WYTD Medical’s ConSpeed® approved in China, the world’s first bubble study-based automated contrast injection system
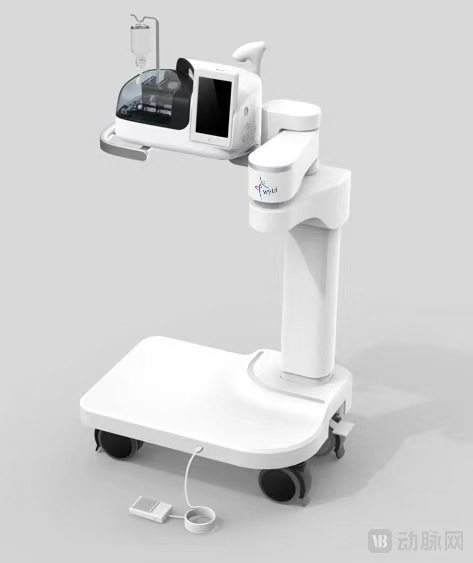
Recently, the ConSpeed® Automated Ultrasound Contrast Injection System, independently developed by WYTD Medical Technology (Shenzhen) Co., Ltd. ("WYTD Medical"), has officially obtained the Medical Device Registration Certificate (Guangdong Medical Device No. 20252061271) in China. The successful approval of this product marks the official market launch of the world's first automated ultrasound contrast injection system. It not only promotes the automation, standardization, and normalization of patent foramen ovale (PFO) diagnosis but also lays the foundation for the development of intelligent and precise diagnosis and treatment in the field of structural heart disease.
As the world's first automated ultrasound contrast injection system developed specifically for bubble studies (agitated saline contrast studies), ConSpeed® has been designed with full consideration of clinical application scenarios. Its intended use is to be used with a disposable mask and disposable contrast injection syringe and accessories to prepare and inject mixed (activated) saline solution during bubble studies for transthoracic echocardiography or contrast-enhanced transcranial Doppler ultrasound. Additionally, the system features a built-in blood pressure monitoring module that can track patient pulse and non-invasive blood pressure in real time, providing dual safety assurance throughout the examination procedure.
The ConSpeed® system consists of a main unit, a touchscreen, and a blowing handle. It can be paired with optional accessories such as a cart, footswitch, and blood pressure cuffs (for adult/pediatric use) to meet the needs of various clinical scenarios. The product is currently available in two models, BS-1 and BS-2, which offer flexible operation and accommodate the usage habits and functional expansion requirements of different departments.
Patent foramen ovale (PFO) is a common congenital heart condition, with a prevalence of approximately 25% in the adult population. Research indicates that PFO is closely associated with various disorders, including cryptogenic stroke, migraine with aura, paradoxical embolism, decompression sickness, and platypnea-orthodeoxia syndrome. Therefore, accurate diagnosis of PFO is clinically significant for reducing potential patient risks.
Among existing diagnostic methods, the bubble study is a common technique used to screen for right-to-left shunt (RLS). However, it has several limitations. Primarily, it relies on manual agitation to mix air, blood, and saline, which leads to inconsistent bubble formation, unstable test results, high operator dependency, poor reproducibility, and uncontrolled microbubble size. Additionally, the test requires the patient to perform a Valsalva maneuver. Since most patients have difficulty performing this maneuver correctly or standardizing it, the effectiveness of the examination and the interpretation of results are often compromised, further limiting the standardization and precision of PFO diagnosis.
ConSpeed® achieves fully automated, uniform mixing and injection of the contrast solution by precisely controlling the aspiration volume, mixing ratio, injection speed, and number of agitations. This ensures the generated microbubbles are consistent in size and stable in distribution, effectively reducing the risk of false-negative or false-positive results and substantially improving test accuracy.
The fully automated process eliminates the subjective errors associated with manual bubble agitation, ensuring highly consistent results across different patients and testing sessions. This provides clinicians with stable, comparable diagnostic data.
ConSpeed® replaces the traditional Valsalva maneuver with an innovative breath-triggered mechanism, requiring only gentle exhalation to complete the test. This design not only significantly reduces reliance on patient cooperation and avoids result deviations caused by improper Valsalva performance but also enhances the controllability and repeatability of the examination, delivering more stable and reliable data for clinical decision-making.
The fully automated design of ConSpeed® significantly streamlines the testing workflow, allowing a single healthcare professional to perform the entire procedure independently. This effectively reduces labor costs, alleviates manual workload, and markedly improves overall testing efficiency.
The approval of the ConSpeed® Automated Ultrasound Contrast Injection System marks a significant milestone not only in WYTD Medical's innovation journey but also signifies the official entry of PFO diagnosis into an era of automation, standardization, and normalization. In the future, the system is poised to play a vital role in key clinical stages such as precise pre-operative screening and post-operative efficacy evaluation, propelling the diagnosis and treatment of structural heart disease into a new era characterized by greater intelligence and high-quality development.
Company Profile
WYTD Medical Technology (Shenzhen) Co., Ltd. established on June 17, 2021, is located in the Shenzhen Biomedical Innovation Industrial Park. It is an innovative enterprise focusing on the field of interventional treatment of heart disease, especially structural heart disease. The company has a top team of experts to; Maximize the quality of life of patients and improve the last firewall of life. For the mission, in line with "Exploiting, innovating, surpassing and win-win." The company is committed to developing into the leader of the most professional medical devices in cardiac science.
