HER2 lung cancer meets its "formidable opponent": Zongaitini, the world's first oral targeted drug, approved in China, achieving a 71% response rate

On August 29, 2025, Boehringer Ingelheim's innovative lung cancer drug—Zongaitini Tablets (also known as Zonggetini)Approved for conditional marketing authorization by China's National Medical Products Administration (NMPA) through the priority review and approval procedure.!For use withPresence of HER2 (ERBB2) activating mutations and previously received at least one systemic treatment for unresectable diseaseLocally advanced or metastatic non-small cell lung cancer (NSCLC)Adult patienttreatment.
The drug's launch brings new hope for patients who previously had limited treatment options, and injects fresh momentum into China's field of HER2-mutant lung cancer treatment, enabling more patients to extend the light of life through precision therapy!
▲ Screenshot from“NMPA”



Zongertinib tablets (also known as Zongertinib, English name: Zongertinib, development code: BI 1810631, brand names: Hernuxeos®, Hernexeos®) is a novel oral HER2-selective tyrosine kinase inhibitor developed by Boehringer Ingelheim. It covalently binds to the HER2 tyrosine kinase domain (including exon 20 insertion mutations) without affecting the wild-type epidermal growth factor receptor.
The drug's global approval process continues to advance: on August 8, 2025, it received accelerated approval from the U.S. FDA for adult patients with unresectable or metastatic non-squamous non-small cell lung cancer (NSCLC) whose tumors have been confirmed by an FDA-approved test to harbor HER2 (ERBB2) tyrosine kinase domain (TKD) activating mutations and who have received prior systemic therapy.The first oral HER2 lung cancer targeted therapy drug, alsoThe world's first oral small-molecule inhibitor targeting this mutation type。
Just one week later (August 15, 2025), the National Comprehensive Cancer Network (NCCN) updated its clinical practice guidelines for NSCLC tumors, listing zongertinib tablets (Hernexeos®) as the preferred subsequent treatment option for patients with advanced/metastatic HER2-mutant NSCLC who have received prior systemic therapy.
And today (August 29, 2025), this major anti-cancer drug has finally been approved for marketing in China! The long-awaited "life-saving drug" for Chinese patients with HER2-mutant lung cancer has officially arrived, bringing a new precision treatment option for patients!


The current approval of zongaitini tablets is primarily based on the excellent data from the BeamionLUNG-1 (NCT04886804) Phase Ia/Ib study, and the related results have been simultaneously published in The New England Journal of Medicine. This study included patients with advanced or metastatic HER2-mutant non-small cell lung cancer (NSCLC) and comprised three cohorts: 75 patients with tyrosine kinase domain mutations (Cohort 1), 31 patients with tyrosine kinase domain mutations who had previously received HER2-targeted antibody-drug conjugate treatment (Cohort 5), and 20 patients with non-tyrosine kinase domain mutations (Cohort 3).
The results show: as of the data cutoff, Cohort 1 has been confirmedThe objective response rate (ORR) reached 71%.(95%CI:60-80),Median duration of response was 14.1 months(95% CI: 6.9 - not evaluable),The median progression-free survival (PFS) was 12.4 months.(95%CI: 8.2-unable to evaluate); Cohort 5 confirmedThe ORR was 48%.(95%CI: 32-65); Cohort 3Confirmed ORR is 30%(95% CI: 15-52) (see figure below).
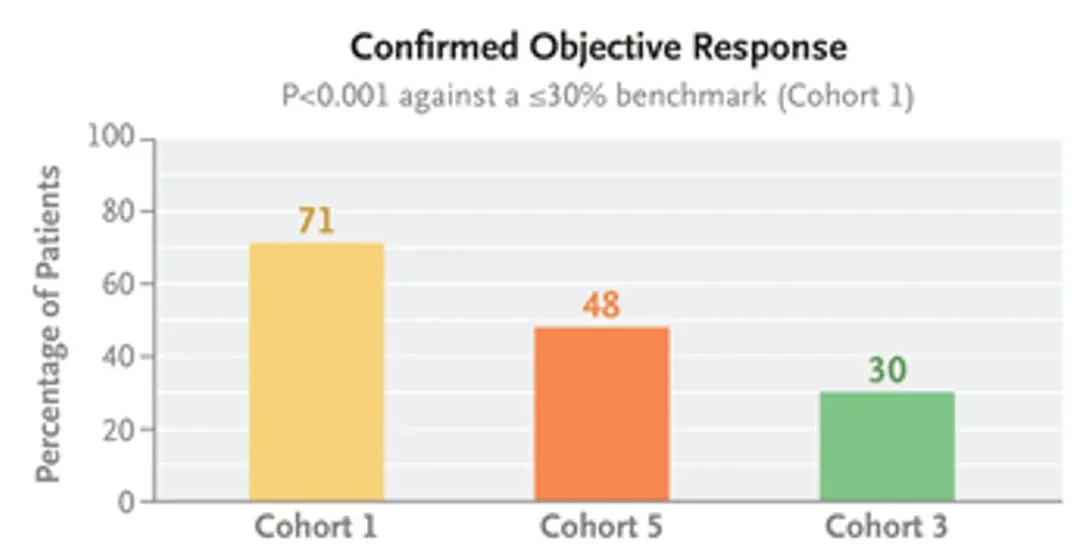
▲Image source“N Engl J Med”, copyright reserved, if intellectual property rights are unintentionally infringed, please contact us for removal.


Believing that after seeing the data above, patients with HER2-positive lung cancer or other solid tumors must have renewed hope! The good news is that clinical trials for HER2-positive solid tumors are currently underway in China, and a large number of patients have already successfully enrolled through the Global Cancer Doctor Network's new cancer drug recruitment center, receiving help from new anticancer drugs!

1) Age ≥18 years, regardless of gender;
2) Histologically or cytologically confirmed HER2-positive locally advanced, recurrent, or metastatic gastric cancer (including adenocarcinoma of the gastroesophageal junction);
3) Prior failure of at least one line of standard therapy (trastuzumab combined with chemotherapy);
4) According to RECIST 1.1: at least one evaluable lesion at baseline;
5) ECOG performance status score of 0 or 1.

Patients wishing to apply for clinical trials of new anti-cancer drugs need to compile recent pathology reports, genetic testing reports, and other documents, then submit them to the Medical Department of the Global Oncologist Network (or call directly).Medical School 4006667998, or send the photo todoctor.huang@globecancer.com, leave contact information in the email for preliminary evaluation.
Our experts will provide you with a comprehensive analysis and interpretation of your test report. We will call you within one business day to recommend a medication plan and match you with suitable clinical trial programs for patient enrollment. Note: As a leading tumor patient service platform in China, Global Oncologist Network promises to keep all participants' personal information confidential and to fully comply with national laws and regulations related to clinical research throughout the process.


Over the past decade, significant progress has been made in lung cancer treatment, reversing survival rates for patients with advanced cancer. In addition to the recently approved zongaitini, many more new anticancer drugs are under development! The editor also hopes that more new anticancer drugs will be approved and marketed in the future, or included in medical insurance coverage, so that more cancer patients can receive treatment and have access to medication!
The good news is that several new anti-cancer drugs and innovative technologies are currently undergoing clinical trials in China, and the "Ark Assistance Program" can also provide patients with free access to both approved and unapproved new drugs. Patients interested in joining clinical trials or seeking help with new anti-cancer technologies may contactGlobal Cancer Doctor Network Medical Department (400-666-7998), understand the detailed inclusion and exclusion criteria, and conduct a preliminary assessment of the condition.


[1]Heymach J V,et al.Zongertinib in Previously Treated HER2-Mutant Non–Small-Cell Lung Cancer[J]. New England Journal of Medicine, 2025, 392(23): 2321-2333.
https://www.nejm.org/doi/10.1056/NEJMoa2503704?url_ver=Z39.88-2003
[2]https://www.nmpa.gov.cn/zhuanti/cxylqx/cxypxx/20250829091544164.html
This article is originally published by Global Oncologist Network and may not be reproduced without authorization.
