended Phase 2 dose.
1Exclusive Target: First Objective CAR-T Activity in Refractory Colorectal Cancer
Colorectal cancer (CRC) is the second leading cause of cancer deaths globally, with approximately half of all patients progressing to advanced stages. According to World Health Organization (WHO) data, there are over 1.9 million new CRC cases annually worldwide, with approximately 900,000 deaths each year from colon or rectal cancer. Patients with metastatic CRC (mCRC) face a poor prognosis, with a five-year survival rate of less than 15%, underscoring the urgent need for new treatment strategies. Guanylyl cyclase C (GCC), expressed in gastrointestinal epithelial cells, is an important target for treating CRC and mCRC.
Currently, ICT has a unique position in developing CAR-T therapies against this target.
LYL273 is a novel, autologous GCC-targeted CAR-T cell therapy candidate for treating mCRC and other GCC-expressing cancers. Data indicates that LYL273 is a lentiviral vector-transduced autologous CAR-T cell product composed of a GCC-targeting CAR for solid tumors, a CD19 CAR, and a cytokine. The inclusion of the CD19 CAR is designed to enhance the in vivo expansion of the solid tumor-targeting CAR-T cells, promote their migration and infiltration into tumors, and increase their cancer-killing capacity within the tumor microenvironment.
In 2024, a study from China published in JAMA Oncology suggested that GCC19CART was safe and tolerable for patients with pretreated mCRC. The article noted that this was the first study known to the authors where a CAR-T therapy demonstrated objective clinical activity in refractory colorectal cancer.
Data from the study showed that among 15 patients receiving either dose level, 6 patients (40%) achieved partial response (PR), 5 had stable disease (SD), and 4 experienced disease progression (PD), resulting in an overall disease control rate (DCR) of 73%. At the higher dose level of 2×10⁶ CAR-T cells/kg, the objective response rate (ORR) and DCR increased to 57.1% and 100%, respectively. Progression-free survival (PFS) was significantly longer in this group compared to the 1×10⁶ lower-dose group (6.0 months vs. 1.9 months), with a median overall survival (mOS) reaching 26.1 months.
In the ongoing U.S. Phase 1 clinical trial, according to Response Evaluation Criteria in Solid Tumors (RECIST) 1.1, patients with refractory mCRC treated with LYL273 achieved a 67% overall response rate and an 83% disease control rate (complete and partial responses plus stable disease), with a manageable safety profile at the highest dose level studied to date.
In August of this year, LYL273 completed key dose escalation in its U.S. Phase 1 trial for patients with relapsed/refractory metastatic colorectal cancer (R/R mCRC) who had received at least two prior lines of therapy. No Grade 3 or higher cytokine release syndrome (CRS) was observed in either the 1×10⁶ or 2×10⁶ CAR-T cells/kg dose cohorts. Notably, LYL273 demonstrated a favorable safety and efficacy profile at the higher dose level: among 5 evaluable patients at the 2×10⁶ CAR-T cells/kg dose, 4 (80%) achieved an objective response, with a median duration of response (mDoR) of 6.9 months.
2Solid Tumor CAR-T: Addressing Key Challenges of Expansion, Potency, and the Tumor Microenvironment
CAR-T therapy for solid tumors must address four key challenges: trafficking and infiltration of CAR-T cells, limited in vivo amplification, restricted tumor-killing capacity, and poor persistence due to T-cell exhaustion.
LYL273 (formerly GCC19CART) is based on ICT's proprietary CoupledCAR® platform, specifically designed for solid tumors. This platform employs a dual CAR approach to stimulate the immune system and enhance CAR-T cell expansion. It upregulates genes associated with tumor cell migration, thereby improving the impaired trafficking and infiltration of CAR-T cells into tumors. Furthermore, it enhances the cytotoxic activity of CAR-T cells within the immunosuppressive tumor microenvironment. Once the CoupledCAR-engineered cells enter solid tumors, the platform stimulates the body to generate and recruit additional immune cells, fostering a more effective and sustained antitumor response.
Dr. Larry Lei Xiao, founder of ICT, stated in a previous interview with VCBeat: "Although CAR-T can only target cell surface antigens, which account for less than 20% of the total cellular antigens, experience from hematologic tumors has shown that targeting a single surface antigen can effectively treat a blood cancer. In principle, therefore, one effective solid tumor target should be sufficient to treat a solid tumor. A considerable number of suitable solid tumor targets can be identified among cell surface antigens. For example, we have already identified GUCY2C for colorectal cancer and TSHR for thyroid cancer."
Dr. Xiao further emphasized, "The inability to target intracellular antigens should not be viewed as a fundamental limitation of CAR-T in treating solid tumors, nor should it be interpreted to mean that CAR-T is potentially less effective than TCR-T or TIL technologies. In reality, if all three technology platforms can identify suitable targets, they will subsequently face the same challenges, such as the immunosuppressive tumor microenvironment. CAR-T will undoubtedly advance and improve more rapidly due to its simplicity and ease of engineering, ultimately emerging as the leading approach."
ICT has now expanded its CoupledCAR® platform to other solid tumor programs, including its prostate cancer candidate PAPCAR-T, which has demonstrated significant clinical activity in data, as well as to other areas with substantial unmet clinical need such as ovarian cancer and thyroid cancer.
Dr. Larry Lei Xiao founded Innovative Cellular Therapeutics (ICT) in 2009 and assumed the role of full-time CEO in early 2016. After returning to China in 2005, Dr. Xiao served as a Principal Investigator at the Shanghai Institute of Biochemistry and Cell Biology. He had previously earned his Ph.D. from the University of Munich, Germany, and conducted postdoctoral research at Johns Hopkins University. Dr. Xiao possesses over 20 years of experience in the fields of embryonic/induced pluripotent stem cells, cell therapy, and gene editing.
As an early entrant in China's CAR-T biotech landscape, ICT has garnered support from multiple healthcare-focused investors and holds a global portfolio of over 100 patents and patent applications. The company completed Series A financing from CDH Investments and Kin Bridge Capital in 2016. In late 2017, it raised RMB 180 million in a Series B round from investors including Volcanics Venture, GTJA Investment Group, and SBCVC. A Series B+ round followed in 2019, led by LH Ventures. In 2021, ICT closed a Series C financing exceeding USD 40 million, led by CDH Venture and Growth Capital with participation from several other institutions.
3A Strategic Partnership: The Long-Term Value of a Cash and Equity Deal
From the transaction details, the "cash plus equity" structure reflects a strong collaborative nature. For ICT, the greater significance of this deal lies in the out-licensing value of GCC19CART, which will showcase and validate its proprietary CoupledCAR® platform for solid tumors to the global biopharmaceutical industry.
For Lyell, the acquisition of LYL273 will strengthen its solid tumor pipeline.
Lyell is a financially robust partner. The announcement indicates that Lyell now expects its 2025 net cash use to be between $155 million and $160 million, excluding the $40 million upfront payment for this transaction, which is below its previous guidance of $175 million to $185 million. Following the transaction closure, Lyell expects its cash reserves will be sufficient to fund operations into 2027, supporting data readouts and progress updates from its clinical program for large B-cell lymphoma, as well as additional clinical data from the Phase 1 trial of LYL273.
Founded in 2018 and headquartered in South San Francisco, California, Lyell focuses on overcoming T-cell exhaustion in cancer immunotherapy and is dedicated to developing next-generation CAR-T cell therapies for patients with hematologic malignancies and solid tumors.
In its early stages, Lyell advanced three T-cell therapy platforms in parallel: CAR-T, TCR-T, and TIL. In 2019, the company entered into an agreement with GSK to collaboratively develop TCR-T pipelines targeting NY-ESO-1 for solid tumors. Its core assets included the CAR-T therapy LYL797, TIL therapy LYL845, and TCR-T therapy NY-ESO-1C239.
However, in June 2024, a Phase 1 trial of its lead product candidate LYL797—a ROR1-targeted CAR-T therapy developed using its proprietary Gen-R technology platform for triple-negative breast cancer—reported one patient death. The patient experienced Grade 5 respiratory failure and died on Day 41. Dose-limiting toxicities related to pneumonitis occurred in 22% of patients, with the most severe events being Grade 3 or higher. Although the FDA did not immediately place the trial on hold, Lyell discontinued all clinical development of LYL797 four months later, along with abandoning all TIL programs, including LYL845.
Following this, Lyell promptly executed a strategic shift—advancing its internal early-stage pipeline while enriching its clinical-stage portfolio through acquisitions and licensing collaborations.
Internally, the company is developing LYL119, a next-generation ROR1-targeted CAR-T therapy built upon the LYL797 foundation and incorporating four novel reprogramming technologies. Preclinical studies indicate that LYL119 has the potential to deliver potent and durable responses and is now advancing into a Phase 1 clinical trial for patients with ROR1-positive solid tumors.
In October 2024, Lyell acquired ImmPACT Bio, paying a $30 million upfront payment and 37.5 million shares of its common stock to secure the rights to its most advanced clinical candidate, LYL314. This next-generation bispecific CD19/CD20-targeted CAR-T cell therapy has received Fast Track designation for the treatment of relapsed/refractory large B-cell lymphoma (LBCL) in the third-line or later setting. Lyell anticipates providing a progress update on the pivotal PiNACLE trial by year-end and plans to submit a Biologics License Application (BLA) in 2027. In April of this year, LYL314 also received the FDA’s Regenerative Medicine Advanced Therapy (RMAT) designation for the treatment of adult patients with relapsed or refractory LBCL after two or more prior lines of therapy.
On the capital front, Lyell maintains strong connections with China and is backed by similarly well-resourced strategic investors. Lyell completed its initial public offering on Nasdaq on June 17, 2021. Its major shareholders include ARCH Venture Partners, GSK, Foresite Capital, Gemini Investments, and Milky Way Investments. Its investor base also features prominent China-based institutions, including WuXi AppTec, Hillhouse Capital, and HSG.
Lyell's founder, Richard D. Klausner, M.D., has nearly three decades of experience in adoptive cell therapy research. An elected member of the National Academy of Sciences, the American Academy of Arts and Sciences, and the National Academy of Medicine, he previously served as the 11th Director of the National Cancer Institute and as Executive Director for Global Health at the Bill & Melinda Gates Foundation. A serial entrepreneur in healthcare, Dr. Klausner formerly held roles as Senior Vice President, Chief Medical Officer, and Chief Opportunity Officer at Illumina. He has also co-founded and helped lead several prominent biotechnology companies, including Juno Therapeutics, GRAIL, Sonoma Bio, and LifeMine Therapeutics.
At the 2021 HSG Global Healthcare Summit, Academician Richard D. Klausner noted in a conversation with Neil Shen, Global Executive Partner of HSG, that the growth of China's biotech industry over the past five years had been impressive in terms of both talent quality and innovation. He emphasized that the biotech sector needs to draw lessons from the technology industry, pointing out that leading companies in biotech are increasingly platform-based enterprises. These are not conventional CROs or tool providers, but rather scalable organizations like Moderna that can develop "apps" or even "killer apps" for everyday use in biotech. Klausner expressed his belief that a new wave of more ambitious and expansion-minded biotech companies would emerge, which would no longer simply aim for early acquisition by large pharma but would instead grow and scale much like technology companies.
For ICT, this moment has now arrived.
