China's SMILE laser market: global giants gather amid import monopoly, when will domestic rise?

Intalight
High-end Ophthalmic Equipment Developer

TowardPi
High-end Ophthalmic Medical Device Developer
The peak season for refractive surgery during summer vacation has just passed, yet the underlying growth drivers of China's ophthalmic consumer healthcare sector are quietly shifting. As profit margins on core high-value consumables are being squeezed by volume-based procurement policies, major listed ophthalmic chains are increasingly pinning their revenue growth hopes on the introduction of advanced equipment and the promotion of high-value-added procedures. Among these, femtosecond laser devices and related refractive and cataract surgeries have become the central focus of this transformation.
ZEISS's latest blockbuster product serves as a clear testament to this trend: In China, within just three months of its launch in 2025, the new-generation robotic all-femtosecond system VISUMAX 800 was installed in 63 healthcare institutions and rapidly performed over 20,000 refractive surgeries.
Unfortunately, despite the current boom in ophthalmic femtosecond laser equipment and related surgeries, there is still no domestically produced equivalent available in the Chinese market. However, that may soon change—a breakthrough for local manufacturers appears to be on the horizon.
For years, China's ophthalmic femtosecond laser equipment market has been characterized by a pattern of import dominance and rapid technological iteration. A notable shift in 2025 is the gathering of all four major global laser systems capable of performing all-femtosecond surgeries in the Chinese market. At the same time, femtosecond laser systems for cataract surgery continue to advance—with upgrades increasingly focused on improving surgical precision, safety, and patient experience. These developments are further strengthening the influence of imported products in the market.
Corneal refractive surgery primarily includes several mainstream techniques such as PRK (photorefractive keratectomy), LASIK (commonly referred to as "half-femtosecond"), and SMILE (small incision lenticule extraction, or "all-femtosecond"). Among these, SMILE involves using a femtosecond laser to perform two scans at different depths within the corneal stroma, creating a lenticule that is then removed through a small incision, thereby correcting myopia. Known for its minimally invasive nature, better preservation of corneal structural integrity, and faster recovery time, SMILE has emerged as one of the most advanced forms of corneal refractive surgery. As a result, competition in this field is now heavily focused on all-femtosecond laser systems, which cost tens of millions of RMB per unit.
In 2011, the SMILE (Small Incision Lenticule Extraction) procedure entered the Chinese market. For over a decade, Zeiss's VisuMax remained the only all-femtosecond laser system available in mainland China, undergoing multiple iterations during this period. The year 2025, however, has marked an explosive expansion in the adoption of all-femtosecond equipment in China, with all four major global SMILE-capable laser systems now concentrated in the market.
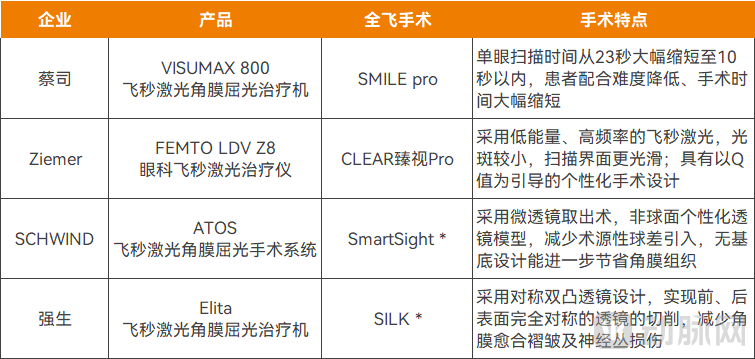
Femtosecond laser devices approved for 2025 (* indicates the device is approved, but the all-femto procedure is not yet approved), Source: National Medical Products Administration, Public information from companies
Specifically, in March 2025, ZEISS launched its new-generation robotic all-femtosecond system, the VISUMAX 800, equipped with the minimally invasive SMILE pro procedure. In May of the same year, Ziemer's already-approved FEMTO LDV Z8 system in China was upgraded with all-femtosecond capabilities, leading to the introduction of the CLEAR Vision Pro procedure based on this platform.
The newly launched products and functionalities have ushered in a new era of more efficient and precise corneal refractive surgery. For example, ZEISS's VISUMAX 800 has dramatically reduced single-eye scanning time from 23 seconds to under 10 seconds, significantly lowering the demands on patient cooperation and shortening overall procedure time. Meanwhile, Ziemer's FEMTO LDV Z8 employs a low-energy, high-frequency femtosecond laser that produces smaller spot sizes and creates smoother scanning interfaces, in addition to offering Q-value-guided personalized treatment planning.
It is worth noting that Ziemer's FEMTO LDV Z8 had already received approval in China for cataract surgery. With the addition of all-femtosecond (SMILE) capabilities, the Z8 has now become the only platform in the world capable of performing all-femtosecond, femtosecond-LASIK (half-femtosecond), and femtosecond laser-assisted cataract surgeries. Compact, mobile, and multi-functional, it significantly reduces procurement and operational costs for healthcare institutions.
Moreover, two femtosecond laser systems—SCHWIND ATOS and Johnson & Johnson's Elita—were also approved for marketing by the NMPA in 2025. Currently, the approved indication scope of both products is limited to femtosecond-assisted LASIK (FS-LASIK) procedures; however, their applications for all-femtosecond (SMILE) procedures have already been approved for use in overseas markets.
The SmartSight all-femtosecond (SMILE) technology of SCHWIND ATOS adopts the lenticule extraction technique and an aspheric personalized lenticule model. This design can effectively reduce the introduction of surgically induced spherical aberration, further preserve corneal tissue, and optimize post-operative visual quality. The SILK procedure of Johnson & Johnson's Elita uses a symmetrical biconvex lenticule design, enabling the ablation of lenticules with completely symmetrical anterior and posterior surfaces. This minimizes corneal healing folds and damage to nerve plexuses, thereby enhancing recovery speed and visual quality. Currently, this procedure has been piloted in Boao, Hainan.
As these two devices obtain approval for and are officially launched for all-femtosecond (SMILE) procedures in China in the next phase, ophthalmologists and patients will have more options to choose from.
While femtosecond laser refractive surgery becomes further popularized, femtosecond laser systems designed for cataract surgery are also continuously advancing—with precision and safety as the core priorities in their technological iterations.
Femtosecond Laser-Assisted Cataract Surgery (FLACS) is capable of performing key procedures such as corneal incisions, anterior capsulotomies, lens fragmentation, and arcuate incisions. Compared with traditional cataract surgery, FLACS offers significant advantages in terms of precision, safety, and postoperative visual quality. Its precision advantage is particularly prominent when implanting refractive intraocular lenses, making it the mainstream choice for high-end cataract surgery.
In 2025, Johnson & Johnson launched the Bausch + Lomb VICTUS 7.0 Navigation Femtosecond Laser Cataract Surgery System, equipped with iris positioning navigation and high-definition scanning dual systems, supporting two personalized astigmatism correction modes; completing a 360° capsulotomy in 1 second, significantly reducing operation time and improving surgical efficiency; featuring innovative aqueous patient interface technology to minimize corneal endothelial damage and subconjunctival hemorrhage. As a classic femtosecond laser cataract surgery device, LENSAR was also upgraded in 2025, introducing a new oval-shaped Patient Interface (PI) in China for better adaptation to patients with small palpebral fissures, reducing intraoperative discomfort and enhancing surgical success rates.
Overall, major imported femtosecond laser devices are accelerating innovation, driving the upgrading of surgeries toward personalization and higher efficiency. Furthermore, the functions of these devices are gradually breaking down scenario barriers, and a trend of "one device for multiple uses" is emerging. Their penetration rate in the domestic ophthalmic market is expected to further increase.
The competition in China's ophthalmic medical market is intensifying, compounded by the squeeze on profit margins of high-value consumables due to volume-based procurement policies. Over the past two years, ophthalmic medical institutions have been under growing pressure to identify new sources of revenue. Financial reports from listed ophthalmic companies for the first half of 2025 reveal that actively introducing and adopting femtosecond laser equipment, along with promoting high-value-added surgical procedures, has become a critical strategy to boost revenue from refractive services and stabilize income from cataract programs.
In the field of refractive surgery, the iterative upgrading of femtosecond laser equipment has clearly become one of the key engines driving business growth.
In the first half of 2025, Aier Eye Hospital reported an 11.14% year-on-year increase in revenue from its refractive surgery segment. A significant driver of this growth has been the widespread patient acceptance and accelerated clinical adoption of new procedures such as Total Refractive Solution (TRS) by Alcon, SMILE 4.0, and SMILE pro. These advanced techniques have contributed to the continuous optimization of the refractive service mix and a rise in the average procedure price. Similarly, Huaxia Eye Hospital achieved a 13.75% year-on-year growth in refractive revenue, partly attributable to its timely introduction and active promotion of advanced techniques like ZEISS's SMILE pro, SMILE 4.0, and Alcon's TRS. This strategic focus on upgrading its portfolio of refractive services has effectively enhanced the value and composition of its surgical offerings.
Both He Eye Specialist Hospital and Bright Eye Hospital also recorded modest growth in their refractive surgery segments. Their financial reports highlighted that the introduction of the latest-generation all-femtosecond equipment and the upgrading of refractive surgical techniques have enhanced their market competitiveness.
In the field of cataract treatment, the impact of the intraocular lens (IOL) volume-based procurement policy on ophthalmic medical institutions has been evident, with most experiencing declines in both revenue and gross margin. Similarly, accelerating technological iteration and upgrading surgical techniques have become critical measures to "rescue" cataract programs. For instance, the introduction of new-generation femtosecond laser systems for cataract surgery and the promotion of FLACS (femtosecond laser-assisted cataract surgery) have emerged as key strategies for institutions to mitigate declining revenue in this segment.
The proactive adoption and promotion of new equipment and surgical techniques by healthcare institutions have directly driven upstream equipment sales. Taking ZEISS's all-femtosecond portfolio as an example: although its previous-generation systems and earlier SMILE 3.0 and SMILE 4.0 procedures still hold significant market share, the latest VISUMAX 800 model has been enthusiastically embraced by both institutions and patients.
According to data released by Zeiss, after its launch, the VISUMAX 800 was officially installed in hospitals in May 2025. In just three months by the end of August, its installations had covered 63 medical institutions across China. Additionally, the number of SMILE pro procedures performed using the VISUMAX 800 exceeded 20,000 within the same three-month period. Liu Yonghua, Head of Zeiss Group China, stated that a decade ago, it took two and a half years to reach 10,000 all-femtosecond (SMILE) procedures. Today, however, the number of SMILE pro procedures has surpassed 20,000 in just three months—representing nearly a 10-fold increase in speed.
In summary, within the fields of refractive and cataract surgery, the trend of high-end equipment—represented by femtosecond lasers—driving revenue growth on the service side is becoming increasingly evident. This not only reflects the structural shift in the sources of revenue and profit for ophthalmic institutions but also underscores the strong patient demand for new technologies and advanced surgical procedures.
Against the backdrop of imported femtosecond laser equipment dominating the market and intensifying iterative competition, the progress of China's domestic femtosecond technology—which has yet to see a product launch—is also worthy of attention. In recent years, Chinese companies have significantly accelerated their efforts, making notable strides in both R&D and product validation, bringing the goal of achieving a breakthrough ever closer.
Recently, the Chinese femtosecond laser devices exhibited during the the 29th Congress of Chinese Ophthalmological Society has drawn attention, becoming an important signal of the rise of technology.
Among them, Fine Vision showcased its independently developed Fine Vision 2000, along with the innovative femtosecond FINE procedure. This product has achieved breakthroughs in multiple aspects: for instance, its scanning time is less than 9 seconds, and it is equipped with automatic watermark center positioning and astigmatism axis spin compensation, which further enhances the accuracy of refractive correction; in addition, the laser-ablated lenticule surface is smooth, and the lenticule separation and extraction process is smooth and easy, reducing irritation to the cornea. The product entered mass production in 2024 and has currently launched registered clinical trials.

Publicly displayed domestic ophthalmic femtosecond laser equipment, source: publicly available corporate information
Another domestically produced femtosecond laser exhibited at this conference is the FEMTOPIA BVM1000 from Andun Medic, which is also undergoing multi-center registration clinical trials.
These two products are not making their public debut for the first time; they have also appeared frequently at other major ophthalmic exhibitions over the past two years. This move not only demonstrates the technological strength of China's domestic devices to the industry, but also signifies a major breakthrough in the product validation of China's domestic equipment. Furthermore, pre-launch warm-up efforts are progressing in an orderly manner, allowing ophthalmologists across the country to familiarize themselves with the products' features in advance.
According to Cao Feng, founder of Fine Vision, the Fine Vision 2000 is expected to be launched and applied clinically in the second half of 2026 to 2027. In other words, the time when the import monopoly of femtosecond laser devices is broken may not be far off.
Besides, companies such as Intalight and TowardPi Medical have also laid out the research and development of ophthalmic femtosecond laser equipment. Chinese enterprises have formed a diversified R & D pattern, laying a foundation for more comprehensive technological and market breakthroughs. However, the current market promotion focus of these enterprises is on other product lines, or they have not yet shown the "true features" of femtosecond laser products.
As mentioned earlier, different femtosecond laser devices correspond to different surgical names. To standardize pricing, in 2025, the National Healthcare Security Administration (NHSA) issued the Interim Guidelines for Establishing Ophthalmology-related Medical Service Price Items. In this document, "femtosecond laser myopia correction surgery" was established as an independent item. Drawing on previous technical standards, the item name was decoupled from laser types and specific devices, and defined as "corneal stromal lenticule extraction fee" based on actual clinical outcomes. This not only facilitates understanding of clinical pricing but also leaves more room for future innovation and translation.
After years of market education, surgical optimization, clinical research, and the development of expert consensus, China’s ophthalmic femtosecond laser market is on the eve of a boom. Driven by factors such as market demand and policy guidance, there is reason to believe that in the near future, China’s domestic femtosecond laser devices will officially "take a seat at the table". This will not only bring more cost-effective options to domestic patients, but also make China’s landscape of high-end ophthalmic equipment more complete.
