Genturn Life completes tens of millions of RMB Angel round to pioneer a new paradigm in human aging reversal
Recently, Genturn Life, a company dedicated to the development of innovative regenerative and anti-aging therapeutics, announced the completion of a tens of millions of RMB angel funding round. This round was co-led by Casstar and Saizhi Bole. The funds will be allocated toward investigator-initiated trial (IIT) research for its core partial reprogramming pipeline and the development of its AI-powered molecular design platform. Previously, the U.S.-based company Life Biosciences announced that its first partial reprogramming therapeutic candidate, ER-100, has received Investigational New Drug (IND) clearance from the FDA. This milestone marks the formal entry of epigenetic reprogramming technology for age reversal into the clinical validation stage, signifying a paradigm shift in the understanding of aging from an "irreversible process" to a "programmable state."
1Core Technological Breakthrough: Redefining the "Safety Boundary" of Cellular Reprogramming
Partial reprogramming technology has demonstrated significant potential in the field of anti-aging, yet safety concerns remain a persistent "ghost" hindering its clinical translation. Conventional transcription factors are prone to triggering uncontrolled gene activation during reprogramming, a risk often rooted in their intrinsic physical properties.
In contrast, Genturn Life has chosen to reconstruct the molecular foundation. Leveraging its proprietary AI protein design platform, Genturn Life utilizes genetic algorithms to engineer the Yamanaka transcription factors by modulating their phase-separation functionality. This approach has led to the development of a transcription factor complex that retains only the initial-phase reprogramming capability—termed "Initiation-Phase Reprogramming" (IPR)—for developing in vivo partial reprogramming products aimed at reversing aging. Compared to similar products developed in the United States, Genturn Life's platform demonstrates enhanced safety and efficacy.
Genturn Life's technological edge lies in two key areas: stronger risk source control and more precise state monitoring. The former involves microscale structural modulation of transcription factors, effectively avoiding safety hazards associated with alterations in genomic spatial architecture and thereby reducing tumorigenic risk at the source. The latter, based on Genturn Life's proprietary IPR (Initiation-Phase Reprogramming) technology, successfully locks the reprogramming process within a "safe window" that efficiently rejuvenates cells without loss of cellular identity, enabling precise reversal of epigenetic age.
In-depth research into molecular mechanisms and continuous evolution of platform capabilities together form the core competitive moat of Genturn Life in the field of aging reversal.
2Pipeline Strategy: Advanced Intervention Targeting High-Barrier Markets
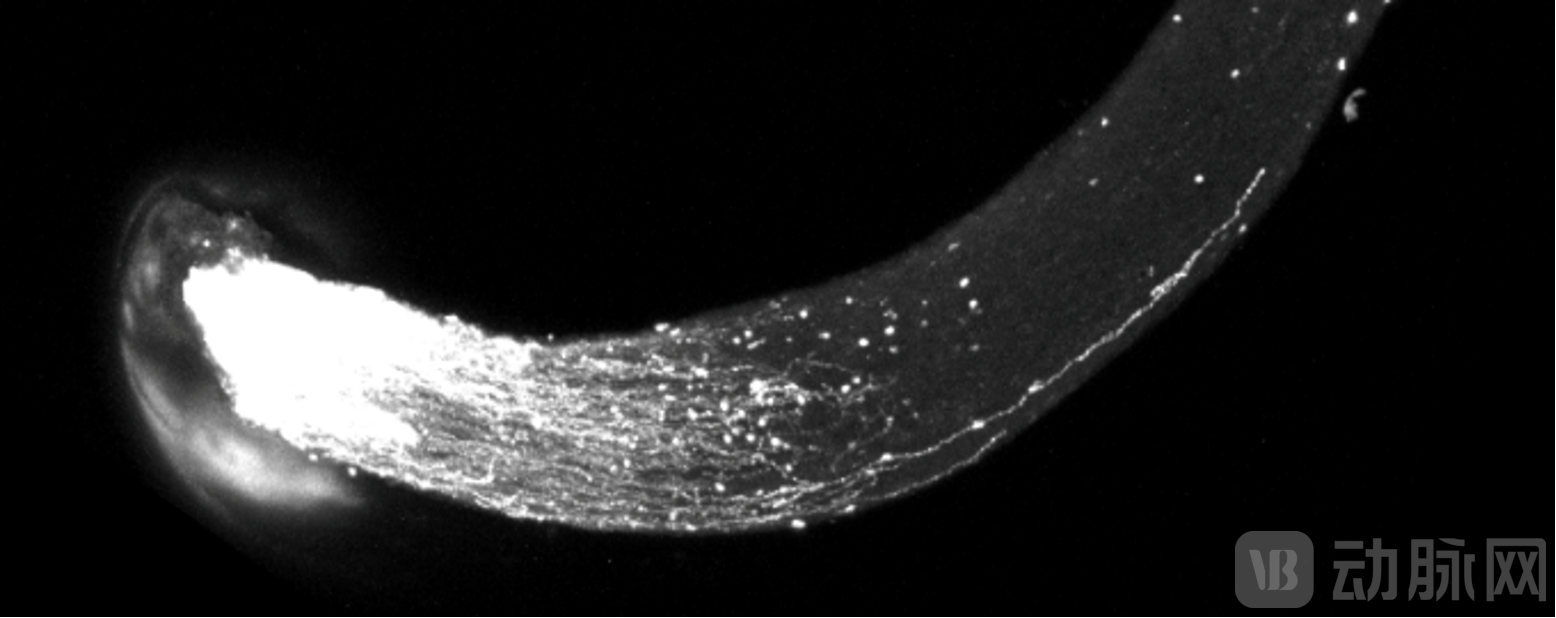
After Drug Administration, Severed Axons in the Mouse Optic Nerve Regenerated and Continued to Extend. (Image Source: Genturn Life)
Based on breakthroughs in this foundational technology, along with deep expertise in vector and delivery systems, Genturn Life has adopted a dual-driver strategy of "reprogramming repair and senescent cell clearance," building a portfolio of highly differentiated product pipelines:
JTL-1201a | Optic Nerve Repair and Axonal Regeneration: Targeting glaucoma and optic nerve injury, this candidate uses AAV to deliver IPR factor genes. In a market where no drugs currently exist to restore damaged retinal ganglion cells (RGCs), this program aims to achieve efficient neural repair and functional recovery through a single administration.
JTL-2201a | Reversal of Idiopathic Pulmonary Fibrosis (IPF): Combining lung-targeted LNPs with circular RNA technology, this candidate is designed to achieve synchronized rejuvenation across multiple alveolar cell types. Unlike conventional drugs that only slow disease progression, Genturn's approach seeks to reverse fibrosis at its root and restore respiratory function.
JTL-3202 | Exploration of Hepatic and Systemic Rejuvenation: Utilizing a unique in vivo CAR-T Senolytic platform, this program aims to precisely identify and eliminate senescent cells accumulated in key organs, enabling targeted or even systemic functional "age reversal."
3The Anti-Aging Industry: From Science Fiction to Reality
The pursuit of "immortality" has existed since ancient times, but it is only in the past decade that the scientific community has truly begun to decipher the underlying code for reversing aging. In 2012, Shinya Yamanaka was awarded the Nobel Prize for discovering that adult somatic cells could be reprogrammed into pluripotent stem cells. This breakthrough, akin to Prometheus' fire, ignited a new era in regenerative medicine. However, a significant gap remains between laboratory success and clinical application: while fully dedifferentiated cells can achieve "rejuvenation," they also carry an extremely high risk of tumorigenicity.
In 2016, the "partial reprogramming" technology developed by the Salk Institute demonstrated that epigenetic age could be reversed without altering cellular identity, marking a milestone breakthrough in the field. Since then, top global capital and technology giants have flocked to this space: from Jeff Bezos' USD 3 billion investment in Altos Labs to Sam Altman's USD 180 million backing of Retro Biosciences and other emerging players. Tens of billions of dollars are now pouring into the partial reprogramming anti-aging sector, transforming the fight against aging into a technological race driven by genomics, proteomics, and computational biology.
