Steep declines in China's NIPT market: how BGI Genomics and Berry Genomics are navigating through global expansion

BGI
Scientific and Technological Service Provider and Precision Medical Service Operator

Berry Genomics
High-throughput Gene Sequencing Technology Developer
Chinese genetic sequencing companies are facing severe challenges.
Interim reports reveal that BGI Genomics recorded a revenue of 1.631 billion yuan in the first half of 2025 (H1 2025), down 12.82% year-on-year. Its net profit after deducting non-recurring gains and losses (net profit after non-recurring deductions) incurred a loss for the first time, plummeting by over 2,600% year-on-year. Within its business segments, the basic research and clinical application services for reproductive health generated 426 million yuan in revenue, accounting for approximately 26% of the total. However, this segment saw a year-on-year decline of 29.80%, which BGI Genomics attributed primarily to a 35% drop in revenue from its non-invasive prenatal testing (NIPT) business.
Another major player, Berry Genomics, also posted disappointing results for H1 2025, with revenue falling 18.91% year-on-year to 447 million yuan and the company reporting a net loss of 27.24 million yuan. Among its businesses, the revenue of medical testing services represented by NIPT was approximately 140 million yuan, accounting for about 31% of the total revenue, with a year-on-year decrease of nearly 30%. Although the operating costs decreased by 13.08% year-on-year, the gross profit also decreased by 10.93% year-on-year.
Amid intensifying competition within China's genetic testing industry, these two leading companies are navigating a painful transition. The sharp decline in their NIPT businesses has led to significant revenue fluctuations, transforming what was once a growth driver into a burden on their overall performance.
Thanks to their first-mover advantage, vast repositories of clinical data, and strong technological platforms, BGI Genomics and Berry Genomics collectively command nearly 70% of the Chinese market. The severe downturn in their core NIPT business will undoubtedly have a substantial impact on both companies, raising the critical question of how these industry leaders should adapt their strategies moving forward.
First, the continued decline in the number of newborns in China has had a direct and profound impact on the NIPT market.
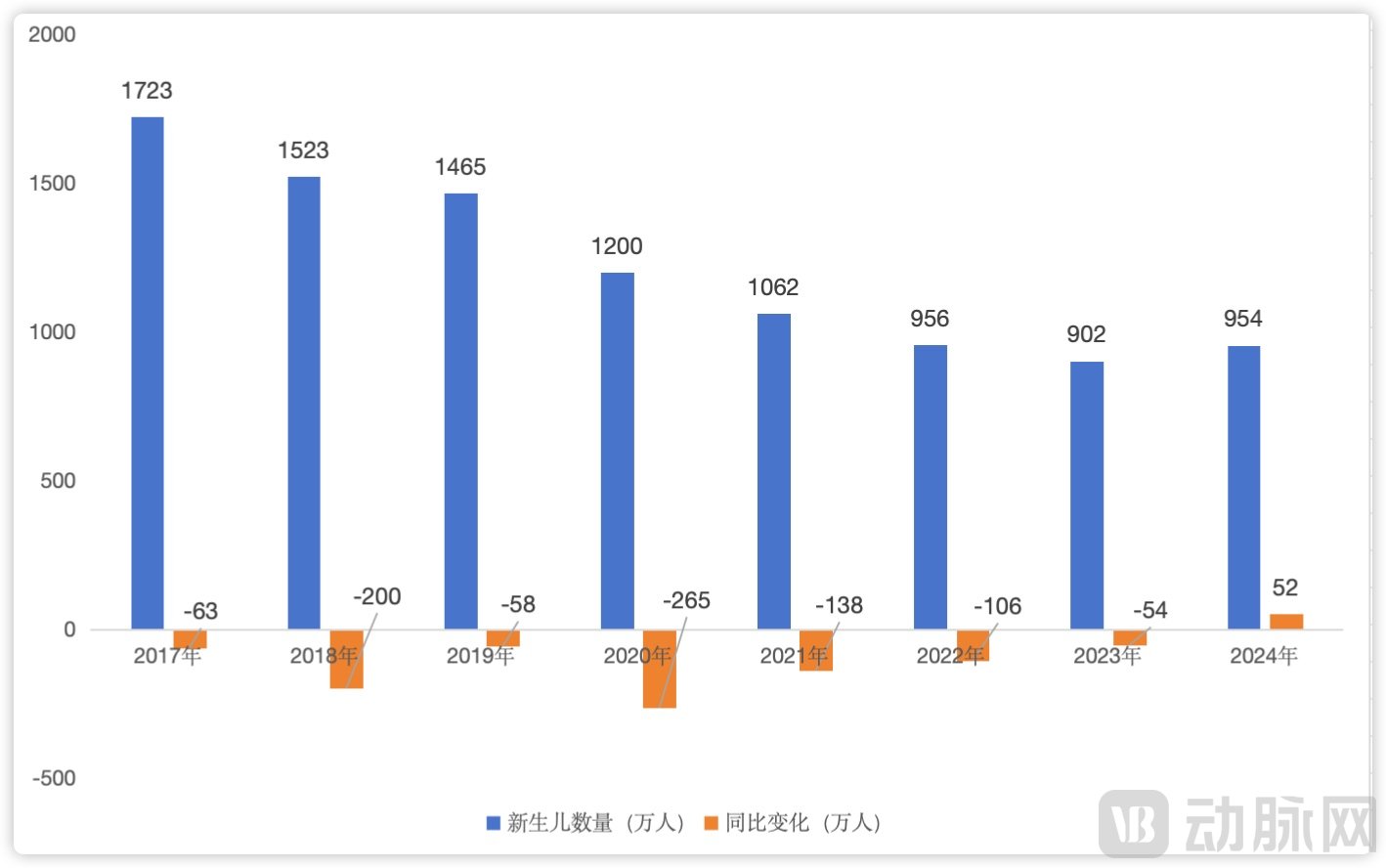
Number and changing trend of Chinese newborns in recent years, data from the National Bureau of Statistics
First, the continued decline in the number of newborns in China has had a direct and profound impact on the NIPT market. After peaking in 2016, China's newborn population experienced a steep, multi-year decline, with a cumulative decrease of approximately 8 million over seven years—a drop of more than 50%. This signifies a sustained and rapid contraction of the NIPT market's potential customer base. In particular, the number of newborns fell below the 10 million mark in 2022 and dropped further to a record low of 9.02 million in 2023. Although a rebound occurred in 2024, the overarching demographic shift has become irreversible. Consequently, the NIPT market has moved beyond its era of high growth and entered a new phase characterized by competition for existing customers, technological advancement, and business diversification.
Secondly, the current state of China's NIPT market is an inevitable outcome of a decade of commercial development and competition.
Viewed chronologically, the development of NIPT in China can be divided into four phases.
The first phase was the technology introduction period. NIPT entered clinical practice around 2010, gaining recognition for its high accuracy and non-invasive advantages. During this stage, the price of NIPT was several thousand yuan.
This was followed by the second phase of rapid expansion. With the removal of the pilot program in 2016—meaning all medical institutions with prenatal diagnosis qualifications could offer this service—the market expanded rapidly. Although the testing price generally remained above one thousand yuan during this period, some regions began exploring ways to reduce the out-of-pocket cost for pregnant women through subsidies or other measures.
The third phase saw local governments actively promoting the adoption of NIPT. Policies were successively implemented in regions such as Beijing, Hebei, and Tianjin, leading to a notable downward trend in end-user pricing. With an increasing number of market participants and intensified price controls, industry competition escalated, and NIPT profit margins began to compress. According to BGI Genomics' financial reports, the gross profit margin of its reproductive health business, represented by NIPT, declined from nearly 80% in 2018 to slightly over 40% in 2023.
In the fourth phase, in July 2024, the Jiangsu Provincial Medical Insurance Bureau released a volume-based procurement plan for NIPT. The plan set the maximum valid bid price at 345 yuan and stipulated that companies quoting no higher than 275 yuan would be directly selected. Compared to previous prices, this represented a drop of over 70%, fundamentally reshaping the industry's pricing logic and profit model. Consequently, the NIPT market has transitioned completely from a past "blue ocean" into a "red ocean" competitive stage, where competition is now centered on cost control and economies of scale.
Looking ahead, companies must navigate the challenges posed by steep price declines and identify new pathways for growth.
With their NIPT businesses under pressure, companies have turned their attention to overseas markets, albeit with distinct strategies. Taking BGI Genomics as an example, the company has chosen to deeply adapt and intensively operate based on the public health needs of different regions.
In Southeast Asia and the Middle East, BGI Genomics has prioritized public welfare projects as its entry point. Through its collaboration with Thailand's Ministry of Public Health, BGI helped advance the inclusion of Non-Invasive Prenatal Testing (NIPT) within the coverage of the government health insurance scheme. The partnership also provides hereditary breast and ovarian cancer screening services covered by the public health insurance. Furthermore, by partnering with multiple hospitals to establish in-house genetic testing laboratories, BGI has successfully implemented its integrated precision medicine solutions directly within healthcare facilities. Building on the experience gained in Thailand, BGI is now promoting related projects in countries like Indonesia and Vietnam. It has also supported the development of local precision medicine and public health services in the United Arab Emirates, India, Vietnam, and Pakistan.
Furthermore, BGI Genomics' joint venture in Saudi Arabia, Genalive, has obtained certifications from the Saudi Ministry of Health and the Central Accreditation Committee for Healthcare Institutions. Capitalizing on the rapidly growing local demand for high-throughput sequencing-based services, BGI is applying its technological expertise to improve the detection, diagnosis, and treatment efficiency for genetic diseases and cancers in the region. This effort not only advances precision medicine and public health in Saudi Arabia but also allows BGI to secure a vital strategic position in the local healthcare ecosystem.
In a parallel development, another BGI joint venture, Borneo Genomics Innovation Sdn Bhd in Brunei, has signed a Memorandum of Understanding (MoU) with the Brunei Ministry of Health for a national cervical cancer screening program. This collaboration, focused on HPV screening, is designed to support the modernization of the country's healthcare system.
The next phase of its public health engagement is technology transfer, an initiative BGI Genomics has already launched in Latin American and Central Asian markets.
In Uruguay, the company established its first regional laboratory in Latin America. This facility also marks the first overseas implementation of BGI's "Fire Eye" air-film structure solution for clinical molecular testing. The laboratory provides end-to-end high-throughput sequencing (NGS) services, covering the entire workflow from sample reception, extraction, and library preparation to hybridization capture, quality control (QC), sequencing, and through to bioinformatics analysis and report issuance. This transfer of technology is poised to enhance the prevention and control levels of diseases, including colorectal cancer—the most common cancer in Uruguay—while also strengthening the overall disease screening capacity across the Latin American region.
Similarly, the Genscreen laboratory, established in Uzbekistan through its collaboration, stands as one of the country's first clinical labs equipped with both high-throughput sequencing (NGS) and mass spectrometry capabilities. Currently deployed for non-invasive prenatal genetic testing and neonatal genetic metabolic disease screening, it has successfully expanded the scope of screened conditions in the local newborn population from just 2 to 48 distinct disorders. An equivalent model of collaboration has been replicated in Kazakhstan with the establishment of the Astana Gene Center.
It is noteworthy that BGI Genomics' strategy of establishing precision medicine laboratories, building joint labs, and forming joint ventures in key overseas countries for technology transfer does more than just promote the application of genetic technology in foreign markets. It has also successfully facilitated the "going global" of Chinese technology, expertise, and standards. While effectively enhancing the capabilities of host countries in areas like birth defect control, precision oncology, and the prevention and management of infectious diseases, this approach has also successfully driven the international promotion and application of its own BGI-CSP Standard certification and evaluation system.
In the first half of the year, BGI Genomics' precision medicine testing business generated revenue of 696 million yuan, a year-on-year increase of 4.80%. This growth was primarily attributable to the continuous expansion in overseas markets, with revenue from key regions like Southeast Asia maintaining steady growth. Simultaneously, business development in emerging markets such as Latin America and South Asia yielded significant results, achieving a doubling in revenue.
BGI Genomics stated in its financial report that it has now completed over 100 technology transfer projects across 36 countries. The company has established collaborations with laboratories in numerous countries and regions, including Croatia, Kazakhstan, Thailand, Uruguay, Vietnam, India, the UAE, Spain, Mexico, Indonesia, Malaysia, Oman, Turkey, Colombia, Singapore, Saudi Arabia, Romania, Rwanda, and South Korea. Future initiatives will focus on projects such as NIFTY®, clinical exome sequencing, carrier screening, Colotect® for colorectal cancer screening, hereditary tumors, and HPV testing.
Berry Genomics has adopted a distinct approach to its global expansion, strategically targeting markets with clear policy directives and strong demand.
A substantive step in its overseas strategy was the establishment of a joint venture with a subsidiary of Saudi Arabia's Ajlan & Bros Holding. This move aligned with Saudi Arabia's "Vision 2030," where the transformation of the healthcare sector is a key component. The vision creates substantial demand for improved healthcare coverage, quality and efficiency of services, as well as digital and intelligent infrastructure.
Berry Genomics is leveraging the joint venture model to introduce its NIPT and other genetic testing products into the Saudi and broader Middle Eastern markets. This initiative aims to provide precise diagnostic and therapeutic solutions for genetic diseases, thereby enhancing and elevating the standard of local healthcare services. The Saudi partner has explicitly acknowledged that Berry's technological and R&D capabilities in specialized fields such as gene sequencing and clinical applications were key factors in the collaboration. Looking ahead, the two parties plan to progressively launch follow-up projects, including the establishment of local genetic testing laboratories.
In Southeast Asia, one of the global regions with the highest incidence of thalassemia, Thailand serves as a pertinent example. According to reports from the Thai Ministry of Public Health, approximately 18 to 24 million people carry the thalassemia gene, representing 30-40% of the total population. In response, the Ministry has implemented a comprehensive thalassemia policy encompassing screening, diagnosis, and treatment.
In the first half of 2025, Berry Genomics obtained CE marking in the European Union for three of its reagent kits: the Human Exome Sequencing Library Preparation Kit (Reversible Terminator Sequencing) for WES, the Fetal Cell-free DNA Library Preparation Kit (Reversible Terminator Sequencing) for NIPT, and the Thalassemia Gene Detection Kit (Single-Molecule Sequencing), CATSA. This certification provides a gateway for expansion into European and Southeast Asian markets. While traditional thalassemia testing methods are associated with relatively high rates of missed and false detection, Berry's third-generation sequencing solution effectively enhances screening efficiency and accuracy. Given the significant need in markets like Thailand, the product's potential is considerable.
Furthermore, products like Whole Exome Sequencing (WES), which command a price of approximately $1,800-$3,600 in the European market—over three times the price in China—present a significant opportunity to leverage technological advantage for arbitrage in overseas markets.
In the first half of 2025, Berry Genomics reported overseas revenue of 64 million yuan, contributing 14.34% of its total revenue, a notable increase from 12% during the same period last year.
Compared to BGI Genomics' "ecosystem" approach of establishing its own platforms and covering the entire industry chain for its global expansion, Berry Genomics has adopted a more focused and flexible model. Instead of pursuing end-to-end control from the technological source to the market front, as BGI does, Berry leverages its technological strengths in specific niches. It aims for targeted breakthroughs and deep collaboration in carefully selected segments. Building on its core advantage in reproductive health, the company is intensively exploring key emerging markets overseas, relying on continuous technological iteration to support its long-term international development. Simultaneously, after consolidating its position in the NIPT market, it is progressively introducing a broader portfolio of genetic disease testing products into suitable overseas markets.
While strengthening their global operations, both companies have also prioritized accelerating internal product iteration, with both choosing to integrate AI assistance.
In August, Berry Genomics launched the GENOisi™ intelligent agent, aiming to empower county-level obstetric and pediatric screening with AI. This initiative is designed to establish a three-tiered linkage model of "primary screening, regional diagnosis and treatment, and national-level research," ultimately building a comprehensive health management system that spans the entire human life cycle.
Currently, Berry Genomics is implementing a project in collaboration with Beijing Children's Hospital and its affiliated medical consortium. This partnership aims to establish a replicable and scalable new model for regional child health management, with related revenue expected to begin contributing in the fourth quarter of 2025. The collaboration primarily encompasses two areas: genetic testing services for neonatal and childhood genetic diseases, and the application of artificial intelligence.
In September 2025, BGI unveiled a comprehensive portfolio spanning upstream core tools, midstream precision health management, and downstream consumer-grade products. The release included the ultra-high-throughput sequencer DNBSEQ-T7+ and multiple health testing products targeted directly at consumers.
Furthermore, BGI has expanded its operations into two major new sectors: public health and agriculture & food. For instance, its "hour-level" rapid pathogen identification solution and the data-driven BGI Zhi Hui Medical System are advancing the development of intelligent, highly responsive public health infrastructure. Concurrently, the company is implementing a strategy to deeply integrate biotechnology into modern agriculture.
In summary, following the significant downturn in their NIPT businesses, both BGI Genomics and Berry Genomics have adopted proactive strategies aimed at driving growth through aggressive overseas expansion, while simultaneously broadening their scope into wider precision medicine fields.
The key to their future success hinges on several critical factors: the seamless transition between legacy domestic operations and new international growth drivers; the effective outward transfer of their technologies and standards; and the ability to establish and maintain a sustainable technological gap advantage within the intensely competitive global market.
Ultimately, the distinct core strengths of each company—BGI Genomics' scaled ecosystem and Berry Genomics' specialized technological expertise—are poised to play decisive roles in shaping their respective future trajectories.
