MGI's $120M deal: what drives China's first life science instrument license-out?

MGI
Gene Sequencing Instruments and Related Reagent & Consumables R&D Manufacturer

Swiss Rockets
Vaccine Developer
On October 28, following approval at the 2025 Fourth Extraordinary General Meeting, MGI Tech Co., Ltd. ("MGI") announced a strategic technology licensing collaboration through its U.S. subsidiaries—MGI US LLC and Complete Genomics, Inc.—with Swiss biotech firm Swiss Rockets AG, with the total transaction value reaching no less than $120 million.
On October 12, MGI disclosed the financial details and transaction structure in an official announcement. This marks China's first-ever license-out of life science instruments and the first global commercialization of a proprietary patented technology system in this sector from China.
The transaction not only represents a pioneering shift from product export to technology export but also establishes a new lightweight licensing model for medical device globalization—featuring milestone payments and a mechanism for generating returns from existing intellectual assets.
Under the licensing agreement, MGI has granted Swiss Rockets authorized access to its CoolMPS™ sequencing technology-related patents, trademarks, know-how, and software—through exclusive and non-exclusive licenses—for the research, development, manufacture, and commercialization of related products in markets outside the Asia-Pacific and Greater China regions.
To support the successful development and commercialization of end products, MGI will provide technical support for the transfer, product development, and commercialization of the CoolMPS™ technology. Swiss Rockets will supply CoolMPS™-based genetic sequencers and consumables to markets including the United States and Europe, with the first CoolMPS™-powered sequencers scheduled for launch in these regions as early as the first quarter of 2026. Additionally, Swiss Rockets will leverage CoolMPS™ sequencing technology to develop cost-effective solutions for disease prevention and health management, advancing the fields of precision medicine and longevity science.
1Pioneer Phase-based Payment Solution for Life Science Instrument Deal
DNBSEQ is the core technology of MGI, utilizing a combination of DNA nanoballs (DNB) and patterned array chips—an architecture consistently employed across all MGI sequencing platforms. StandardMPS™ and CoolMPS™ represent two distinct sequencing chemistry approaches that can be implemented on various DNBSEQ platforms.
The CoolMPS technology, which is the subject of this transaction, originated during a period when MGI was facing competitive pressure from international giants and seeking to expand its proprietary innovations. It was first introduced at the AGBT conference in 2019. At that time, MGI was engaged in patent litigation with Illumina, which initially hindered the U.S. launch of CoolMPS, leading to its formal commercial release in 2020. Following MGI's legal victory and the subsequent $325 million settlement, CoolMPS was not positioned to replace StandardMPS as the primary chemistry but rather serves as a complementary and differentiated sequencing solution, advancing in parallel with the established StandardMPS system.
As the first sequencing chemistry based on base-specific antibodies, CoolMPS™ employs unlabeled reversible terminators for polymerization and utilizes specific antibody recognition for base identification. This antibody-based approach eliminates the need for chemical modification of nucleotides, keeping the newly synthesized DNA strands in their natural state. It avoids interference from "scarring" caused by fluorescent label removal, thereby supporting efficient and stable continuous DNA polymerization. Furthermore, since a single antibody can conjugate with multiple fluorophores, the detection signal is significantly enhanced. This allows DNA nanoballs (DNBs) of the same density to produce stronger fluorescence and a higher signal-to-noise ratio, effectively improving sequencing accuracy and increasing read length.
In summary, CoolMPS™ delivers significantly longer reads with higher accuracy and lower error rates, making it suitable for applications requiring "long reads with high precision," such as HLA high-resolution typing, low-frequency mutation detection in FFPE samples, and long-fragment assembly. In contrast, StandardMPS™, which uses direct fluorescently labeled nucleotides, remains MGI's primary sales driver due to its lower cost, mature supply chain, and comprehensive platform compatibility. It is ideally suited for routine high-throughput scenarios like WGS, WES, and RNA-seq.

MGI Discloses CoolMPS Sequencing Data at AGBT 2020 Conference
The balanced development of these two technologies was disrupted in 2024. At the AGBT conference that year, MGI launched StandardMPS™ 2.0, which enables DNBSEQ platforms to achieve a sequencing data quality of Q40 > 85%. This significantly improves the accuracy and sensitivity of Whole Genome Sequencing (WGS) while reducing the required sequencing data volume. Currently, the T7+ system empowered by StandardMPS™ 2.0 can deliver over 14 Tb of high-quality, high-throughput data with Q40 > 80% within 24 hours. Potentially, StandardMPS™ 2.0 bridges the target customer markets of both StandardMPS™ and CoolMPS™, providing a more cost-effective solution for mid-to-high-end applications.
Simultaneously, CoolMPS™ has continued to advance. Concurrently, leveraging the CoolMPS™ sequencing biochemistry, MGI introduced the new mid-to-high-throughput sequencer DNBSEQ-G800. This platform achieves a sequencing accuracy of Q40 > 90% and supports continuous reads up to 600 bp, offering the longest read length among short-read sequencers and facilitating the decoding of challenging genomic regions. At the time of its release, MGI opened applications for early access in the U.S. market, collaborating with early users to explore the stability of Q50 sequencing quality. Consequently, the positioning of CoolMPS™ as a premium, internationally-focused technology for advanced applications and future capabilities has become increasingly clear.
The announcement indicated that the CoolMPS™ sequencing technology overlaps with the company's current core R&D focus on StandardMPS™, while also facing constraints from certification processes and technology transition costs. This clarifies the underlying rationale for MGI's licensing partnership—to activate the value of its existing technology, retain autonomy in core markets, and utilize a capital-light licensing model to enable high-end technology to "ride on others' ships" for global expansion cost-effectively.
This approach mirrors models that leverage shared core technologies and platforms to revitalize existing assets. By partnering with international firms that provide established channels, R&D capabilities, manufacturing facilities, and regulatory expertise, the licensed technology can advance more efficiently in global markets. Simultaneously, the Chinese innovator gains cash flow, equity investment, and potential milestone payments, royalties, and commercial profit-sharing (e.g., co-development/co-commercialization models). This model has been repeatedly validated in the overseas expansion of innovative Chinese pharmaceuticals. With this agreement, MGI has realized the first-ever overseas licensing case for a Chinese life science instrument company, elevating the transaction value to over US$100 million in the process.
Upon the official effectiveness of this licensing agreement, MGI will receive a one-time, non-refundable upfront payment of $20 million from Swiss Rockets. This substantial initial payment, representing approximately one-sixth of the total minimum transaction value, is particularly notable. If the agreement is fulfilled through the exclusive license term, MGI is expected to receive a total of no less than $120 million, comprising the upfront payment, milestone payments ($20 million), and tiered royalties based on a percentage of the net sales of the licensed products. This structure is projected to generate significant immediate profits and establish a long-term, stable revenue stream, substantially improving MGI's profitability and cash flow position.
2A New Pathway for Globalization Amid Uncertainty
Looking ahead, this licensing deal may create a chain reaction with the previous patent dispute with Illumina, collectively advancing MGI's global strategy and intensifying transformation in the global gene sequencing market.
In Illumina's home turf—the international market—escalating geopolitical volatility over the past two years has constrained MGI's overseas expansion. In 2024, sequencing revenue in Europe and Africa declined to RMB 344 million (year-on-year -6.24%) amid intensified competition and geopolitical conflicts. Due to postponed genomic projects in Middle Eastern countries, sequencing-related revenue in Asia Pacific fell to RMB 225 million (year-on-year -30.25%). Impacted by policies such as the "Biosafety Act," the Americas market recorded sequencing-related revenue of RMB 172 million (year-on-year +8.28%). Entering 2025, overseas revenues showed some recovery: January-September figures reached RMB 110 million in Asia Pacific, RMB 260 million in Europe-Africa, and RMB 140 million in the Americas.
In China, however, the core market continues to expand, and order conversion rates have significantly improved following Illumina's sanctions. A report from the National Center for Clinical Laboratories revealed that MGI's sequencing platforms surpassed Illumina for the first time in 2023 to become the most widely used platforms in Chinese laboratories. According to China Research P&H, Illumina's market share in China plummeted from 64.5% in 2021 to 54.2% in 2023, while MGI's share surged from 19.9% to 32.6% during the same period. By 2024, MGI's instrument installations reached 56.4%, achieving market leadership.
In the first half of 2025, MGI's sequencer shipments increased by 40% year-on-year, with the T7 series—"the world's highest daily throughput genetic sequencer"—accounting for 62% of shipments. Cumulative global instrument installations surpassed 5,300 units. Although revenue for the first half reached RMB 1.114 billion, reflecting a slight decrease of 7.9% year-on-year, the net loss attributable to shareholders narrowed to RMB 104 million, representing a 65.3% improvement. In the latest Q3 report, MGI's single-quarter revenue reached RMB 755 million, a 14.45% year-on-year increase, with profit turning positive—surging 110.98% year-on-year. For the first nine months, the net loss attributable to shareholders continued to narrow to RMB 120 million, reducing losses by 74% compared to the same period last year.
From a business model perspective, MGI's full-portfolio sequencer products and solutions operate on a classic "razor-blade" model: instruments (the razors) are sold at lower profit margins to secure customer accounts, while sustained profitability is achieved through recurring sales of high-margin consumables and reagents (the blades). This model demonstrates characteristic "installed base leverage."
In the Chinese market, MGI has transitioned from the "customer acquisition phase" to the "installed base monetization phase." In 2024, revenue from sequencing consumables reached RMB 1.373 billion (yoy +1.43%), while revenue from sequencing instruments amounted to RMB 960 million (yoy +6.92%).
The strength of the razor-blade model lies in creating application-specific barriers through the deep integration of instruments and consumables, securing a long-term, stable customer base and recurring revenue stream. As previously noted, CoolMPS™ sequencing targets Q50 quality standards, focuses on premium applications, and commands higher pricing and gross margins. Meanwhile, the StandardMPS™ 2.0 reagent kit, though priced higher than the 1.0 version, reduces the required data volume for equivalent applications, effectively lowering the cost per gigabase. This creates market opportunities for both "same-volume price upgrades" and "mid-to-high-end application substitution."
This agreement enables the commercialization of CoolMPS™ technology and products in overseas markets through Swiss Rockets, a fully foreign-owned entity, thereby mitigating potential avoidance from U.S. clients concerned about geopolitical tensions or Biosafety Act compliance issues. At a deeper strategic level, the partnership is expected to drive incremental growth in MGI's core "razor-blade" model by promoting premium consumable adoption—enhancing global recognition of MGI's technological capabilities and brand presence while advancing its globalization strategy through collaboration.
Concurrently, MGI retains all rights to CoolMPS™ in the Asia-Pacific and Greater China markets, directly competing with Illumina in its most dominant premium segment globally. Public data indicates Illumina maintains an installed base of over 3,000 instruments in China, with 2024 Greater China revenue reaching approximately RMB 2.2 billion, of which demand for ultra-high-throughput systems accounted for over 60%.
Leveraging Swiss Rockets' strong local resources and R&D capabilities will fully unlock the value of CoolMPS™ sequencing technology, creating synergies with MGI's established and commercially proven StandardMPS™ technology.
3The Cooperation Model
The licensing partner, Swiss Rockets AG, is a Basel-based innovative enterprise founded in 2018. It aims to function as a leading biotech startup incubator and accelerator, providing comprehensive support including funding, legal and regulatory guidance, as well as laboratory and manufacturing facilities. The organization brings together several specialized biotech companies with leading expertise in their respective fields. With subsidiaries in Switzerland, Serbia, and the United States, it maintains distinct local channel advantages. Oncology and infectious diseases represent its core R&D focus areas.
In March 2025, Emergent BioSolutions—a U.S. public health company—entered into an investment agreement with Swiss Rockets and established a strategic collaboration with Rocketvax AG, a vaccine company under Swiss Rockets. Emergent will lead the manufacturing and commercialization in the U.S. for four of Rocketvax's pipeline candidates targeting infectious diseases, cancer, and autoimmune disorders. One candidate has already secured a Letter of Intent with the U.S. National Institutes of Health (NIH).
Other portfolio companies include Torqur AG, which is developing next-generation dual PI3K/mTOR inhibitors for targeted cancer therapy, and Torpedo Therapeutics, a nuclear medicine innovator advancing Terbium-161 (¹⁶¹Tb)-based next-generation radioligand therapies.
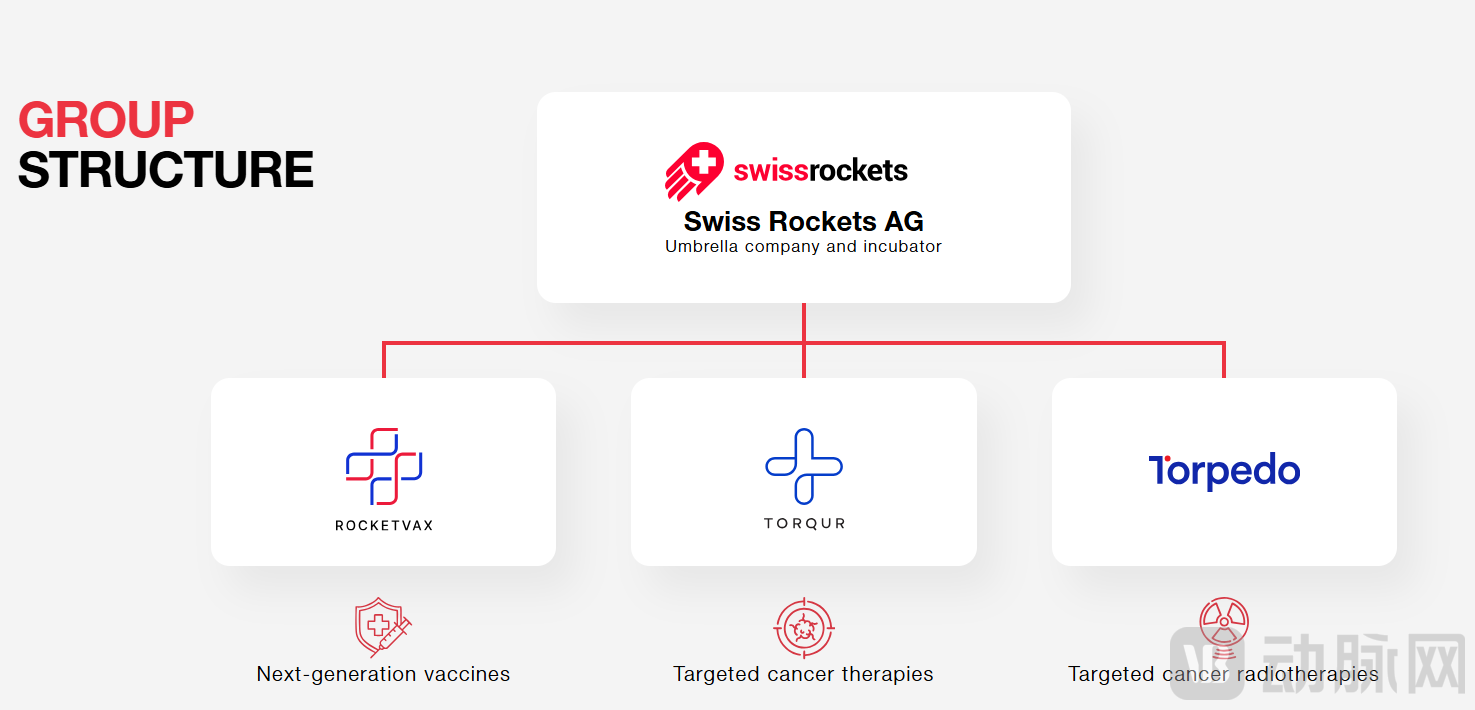
Current Group Structure of Swiss Rockets
According to Swiss Rockets' official news, this agreement will enable the company to focus future development on products utilizing CoolMPS™-based DNA barcoding technology, aiming to achieve faster, more accurate, and cost-effective whole genome sequencing. Dr. Vladimir Cmiljanovic, Founder, CEO, and Chairman of Swiss Rockets, emphasized that the collaboration will complement the group's portfolio of advanced precision therapy companies by providing economical, precise, and information-rich multi-omics testing for preventive healthcare and early diagnosis, as well as delivering effective treatment solutions in the fields of precision medicine and longevity science.
Notably, Swiss Rockets plans to launch CoolMPS™-powered sequencers in European and American markets as early as Q1 2026, targeting applications in whole genome sequencing, RNA sequencing, and microbiome analysis. Moving forward, it will be particularly interesting to observe how the "long-read, high-accuracy" CoolMPS™ technology synergizes with Swiss Rockets' innovative drug development pipelines, potentially enabling new collaborations and breakthroughs.
As early as 2023, Illumina made significant moves into drug development by partnering with Nashville Biosciences to unlock 250,000 clinically-linked whole genome sequences for pharmaceutical R&D, collaborating with Janssen Pharmaceutical on precision medicine and companion diagnostics, and launching a strategic partnership with AstraZeneca to accelerate drug target discovery using AI-powered genomic interpretation and analysis.
The true value lies within the data. As genome-wide association studies mature, vast repositories of multi-omics data and hundreds of thousands of genetic profiles await exploration. Leveraging genomic and genetic data to identify new targets and improve drug development success rates is becoming a reality. Ultimately, global sequencing giants are fundamentally focused on stimulating new sequencing demand, expanding downstream business models, and discovering sustainable long-term growth drivers.
The partnership between incubator group Swiss Rockets and MGI may potentially forge a new pathway in the global gene sequencing market.
