Antengene lands up to $1.18B UCB deal for autoimmune TCE bispecific
Hong Kong-listed Antengene Corporation Limited (Antengene, SEHK: 6996.HK) announced that it has entered into an exclusive global licensing agreement with UCB, a Belgian biopharmaceutical company, granting UCB the rights to develop, manufacture, and commercialize ATG-201, a CD19×CD3 bispecific T-cell engager targeting B cell-mediated autoimmune diseases.
According to the announcement, Antengene will receive USD 80 million (comprised of an upfront payment of USD 60 million and near-term milestone payments of USD 20 million), and is eligible to receive future milestone payments of over USD 1.1 billion, along with tiered royalties based on future net sales. This collaboration not only opens a pathway for the global development of ATG-201, but also further validates the industry trend of T-cell engager therapies expanding from oncology into autoimmune indications.

ATG-201: A CD19×CD3 Bispecific Antibody Featuring "2+1" Structure with Masking Technology
ATG-201 is a second-generation T-cell engager candidate independently developed by Antengene, featuring a "2+1" bivalent structure: one end bivalently targets the CD19 antigen on the surface of B cells, while the other end monovalently binds to the CD3 molecule on the surface of T cells. This design incorporates steric hindrance masking technology and a CD3 sequence with fast-on/fast-off kinetics, thereby minimizing the risk of cytokine release syndrome while effectively depleting B cells.
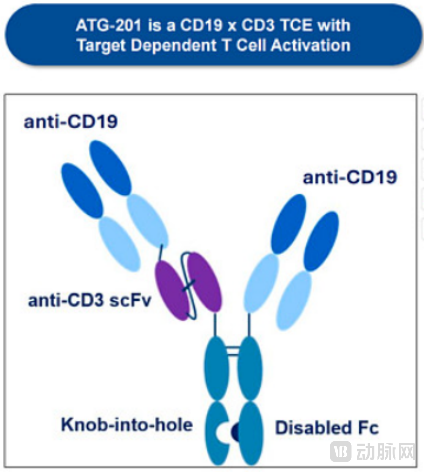
Cryo-electron microscopy structural analysis has revealed that when ATG-201 is not cross-linked with CD19, its CD3 binding site is sterically masked by its own structure. It activates T cells only upon encountering CD19-positive B cells, effectively avoiding non-specific attack on healthy cells.
Preclinical study results have validated the design advantages of ATG-201. In vitro, using peripheral blood mononuclear cells derived from patients with systemic lupus erythematosus, ATG-201 demonstrated significantly superior B cell depletion capacity compared to a control T-cell engager, while simultaneously inducing markedly lower levels of cytokine release. In CD34+ cell-humanized mouse models, a single administration achieved complete and durable B cell depletion in peripheral blood, bone marrow, and spleen, with cytokine peak levels substantially lower than those in the control group.
Notably, compared to first-generation T-cell engagers, ATG-201 resulted in significantly reduced expression of T cell exhaustion markers (such as PD-1 and Tim-3) in in vitro culture systems, indicating a substantial improvement in mitigating chronic T cell overstimulation. Furthermore, in autoimmune mouse models (such as the MOG-EAE and MRL-lpr SLE models), surrogate molecules of ATG-201 demonstrated excellent therapeutic efficacy, significantly delaying disease progression. Experiments in non-human primates further showed that a monkey surrogate of ATG-201 achieved deep and durable naive B cell depletion while inducing only low-level, transient cytokine release.
In summary, leveraging its dual characteristics of targeted activation and low toxicity, ATG-201 has demonstrated a differentiated advantage in preclinical studies of "precisely killing aberrant B cells while safely preserving normal cells," providing a strong rationale for advancing into clinical trials. Antengene expects to initiate clinical trials for ATG-201 in the first quarter of 2026.
From AnTenGager to Clinical Candidates: Expanding Therapeutic Reach in Oncology and Autoimmunity
Behind this licensing deal, what is also noteworthy is the alignment between the two parties in terms of pipeline stage, technological approach, and global capabilities.
Antengene is a research-driven, commercial-stage biotechnology company focused on discovering and developing first-in-class or best-in-class therapies for diseases with significant unmet medical needs. Antengene possesses a robust research and development pipeline spanning multiple stages from preclinical to commercial. In addition to ATG-201, its in-house discovered pipeline includes the CLDN18.2 antibody-drug conjugate ATG-022, the oral CD73 inhibitor ATG-037, the PD-L1×4-1BB bispecific antibody ATG-101, the CD24-targeting macrophage activator ATG-031, and the PRMT5-MTA small molecule inhibitor ATG-042, among others. These candidates cover a range of indications including oncology and autoimmune diseases.
Antengene's independently developed second-generation T-cell engager platform, AnTenGager, employs a "2+1" bivalent binding structure and steric hindrance masking technology. This enables targeting of low-expression antigens and reduces the risk of cytokine release syndrome, demonstrating broad application potential in autoimmune diseases, solid tumors, and hematologic malignancies.
Antengene has accumulated extensive experience in advancing innovative drugs to commercialization. To date, Antengene has obtained 32 investigational new drug approvals in the United States and Asia-Pacific markets, and has secured 10 new drug application approvals in the Asia-Pacific region. Its first commercialized product, XPOVIO® (generic name: selinexor), has received marketing approvals in nine markets, including Mainland China, Taiwan China, Hong Kong China, Macau China, South Korea, Singapore, Malaysia, Thailand, Indonesia, and Australia. Furthermore, it has been included in national insurance schemes in five of these markets.
As the global partner, UCB was founded in 1928 and is headquartered in Brussels, Belgium, employing over 9,000 people in approximately 40 countries. UCB has a long-standing focus on developing innovative therapies for immune system and central nervous system diseases, reporting revenues of approximately 7.7 billion euros in 2025.
By incorporating ATG-201 into its pipeline, UCB gains an early-stage autoimmune disease candidate with mechanistic expansion potential, supplementing its portfolio in B cell-related diseases. For Antengene, this collaboration not only provides financial support but, more importantly, enables the acceleration of the project's overseas clinical advancement and regulatory pathways by leveraging UCB's global development and commercialization network.
TCE Advances into Autoimmunity
In recent years, the autoimmune field has progressively evolved from a "long-term chronic disease management market" into a high ground for high-value innovative biologic transactions. According to public deal data, since 2020, significant transactions centered on key targets and differentiated mechanisms have continuously emerged. In 2020, Johnson & Johnson acquired Momenta for $6.5 billion, with the core asset being an FcRn (neonatal Fc receptor) antibody. In 2022, Amgen acquired Horizon for $27.8 billion, betting on an IGF-1R (insulin-like growth factor 1 receptor) antibody pipeline. Entering 2024, the participation of Chinese innovative pharmaceutical companies in this field has significantly increased. Companies such as InnoCare Pharma, Keymed Biosciences, and Genrix Biopharmaceutical have successively out-licensed their autoimmune bispecific antibodies. In early 2026, Helixon reached a collaboration with Sanofi, with a total deal value of $2.56 billion.
Against this backdrop, T-cell engagers have begun to enter the autoimmune landscape. T-cell engagers are essentially a class of immunotherapies that utilize bispecific or multispecific antibodies to achieve precise engagement between T cells and target cells. Their core mechanism bypasses antigen presentation, directly activating T cells to eliminate target cells. Currently, the approximately 10 approved T-cell engager products globally remain primarily concentrated in the fields of hematologic malignancies and, to some extent, solid tumors, targeting antigens such as CD19, CD20, BCMA, and GPRC5D. However, mechanistically, the profound B cell depletion capability of T-cell engagers provides a realistic foundation for their application in B cell-driven diseases such as systemic lupus erythematosus.
In recent years, numerous global large pharmaceutical companies and emerging biotech firms have placed significant bets on autoimmune T-cell engagers. In late 2024, U.S.-based Candid Therapeutics entered into three T-cell engager research and development collaborations with multiple Chinese companies within a single day, with total potential transaction value exceeding $1.32 billion. Harbour BioMed's BCMA/CD3 bispecific antibody HBM7020 (targeting autoimmune indications) has entered into an exclusive licensing agreement with Otsuka, with a total transaction value of approximately $670 million. It is evident that while autoimmune T-cell engagers are still in their early stages, they are progressively entering the realm of industrial collaboration, with relevant molecules and technology platforms provided by Chinese innovative pharmaceutical companies becoming a significant source of external sourcing for multinational corporations.
From a longer-term perspective, the application of T-cell engagers in autoimmunity remains in early-stage exploration, and the core challenges for their clinical translation are not yet fully resolved. On one hand, compared to oncology, the tolerance for safety risks in autoimmune diseases is considerably lower. Mitigating the risk of cytokine release syndrome through structural design and dosing strategies remains a critical challenge. On the other hand, how T-cell engagers will differentiate from or synergize with existing established therapies (such as anti-CD20 monoclonal antibodies and other B cell depletion strategies) will also directly impact their clinical positioning and commercial potential.
As programs like ATG-201 advance into the clinic, the industry will gradually accumulate key evidence regarding whether profound B cell depletion can translate into durable disease remission. The answer to this question will not only determine the success or failure of individual products but will also define the developmental boundaries of T-cell engager technology in the autoimmune field, making this a space worth watching closely in the years ahead.
