Leadinno Medical Valley closes nearly RMB 200M Series B to build full-stack neuromodulation platform
Beijing Leadinno Medical Valley Technology Development Co., Ltd. ("Leadinno Medical Valley"), a provider of innovative solutions for the neuromodulation and brain-computer interface (BCI) industry chain, announced the completion of nearly 200 million yuan in Series B financing. This round was co-led by Shunxi Fund and E-Town Capital, with follow-on investments from Galaxy Capital, Guangzhou Industry Investment Capital, and Yizhuang Investment. Genesis Partners has served as the company's long-term strategic advisor.
In recent years, Leadinno Medical Valley has maintained a steady annual financing rhythm. The proceeds from this round will be primarily allocated to three key areas:
Product Line Expansion: Building upon its wireless powered spinal cord stimulation (SCS) products, the company will rapidly extend its portfolio to include peripheral nerve stimulation (PNS), sacral nerve stimulation (SNS), and deep brain stimulation (DBS), forming a comprehensive wireless powered neuromodulation product matrix.
Production Capacity Upgrade: New production bases will be established in Beijing E-Town and Shaoxing Keqiao to meet the demands of registration clinical trials and future commercialization.
Talent and Team Development: The company will continue to strengthen its R&D, production, and technical support teams to build a top-tier, full-chain team in China.

As one of China's few platform-based, full-stack neuromodulation companies, Leadinno Medical Valley has consistently focused on two core technology platforms since its establishment in 2017: wireless power transfer and implantable chips.
"Neuromodulation products must not only be functional but also address patient comfort and portability for long-term implantation, requiring diverse product solutions," said Xu Tianrui, founder of Leadinno Medical Valley. He explained that spinal cord stimulation (SCS), peripheral nerve stimulation (PNS), and deep brain stimulation (DBS) have vastly different requirements for power transfer efficiency and wearable designs. Through seven years of R&D iteration, Leadinno Medical Valley has broken through the limitations of single power transfer pathways, achieving multiple distinct transfer methods. Beyond supporting conventional stimulation modes, the platform also enables high-frequency stimulation and burst stimulation modes, providing customized solutions for various indications and application scenarios—covering spinal cord stimulation (SCS), peripheral nerve stimulation (PNS), sacral nerve stimulation (SNS), and deep brain stimulation (DBS).
Traditional implantable pulse generators (IPGs) face challenges such as complex surgeries, frequent postoperative adverse events, limited battery life, and high costs—primarily due to the large size of implanted batteries. In contrast, Leadinno Medical Valley's wireless powered products utilize wireless energy transfer from an external device to the implant, completely eliminating the need for an implanted battery. This innovation enables simplified surgery (a single minimally invasive procedure requiring only 30-40 minutes), a compact and fully flexible implant (with a lead-like electrode form factor), eliminates battery life concerns through external powering, and reduces overall costs.

Leadinno Medical Valley Wireless-Powered Spinal Cord Stimulation System is the first wireless implantable SCS product produced in China.
"Small size, low power consumption, and high performance are the three core metrics for implantable neuromodulation chips," Xu Tianrui told VCBeat. After five years of meticulous development, Leadinno Medical Valley has accumulated substantial intellectual property and technical know-how in the field. Its self-developed, dedicated implantable chip platform achieves a miniaturized design of just 1×3 millimeters, meeting the requirements for minimally invasive implantation while offering exceptional low power consumption and high performance. The chip platform covers nearly all functions of traditional IPG products, supporting not only various stimulation modes such as low-frequency, high-frequency, burst, and Differential Target Multiplexing (DTM) but also exploring several different processing solutions for closed-loop stimulation.

These two platforms enable Leadinno Medical Valley to expand horizontally across multiple indications and application scenarios, while simultaneously advancing vertical integration for performance iteration and closed-loop management. Furthermore, the platforms are designed for seamless integration with brain-computer interface (BCI) technology.
SCS Clinical Feedback Positive, PNS Type Inspection Imminent
Leadinno Medical Valley's first wireless powered spinal cord stimulation (SCS) product initiated its registration clinical trial earlier this year. The study is being conducted across 15 clinical centers nationwide and plans to enroll 70 patients. Currently, more than half of the target enrollment has been completed. Preliminary results indicate that the product reduces surgery time from the 2-3 hours required for traditional IPG products to just 30-40 minutes, simplifies the surgical procedure, and delivers significant analgesic efficacy. Patient pain scores have decreased from severe to mild, with notable improvements in sleep quality and emotional well-being.
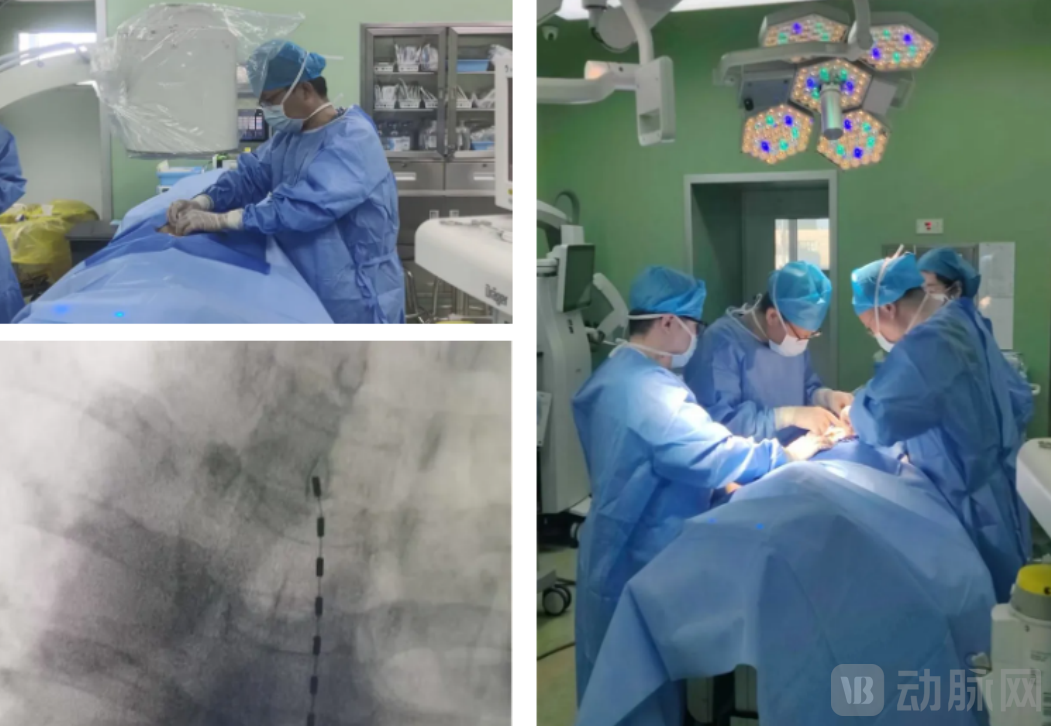
In April 30, 2025, a team led by Professor Fan Bifa from the Pain Department of China-Japan Friendship Hospital and the National Clinical Research Center for Pain Medicine successfully completed the first domestic clinical enrollment for the globally innovative "Implantable Wireless Spinal Cord Neuromodulation System." A patient suffering from refractory herpes zoster neuralgia experienced a reduction in pain score from 6 preoperatively to 1-2 postoperatively
"Globally, the PNS field remains a blue ocean. The form factor of traditional IPG products cannot meet the implantation requirements for PNS applications, making wireless powered products the future direction for PNS development," Xu Tianrui explained. Similar to the company's SCS product, its self-developed peripheral nerve stimulation (PNS) product also adopts wireless power transfer technology. The PNS product utilizes a new wireless energy transfer solution, featuring a smaller implanted electrode and a lighter external stimulator, better meeting patients' needs for daily and long-term wear.
It is reported that Leadinno Medical Valley's PNS product is expected to complete type testing shortly and is planned to enter the registration clinical trial phase next year.
Notably, Leadinno Medical Valley's SCS and PNS products demonstrate strong synergistic effects. The SCS product primarily targets moderate to severe chronic pain in the trunk region, while the PNS product is suitable for mild to moderate pain in the head, face, and limbs. By complementing each other, they provide a comprehensive solution for patients with chronic refractory pain, ensuring appropriate treatment options for patients across different disease stages, pain intensities, and affected body regions.
China's Neuromodulation Enters a Leap Window
During this critical window of technological advancement in the field of neuromodulation, Chinese companies are building a solid industrial foundation through complete local supply chains and rapid iteration capabilities. Meanwhile, the vast patient population provides abundant clinical samples for research, and the synergistic development of AI algorithms and brain-computer interface (BCI) technology is driving the industry toward breakthroughs in closed-loop intelligent systems. Against this backdrop, Leadinno Medical Valley has established a development path that starts from clinical needs → progresses through innovative design and supply chain R&D (with self-developed core components) → and aims to create globally Best-in-Class (BIC) products. The company's wireless power transfer and self-developed chip technology platform already enable seamless integration between neuromodulation and BCI products, paving the way for further clinical innovations.
Currently, the company has established collaborations with over 20 clinical centers led by the China-Japan Friendship Hospital and is partnering with research institutions such as Peking University and the Chinese Academy of Sciences to advance fundamental research in neuromodulation, forming a comprehensive neuromodulation-oriented solution. Simultaneously, it is actively working with brain-computer interface (BCI) enterprises to explore cutting-edge areas including consciousness disorder arousal, lower limb motor rehabilitation after spinal cord injury, and post-stroke rehabilitation. Leveraging its dual-platform advantages in wireless power transfer and self-developed chips, the company is building a product matrix that covers diverse indications and patient tiers while accelerating its global expansion.
