Beyond the deal: Pfizer snaps up another China GLP-1 asset with $495M Sciwind pact
On February 24, Sciwind Biosciences and Pfizer China announced a strategic commercialization collaboration for Ecnoglutide injection, a new-generation cAMP-biased GLP-1 receptor agonist.
Under the agreement, Pfizer will obtain exclusive commercialization rights for the product in Mainland China, marking an important first step to advance Pfizer's global strategy in the metabolic field in China. Sciwind Biosciences will remain the Marketing Authorization Holder (MAH) and will be responsible for the research and development, registration, manufacturing, and supply of the product.
Sciwind Biosciences is eligible to receive up to $495 million in payments from Pfizer, including upfront, regulatory, and sales milestone payments.
The World's First Approved cAMP-Biased GLP-1 Receptor Agonist
GLP-1 receptor agonists can be categorized into two main classes: biased and non-biased. Non-biased agonists, while activating the cAMP signaling pathway, promote β-arrestin recruitment, which in turn induces GLP-1 receptor internalization and desensitization. In contrast, biased GLP-1 receptor agonists such as Ecnoglutide, while preferentially activating the cAMP signaling pathway, minimize β-arrestin recruitment. This signaling bias limits receptor desensitization and downregulation, allowing more receptors to remain on the cell surface and sustain therapeutic signal transduction. The unique mechanism of biased agonists is a key factor in enhancing clinical efficacy and improving metabolic benefits.
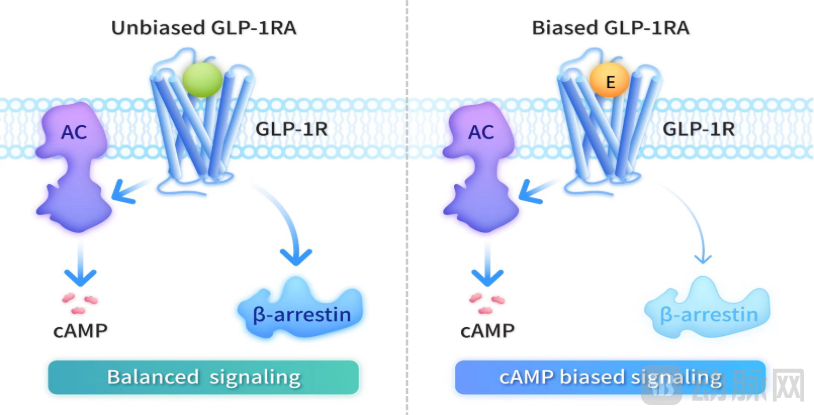 Non-biased and Biased GLP-1RA, Image Source: Sciwind Official Website
Non-biased and Biased GLP-1RA, Image Source: Sciwind Official Website
Independently developed by Sciwind Biosciences as a new-generation cAMP-biased GLP-1 receptor agonist, Ecnoglutide injection has demonstrated robust efficacy and a favorable safety profile across multiple clinical studies, owing to its unique biased mechanism. In Chinese patients, it achieved a placebo-adjusted mean weight loss of 15.1%, with 92.8% of patients attaining clinically meaningful weight loss, and over 80% of patients achieving glycemic targets (HbA1c < 7.0%).
Based on its favorable safety and tolerability profile, on January 30, 2026, Ecnoglutide injection was approved by the NMPA for glycemic control in adults with type 2 diabetes, becoming the world's first approved cAMP-biased GLP-1 receptor agonist. In addition, its marketing authorization application for long-term weight management in adults has been accepted by the NMPA.
The successful approval of Ecnoglutide injection is underpinned by Sciwind Biosciences' three core technology platforms:
BiasVantage™ Biased Agonist Discovery Platform: This platform leverages structure-based rational design and comprehensive signaling assays to create precision therapeutics with enhanced efficacy and reduced desensitization. The platform develops signaling-selective ligands that offer extended receptor activity through reduced desensitization, leading to excellent therapeutic outcomes compared to unbiased approaches. Its signaling assay systems can quantify pathway-specific activity including cAMP production and β-arrestin-mediated receptor internalization, significantly accelerating discovery compared to traditional screening approaches and ensuring only the most promising candidates advance to clinical development.
OralVantage™ Oral Peptide Delivery Platform: This platform overcomes major challenges in developing oral peptide therapeutics by addressing enzymatic degradation and poor gastrointestinal absorption through enhanced molecular stability and advanced formulation technologies. The platform enables peptides to achieve clinically meaningful exposure by combining rational peptide design with innovative drug delivery strategies that significantly improve absorption across the gastrointestinal tract. This approach simultaneously addresses both peptide stability and absorption challenges, offers versatility across broad peptide structures and disease indications, and creates robust intellectual property protection.
HaleVantage™ Half-life Extension Platform: This platform improves pharmacokinetic properties through fatty acid modification and antibody-peptide conjugation to enable extended dosing intervals from weekly to monthly or potentially longer. The platform enhances patient convenience and compliance by extending peptide half-life through systematic optimization including selection of conjugation sites, refinement of peptide sequences to optimize enzymatic stability, and structural tuning of fatty acids. It utilizes various modification strategies offering customized approaches tailored to specific therapeutic demands and integrates pharmacokinetic evaluation systems across multiple animal models for early identification of optimal modifications.
Together, these three platforms constitute Sciwind Biosciences' core technological advantages in metabolic disease drug development, providing strong support for the innovative design, clinical efficacy, and commercial advancement of Ecnoglutide injection. Beyond Ecnoglutide, leveraging its core technology platforms, Sciwind Biosciences has also established multiple innovative drug pipelines at various stages of development, with indications covering specialized areas including metabolic dysfunction-associated steatohepatitis (MASH), obstructive sleep apnea (OSA), and influenza. Sciwind Biosciences has also entered into license and collaboration agreements with companies such as Verdiva Bio Limited and HK inno.N Corporation.
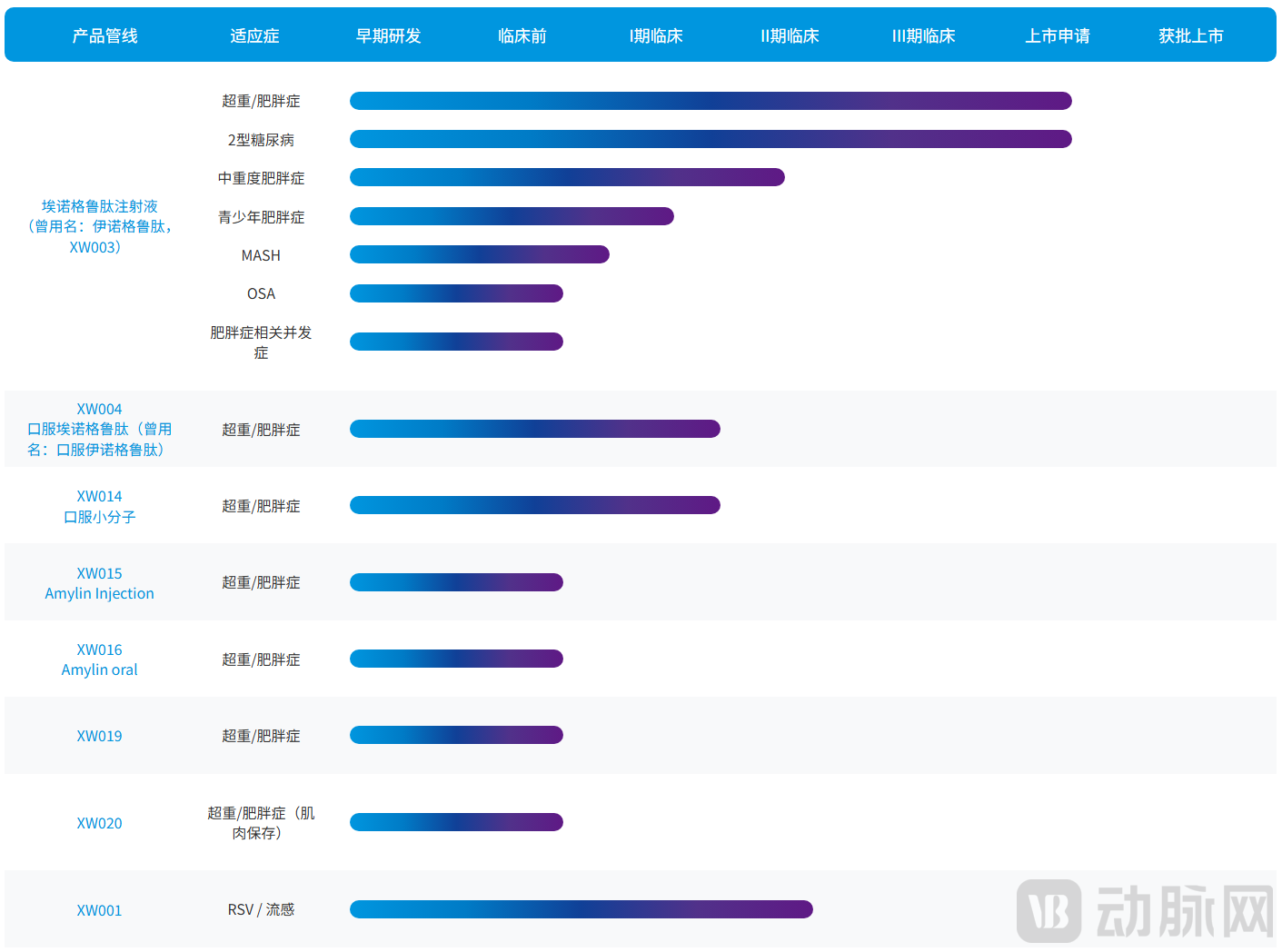 Sciwind's Pipeline Layout, Source: Sciwind Official Website
Sciwind's Pipeline Layout, Source: Sciwind Official Website
$11.78 Billion: Pfizer's Three GLP-1 Deals in Four Months
This collaboration underscores Pfizer's long-term commitment to playing a leading role in the next generation of chronic weight management therapies. Within less than four months, Pfizer has completed three consecutive transactions in the GLP-1 field (including this $495 million collaboration with Sciwind Biosciences), with total collaboration value reaching $11.78 billion.
In November 2025, Pfizer announced the successful completion of its acquisition of Metsera, with a total transaction value of approximately $9.2 billion. Through this acquisition, Pfizer obtained a pipeline including MET-097i (a once-weekly and once-monthly injectable GLP-1 receptor agonist) and MET-233i (a monthly injective amylin analog candidate).
In December 2025, Pfizer entered into a collaboration with YaoPharma, a subsidiary of Fosun Pharmaceutical, to jointly develop, manufacture, and commercialize YP05002, a GLP-1 receptor agonist for chronic weight management. Under the terms of the agreement, YaoPharma will complete the ongoing Phase 1 clinical trial of YP05002 and grant Pfizer an exclusive global license to further develop, manufacture, and commercialize YP05002 worldwide. YaoPharma will receive an upfront payment of $150 million and is eligible for up to $1.935 billion in milestone payments.
Pfizer's frequent strategic moves in the GLP-1 field can be attributed to several key factors.
First, the GLP-1 class represents one of the fastest-growing segments in the global pharmaceutical landscape. According to a Goldman Sachs analysis report from May 2025, the global market size for GLP-1 drugs is projected to reach $95 billion by 2030 and could expand further to $120 billion by 2035. In what began as the "century-long battle of weight loss drugs" dominated by Novo Nordisk's semaglutide and Eli Lilly's tirzepatide, Pfizer has naturally chosen to participate, seizing this historic opportunity through rapid licensing deals.
Second, Pfizer faces an impending patent cliff. The company expects that between 2025 and 2030, blockbuster drugs including Eliquis and Ibrance will face patent expirations, representing an estimated revenue at risk of approximately $17 billion. In its 2026 guidance, Pfizer has already factored in approximately $1.5 billion in expected impacts from patent expirations. To offset the revenue erosion from patent expirations, Pfizer plans to initiate approximately 20 pivotal clinical trials in 2026, including 10 centered on obesity assets and 4 focused on PD-1×VEGF bispecific antibodies, with R&D resources concentrated on high-growth areas such as bispecifics and GLP-1.
Third, the Chinese market holds significant strategic importance. Currently, the obesity rate among Chinese adults stands at 14.1%, which is closely associated with metabolic diseases including hypertension, hyperlipidemia, and hyperglycemia. "Healthy weight management" has been officially incorporated into the Healthy China Initiative. By collaborating with Chinese pharmaceutical companies (such as Fosun Pharmaceutical's subsidiary YaoPharma and Sciwind Biosciences), Pfizer can leverage local R&D advantages to accelerate its long-term strategic expansion in the metabolic field, while rapidly accessing the Chinese market to meet the growing demand of domestic patients for innovative metabolic disease treatments, thereby enhancing its competitiveness in the Chinese market.
Furthermore, Pfizer's GLP-1 collaborations are not without strategic selectivity. Examining its recent partnerships reveals that Pfizer is pursuing differentiated breakthroughs in technological directions including ultra-long acting, oral formulations, and precision targeting, aiming to avoid direct competition with giants such as Novo Nordisk and Eli Lilly in traditional GLP-1 products.
In summary, Pfizer's frequent strategic deployments in the GLP-1 field reflect comprehensive considerations encompassing market potential, patent pressures, strategic development, and technological innovation. Through differentiated products and collaboration models, Pfizer aims to secure a significant position in this high-growth therapeutic area.
