Genrix Bio and CMS strike RMB 510M licensing deal for two Class 1 innovative biologics

GENRIX BIO
Developer of Novel Monoclonal Antibody Drugs

CMS
Professional Drug Developer

Rxilient Medical
Dental Tool Manufacturer and Wholesaler
On September 22, 2025, innovative biopharmaceutical company Chongqing Genrix Biopharmaceutical Co., Ltd. ("Genrix Bio") announced that it has entered into separate exclusive commercialization collaboration agreements with Tibet Kangzhe Pharmaceutical Development Co. (“Tibet Kangzhe”) and RXILIENT MEDICAL PTE. LTD, the subsidiaries of China Medical System Holdings Limited ("CMS") . The collaboration assets include two investigational monoclonal antibody products: Vecantoxatug Injection (GR2001, "Vecantoxatug") and Silevimig Injection (GR1801, "Silevimig").
Under the agreements, CMS has obtained exclusive commercialization rights for GR2001 (Vecantoxatug) and GR1801 (Silevimig) in mainland China, while RXILIENT has secured the relevant rights for the Asia-Pacific (excluding mainland China), Middle East, and North Africa markets. Genrix Bio is set to receive upfront and potential milestone payments totaling approximately RMB 510 million from these collaborations.
About Genrix Bio
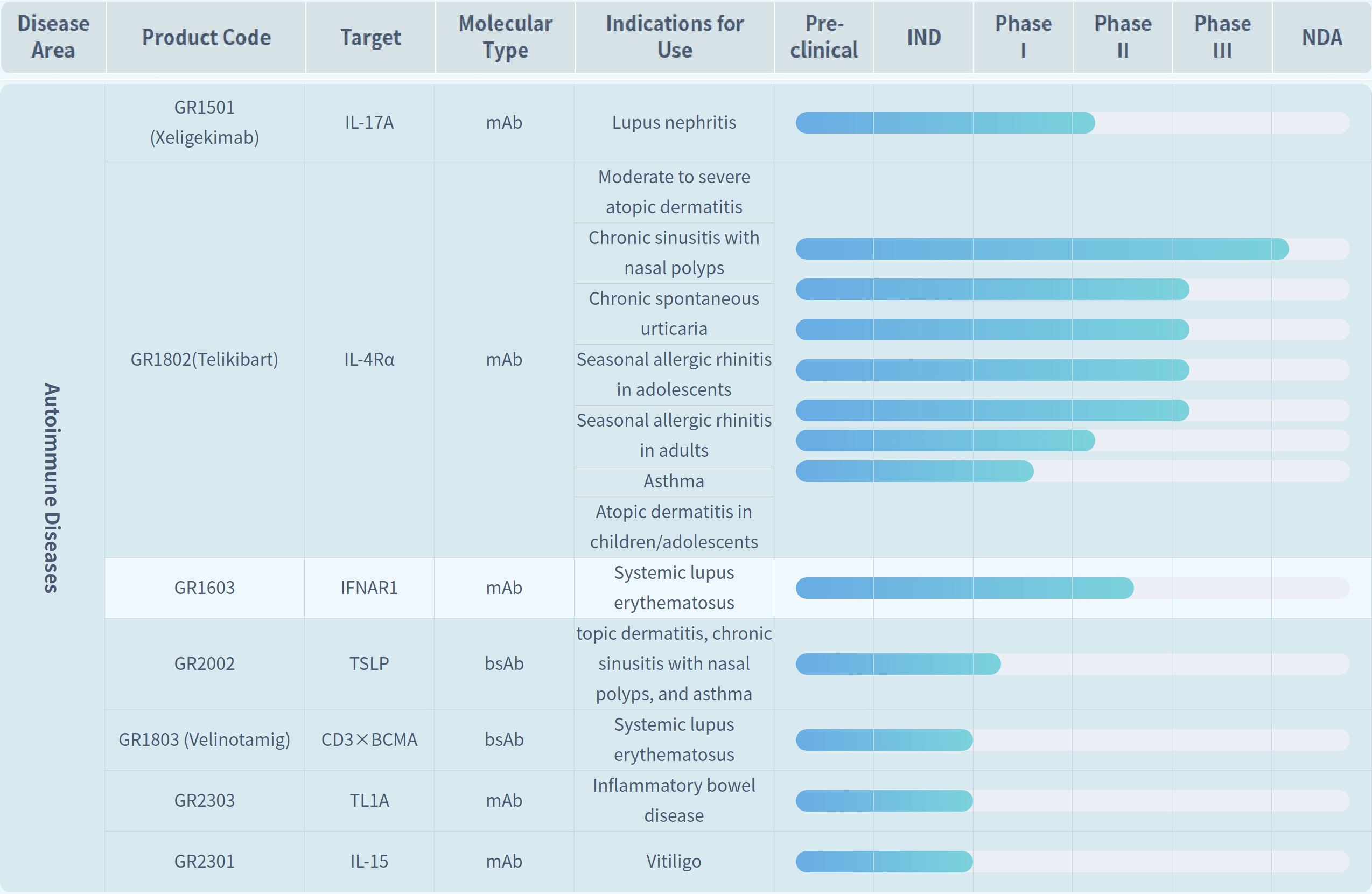
Founded in 2015, Genrix Bio is an innovative biopharmaceutical company driven by antibody drug discovery technology to address significant clinical needs. The company focuses its R&D efforts on three major therapeutic areas: autoimmune diseases, infectious diseases, and oncology. Concurrently, it has built capabilities in process development, quality systems, and industrial-scale manufacturing to enhance the efficiency of translating R&D achievements into tangible outcomes.
Source: Genrix Bio
In the autoimmune disease area, GR1501 (Xeligekimab) is a Class 1 therapeutic biological product, a monoclonal antibody injection targeting IL-17A. It was approved in August 2024 for the treatment of moderate to severe plaque psoriasis, and subsequently received approval in January 2025 for the treatment of ankylosing spondylitis (radiographic axial spondyloarthritis). Another core asset, GR1802 (Telikibart), targets allergic diseases and has currently advanced to the NDA (New Drug Application) stage for its lead indication.
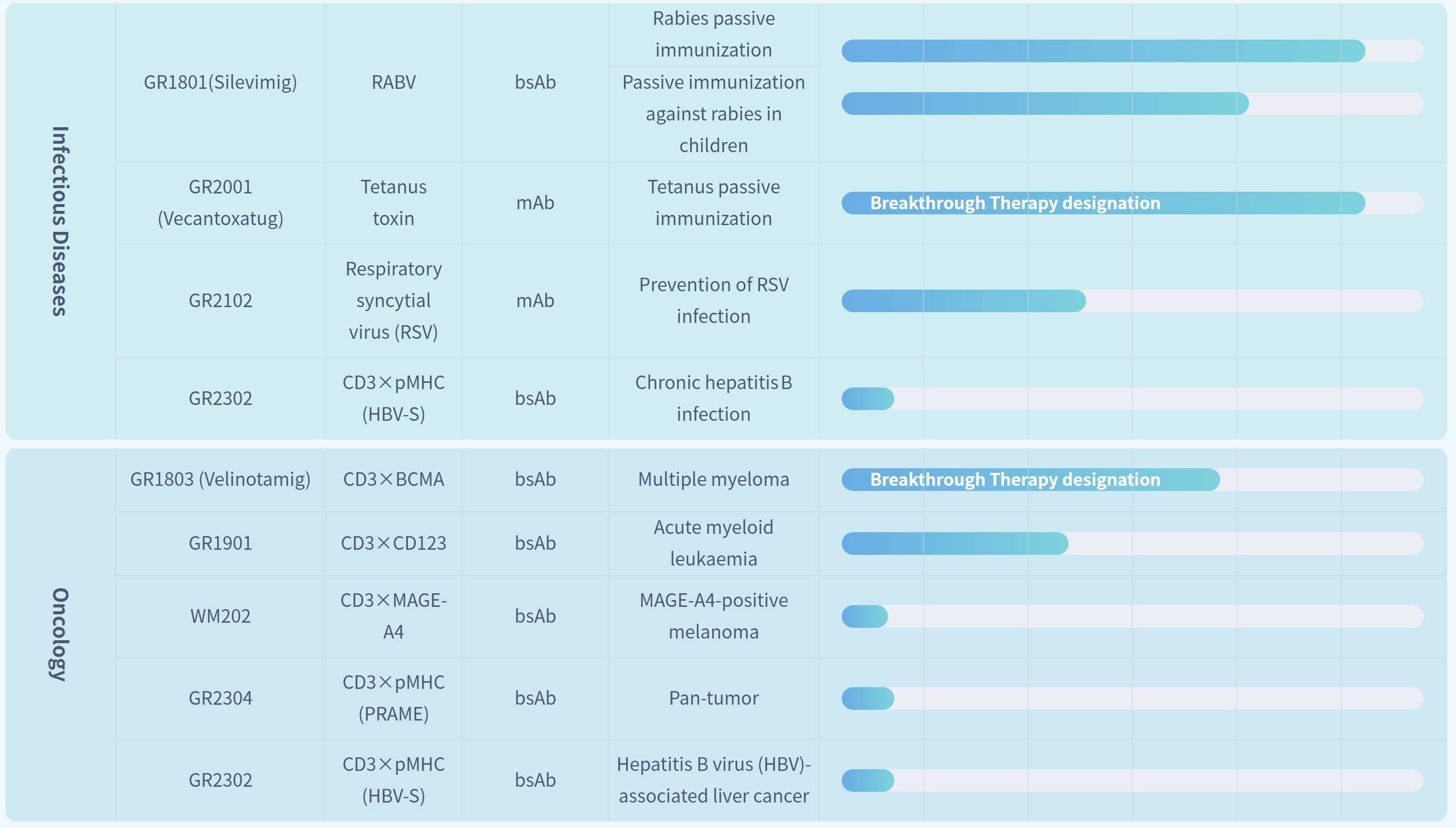
In the infectious diseases area, GR1801 (Silevimig) and GR2001 (Vecantoxatug)—the key assets that Genrix Bio has focused on developing—are exactly the core products of the RMB 510 million exclusive licensing collaboration with CMS. Both products are developed with the goal of replacing traditional human immunoglobulin, and hold potential advantages in large-scale production, batch consistency, and blood source safety.
Among them, GR1801 features a recombinant fully human bispecific antibody design, targeting multiple epitopes of the Rabies Virus (RABV) G protein, and its New Drug Application (NDA) has been accepted by the Center for Drug Evaluation (CDE) of the National Medical Products Administration (NMPA). GR2001, on the other hand, is a recombinant humanized monoclonal antibody targeting Tetanus Neurotoxin (TeNT); it has not only been included in China’s "Breakthrough Therapy" designation list, but also had its NDA submitted. The technological innovation and clinical progress of these two products serve as important cornerstones for this strategic collaboration.
In the oncology area, Genrix Bio has actively deployed its pipeline based on its bispecific antibody technology platform. Among these pipeline assets, GR1803 has obtained Breakthrough Therapy designation, while some other pipeline candidates have entered the preclinical research stage.
About CMS
CHINA MEDICAL SYSTEM HOLDINGS LIMITEDAs a pharmaceutical company with outstanding commercialization capabilities in the Hong Kong stock market,Build a diversified business portfolio spanning antibiotics, vaccines, and biologics.As of the end of 2024,Its innovative product pipeline has nearly 40 candidates, of which about 10 are in clinical stages, and 5 innovative drugs have been successfully approved for marketing in China.With a sales network covering 30 provinces, autonomous regions, and municipalities across China, as well as a professional promotion team of over 2,000 people, CHINA MEDICAL SYSTEM HOLDINGS LIMITED has formed a significant advantage in the field of innovative drug commercialization, particularly excelling in converting early-stage R&D achievements into market value.
Notably, CHINA MEDICAL SYSTEM HOLDINGS LIMITED has also obtained the commercialization rights for the product in the Asia-Pacific, Middle East, and North Africa regions through its subsidiary RXILIENT. Leveraging its accumulated registration experience and localized operational capabilities in emerging markets, the company not only achieves a global layout for the product but also maximizes the release of innovative value through a division of labor model where "the R&D side focuses on innovation and the commercialization side concentrates on implementation," allowing both parties to share risks.
Accelerating Global Expansion
This collaboration is not merely a case of "licensing for funds," but rather the result of the overlap between industry context and corporate strategic choices.
First, from the perspective of industry context, the passive immunity field has long relied on human immunoglobulins (such as human tetanus immunoglobulin (HTIG) for tetanus and rabies immunoglobulin (RIG) for rabies). However, their limitations are obvious: reliance on donated resources, potential blood source safety risks, batch-to-batch variations, and pressures from costs and cold chain logistics, all of which have led to a long-standing issue of insufficient supply. Against this backdrop, recombinant monoclonal antibodies or bispecific antibodies are regarded as an urgent innovative approach to fill this gap. Genrix Bio’s GR2001 and GR1801 are representative products that target and address this gap.
Second, geographical differences in market demand also serve as a driver for this licensing deal. Tetanus and rabies remain prominent public health challenges in China, the Asia-Pacific region, the Middle East, North Africa, and other areas. The limited supply of traditional preparations has made recombinant antibodies—with their standardized and scalable production—an urgent need. Under the terms of this transaction, CMS has exclusive responsibility for the Chinese mainland market, while RXILIENT is tasked with advancing the products in the Asia-Pacific (excluding the Chinese mainland), the Middle East, and North Africa. This arrangement is precisely a regional licensing strategy based on differences in demand and distribution channels across various regions.
Finally, Genrix Bio's R&D capabilities and commercialization readiness have improved significantly. In the first half of 2024, its R&D investment reached RMB 283.5 million, and R&D personnel accounted for approximately 65% of its total staff—both figures highlight its R&D-driven strategic orientation. The clinical data of its core investigational product GR1501 (Xileqikimab) is impressive: in the plaque psoriasis trial, the PASI75 response rate reached 90.7% at Week 12 (vs. 8.6% in the placebo control group), and the PGA 0-1 response rate hit 74.4% (vs. 3.6% in the placebo control group). These results fully validate the company's R&D strength and the clinical value of the product.
At the same time, CMS has rich experience in innovative drug in-licensing and biological product commercialization, with extensive coverage and fast market penetration capabilities. It is expected to accelerate the translation of GR2001 and GR1801 from regulatory registration to clinical application.
Of course, the licensing journey is not without challenges. Risks in regulatory review, consistency of manufacturing processes, difficulties in cold chain logistics, product pricing and medical insurance access, as well as differences across regional markets, will all test the ultimate outcome of the products' commercialization.
This RMB 510 million licensing collaboration represents not only a crucial leap forward in the commercialization of Genrix Bio's R&D achievements, but also a key strategic move by CMS in the infectious diseases field. It is expected that these two products will successfully overcome commercialization hurdles, and while addressing unmet clinical needs, they will also set an example for more innovative projects to explore a balance between commercial value and social value.
