HighTide unveils Phase III T2DM results for its first-in-class drug at EASD 2025, targeting multibillion-dollar CKM market
On September 16, at the 61st EASD Annual Meeting in Vienna, Austria, Professor Linong Ji from Peking University People's Hospital presented that HTD1801 met the primary efficacy endpoint in its Phase III clinical trial for type 2 diabetes (T2DM), with additional cardiorenal and metabolic benefits observed.
The SYMPHONY 2 (NCT06353347) trial is a randomized, double-blind, placebo-controlled Phase III study conducted across 61 clinical sites in China. It was designed to evaluate the efficacy and safety of HTD1801 in adult patients with T2DM who had inadequate glycemic control despite metformin therapy.
Regarding the primary efficacy endpoint, after 24 weeks of treatment with HTD1801 in combination with metformin, the mean change in glycated hemoglobin (HbA1c) from baseline was -1.2%, which was significantly superior to the placebo group. Notably, in the severe patient population with high baseline HbA1c (≥8.5%), the mean change in HbA1c from baseline reached -1.6% in the HTD1801 group.
Simultaneously, HTD1801 met multiple secondary efficacy endpoints, including improvements in fasting and postprandial blood glucose, lipid parameters, as well as markers of inflammation and oxidative stress, indicating its potential for comprehensive management of multiple metabolic abnormalities. In patients with mild renal impairment, the HTD1801 group demonstrated an increase in estimated glomerular filtration rate (eGFR, a key indicator of renal function), suggesting a potential renoprotective effect. Furthermore, across the overall study population, the HTD1801 group showed significant reductions in lipid parameters such as LDL-C, along with markers of inflammation and oxidative stress, indicating potential cardiovascular benefits.
Indeed, the concepts of "one drug, multiple effects" and "treating different diseases with the same approach" are increasingly becoming clinical trends in the management of metabolic-related comorbidities such as type 2 diabetes (T2DM). Chronic hyperglycemia and other metabolic abnormalities (e.g., obesity, dyslipidemia, hypertension) in T2DM patients ultimately lead to damage in various organ systems, resulting in both microvascular and macrovascular injuries, and eventually causing life-threatening complications.
HTD1801, a novel molecular entity derived from traditional natural ingredients, employs a globally unique dual-target synergistic mechanism to achieve concurrent management of both metabolism and inflammation. As an innovative metabolic drug, it is poised to deliver a cutting-edge treatment option for cardiovascular-kidney-metabolic (CKM) system diseases.
1Improved Glycemic Control and Insulin Sensitivity
According to the prospectus of HighTide Therapeutics, HTD1801 was developed based on the traditional natural products berberine and ursodeoxycholic acid. Through the construction of a unique microstructure, it forms a globally first-in-class new molecular entity (NME). As an oral anti-inflammatory and metabolic modulator, HTD1801 possesses a novel and unique dual-target synergistic mechanism. It exerts its biological activity by activating AMPK and inhibiting the NLRP3 inflammasome, targeting the core mechanism of insulin resistance and intervening from both metabolic dysfunction and inflammation pathways.
Activating the AMPK Pathway and Regulate Energy Homeostasis
AMPK (AMP-activated protein kinase) is a key enzyme that regulates cellular energy homeostasis. By activating AMPK, HTD1801 improves insulin sensitivity, promotes fatty acid oxidation, inhibits lipotoxicity, enhances mitochondrial function, and strengthens cellular capacity to cope with metabolic stress. These actions collectively alleviate oxidative stress and renal tubular injury, thereby effectively ameliorating metabolic diseases and their associated multi-organ dysfunction.
Inhibiting the NLRP3 Inflammasome and Alleviate Chronic Systemic Inflammation
The NLRP3 inflammasome is a critical regulator of inflammatory responses, capable of triggering multi-level inflammatory cascades and pyroptosis. It is closely associated with glucose metabolism disorders, insulin resistance, and endothelial dysfunction. By inhibiting the activation of the NLRP3 inflammasome, HTD1801 reduces the release of inflammatory cytokines such as IL-1β and IL-18, thereby blocking inflammation-driven metabolic dysregulation. This mechanism improves insulin sensitivity and metabolic function, while also exerting anti-fibrotic effects in key organs including the liver, kidneys, and lungs. Consequently, HTD1801 mitigates the multi-organ damage caused by systemic chronic inflammation.
Among current clinical-stage investigational drugs, HTD1801 is the only dual-targeted product capable of simultaneously modulating both AMPK and NLRP3. As a result, the newly released data demonstrate that HTD1801 provides multi-faceted metabolic benefits, including improvements in insulin resistance, glycemic control, lipid metabolism, and inflammatory markers.
Regarding glucose-lowering efficacy, in the SYMPHONY 2 trial (in combination with metformin), the HTD1801 treatment group achieved a mean change in HbA1c from baseline of -1.2%, which was significantly superior to the placebo group. Notably, in the severe patient subgroup with high baseline HbA1c (≥8.5%), the mean reduction in HbA1c from baseline reached -1.6% with HTD1801.
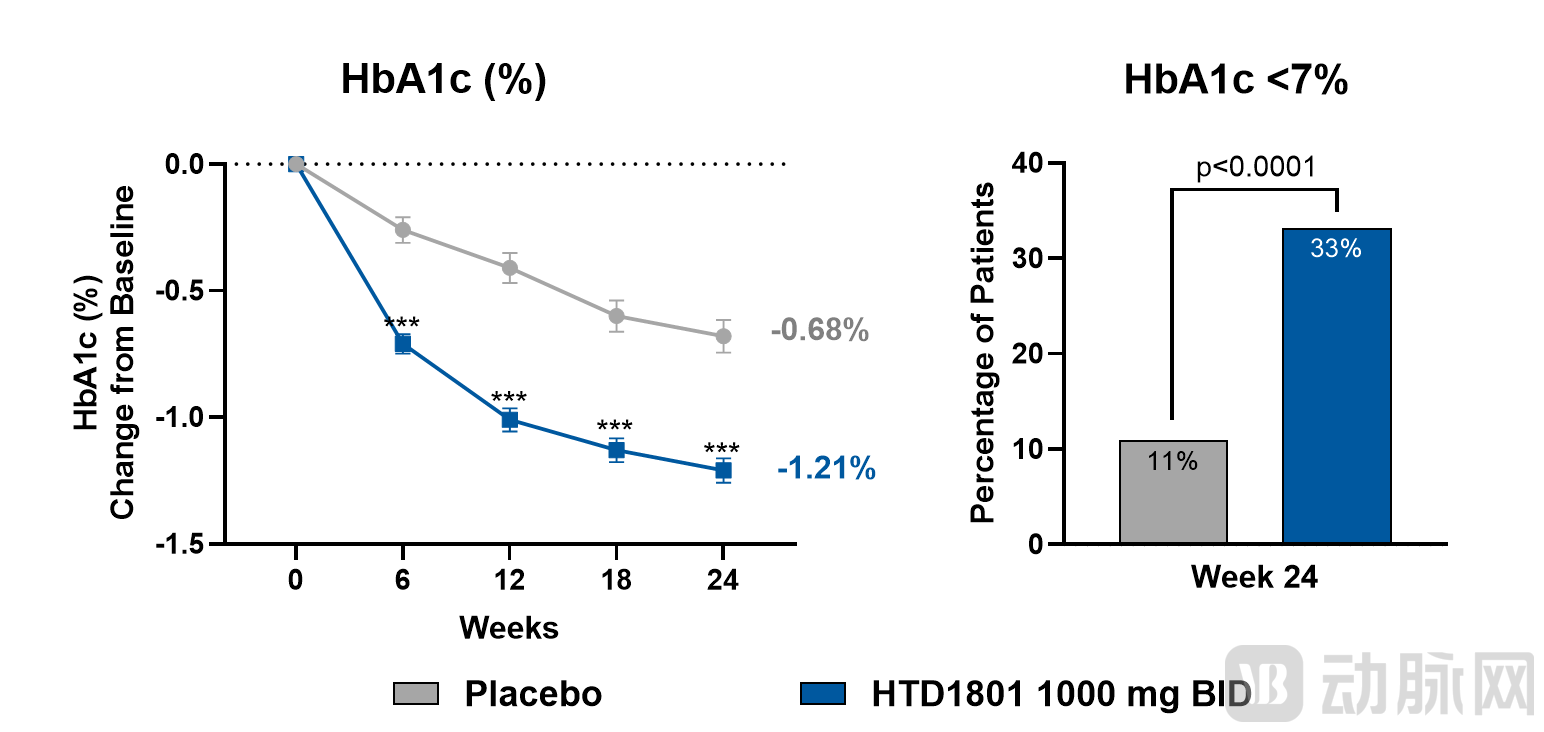
(Left - Change in HbA1c from baseline; Right - Proportion of patients achieving HbA1c < 7%)
Specifically, the HTD1801 group demonstrated significant reductions in HbA1c, LDL-C, and non-HDL-C as early as Week 6, with sustained improvements through Week 24, indicating a clinically meaningful profile characterized by rapid onset and durable efficacy. At Week 24, the proportion of patients achieving HbA1c <7% was approximately three times higher in the HTD1801 group compared to the placebo group (33% vs. 11%).
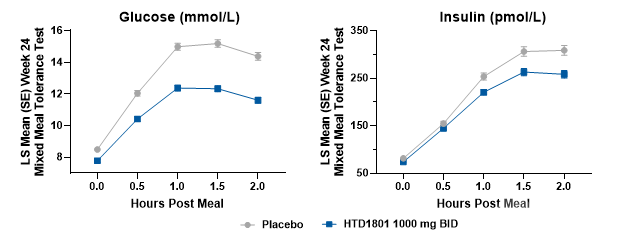
(Left - 24-week MMTT blood glucose curve; Right - 24-week MMTT insulin curve)
Compared to the placebo group, the HTD1801 group demonstrated a reduction in the area under the curve (AUC) for blood glucose and a significant decrease in the AUC for insulin at Week 24. These results indicate that HTD1801 not only effectively lowers blood glucose levels but also reduces insulin requirements, demonstrating an insulin-sparing effect.
In patients with T2DM and insulin resistance, pancreatic β-cell function is impaired, leading to abnormal insulin secretion patterns. Following treatment with HTD1801, a reduction in the insulin AUC was observed alongside significant decreases in both fasting and postprandial blood glucose levels. This suggests that by ameliorating insulin resistance, HTD1801 enhances the body's sensitivity to insulin, enabling a given amount of insulin to exert a greater glucose-regulating effect and restoring more efficient insulin-mediated metabolic control. This "unloading" effect on β-cells is expected to help preserve pancreatic β-cell function in T2DM patients over the long term, thereby providing sustained clinical benefits.
From a clinical perspective, the inability to sustain long-term therapeutic efficacy represents a significant unmet need in the treatment of type 2 diabetes (T2DM). The insulin-sparing effect of HTD1801 effectively addresses this critical clinical demand.
Building upon previously released data from the SYMPHONY 1 trial, which evaluated HTD1801 in T2DM patients with inadequate glycemic control despite diet and exercise intervention, HTD1801 not only effectively improves insulin resistance but also avoids the risks of water retention and weight gain. Long-term, HTD1801 treatment is anticipated to slow disease progression, improve cardiovascular risk factors, and holds the potential to deliver sustained health benefits for patients with diabetes.
2 Aiming at the Billion-Dollar CKM Market
In November 2023, the American Heart Association's latest guidelines introduced the concept of "Cardiovascular-Kidney-Metabolic (CKM) syndrome," highlighting the close interconnections and vicious cycle among cardiovascular disease (CVD), chronic kidney disease (CKD), and metabolic disorders—particularly obesity, diabetes, and hypertension. The guidelines emphasize the necessity of managing these conditions as an integrated whole.
Behind these guidelines lies the vast and urgent unmet need within the T2DM patient population. According to the latest data from the 11th IDF Diabetes Atlas, approximately 589 million adults (aged 20–79) worldwide are currently living with diabetes. This number is projected to rise to 850 million by 2050, representing a 45% increase, with over 90% of cases being type 2 diabetes (T2DM). In 2024, an estimated 3.4 million deaths globally were attributed to diabetes or its complications, accounting for 9.3% of all-cause mortality among adults.
China, with the largest diabetic population globally, has an estimated 150 million people living with the disease. The Chinese Guidelines for Diabetes Prevention and Treatment (2024 Edition) indicate that the comprehensive control rate for blood glucose, blood pressure, and lipid levels remains critically low, at only 4.4% to 5.6%, highlighting an urgent need for improved integrated management. The iCaReMe China study, led by Peking University People's Hospital, revealed that among a cohort of 9,000 T2DM patients, 42.8% and 38.9% had comorbid dyslipidemia and cardiovascular-kidney-metabolic (CKM) syndrome, respectively.
Therefore, "one drug, multiple effects" has become a core expectation in treating metabolic abnormality-driven CKM syndrome. At its mechanistic core, HTD1801 precisely targets two key pathways—chronic inflammation and metabolic dysregulation—to simultaneously improve glycemic and lipid profiles, reduce inflammation, and enhance cardiovascular-metabolic parameters. This integrated approach delivers systemic and comprehensive benefits for holistic management.
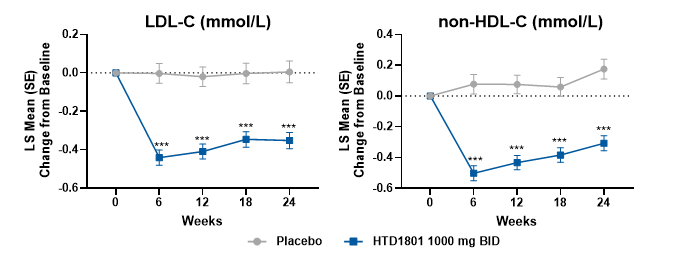
(Left-LDL-C level; Right-non-HDL-C level)
In the SYMPHONY 2 trial, significant reductions in low-density lipoprotein cholesterol (LDL-C) and non-high-density lipoprotein cholesterol (non-HDL-C) were observed in the HTD1801 group as early as Week 6, and these improvements were sustained through Week 24, indicating potential lipid and cardiovascular benefits. LDL-C and non-HDL-C are recognized as the primary and major risk factors for dyslipidemia and cardiovascular risk in diabetic patients, as well as key efficacy endpoints for lipid-lowering interventions.
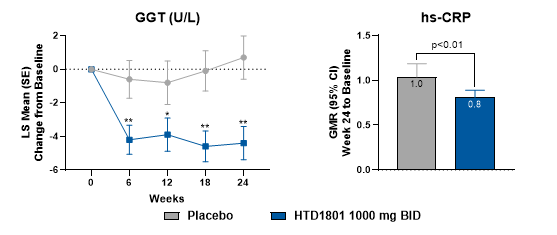
(Left-GGT level; Right-hs-CRP level)
The data also demonstrated sustained and significant reductions in markers of inflammation and oxidative stress, such as γ-glutamyl transferase (GGT) and high-sensitivity C-reactive protein (hs-CRP). Furthermore, the incidence of major adverse cardiovascular events (MACE) was 2% in the HTD1801 group, lower than the 4% observed in the placebo group, indicating a consistent trend toward cardiovascular benefit.
In a pooled analysis of two Phase III clinical trials, the HTD1801 treatment group demonstrated greater stability in eGFR compared to the placebo group in the overall population, along with stable electrolyte levels. Notably, consistent renal benefits were observed with HTD1801 in T2DM patients with mild renal impairment (60 ≤ baseline eGFR < 90), regardless of whether they were treatment-naïve or had inadequate glycemic control after metformin therapy.
After HTD1801 treatment, patients with baseline eGFR levels indicating mild renal impairment showed significant improvement in eGFR compared to both the placebo group and their own baseline levels, suggesting HTD1801's potential for early renal intervention and functional recovery. It should be noted that currently approved drugs with established renal benefits generally only slow the decline in eGFR compared to placebo rather than achieving an improvement over baseline levels.
3HighTide Therapeutics' Capital Story Enters the Realization Phase
According to the 2025 interim report, HighTide Therapeutics is expected to submit a New Drug Application (NDA) for the T2DM indication by the end of 2025, with the 52-week long-term data from the two SYMPHONY studies also scheduled to be read out soon.
Currently, multiple pipelines covering CKM syndrome are advancing in global synchronized clinical development, including those for chronic kidney disease, hypertriglyceridemia, and obesity, which have already demonstrated clinical benefits. As of the time of writing, HTD1801 has been granted two Fast Track designations and one Orphan Drug designation by the FDA.
Fundamentally, the expansion of indications is based on the demonstrated efficacy of HTD1801 in improving metabolic and anti-inflammatory pathways.
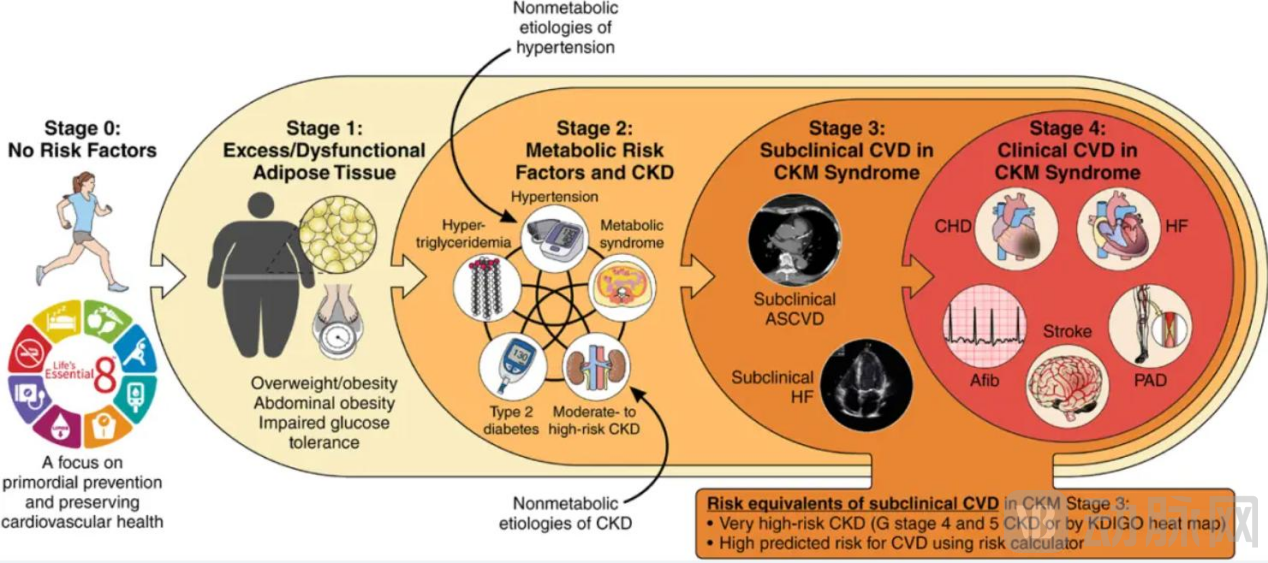 Summary of Scientific and Clinical Management Evidence for Cardiovascular-Kidney-Metabolism (CKM) Syndrome
Summary of Scientific and Clinical Management Evidence for Cardiovascular-Kidney-Metabolism (CKM) Syndrome
From a commercial perspective, expanding indications is a crucial forward-looking strategy. The treatment approach of "managing diverse diseases through a unified mechanism" and "addressing both symptoms and root causes" with a CKM-focused drug means that HTD1801, upon approval, has the potential to broaden its clinical application scenarios and significantly enhance its market scope through a "T2DM + comorbidities" indication strategy. In the long term, the core compound patent for HTD1801 is protected until 2040, while method-of-use patents for new indications such as kidney disease can extend protection at least until 2045.
For HighTide, an extended product lifecycle signifies technological validation, R&D innovation, and potential business development (BD) opportunities. Concurrently, HighTide is developing follow-up innovative drug candidates beyond HTD1801, focusing on multiple potential indications. The company has already disclosed seven pipelines, including peptide-based therapeutics and mitochondrial uncouplers, targeting conditions such as obesity, neurodegenerative disorders, and alcoholic hepatitis.
According to the prospectus, the global market for metabolic diseases is projected to reach $458 billion by 2032. With HTD1801—a first-in-class innovative drug derived from natural products and featuring a multi-mechanism approach—what story will HighTide Therapeutics tell in the field of metabolic diseases? The industry awaits with great anticipation.
