Venturing into the deep end: Decoding Dupilumab's "systematic breakthrough" in pediatric indications
A new development has emerged in global pediatric immune-related drug therapies.
Recently, Japan's Ministry of Health, Labour and Welfare officially approved Dupilumab (Dupixent®) for the treatment of children aged 6 to 11 years with severe or refractory bronchial asthma whose symptoms are inadequately controlled by existing therapies. This marks the 50th country worldwide where Dupilumab has been approved for asthma in this age group.
Behind this approval lies Dupilumab's ongoing strategic focus on expanding its indications in pediatric and adolescent populations.
In fact, the pharmaceutical industry has long faced a structural challenge: the approval of pediatric indications for new drugs often lags significantly behind those for adults. This delay can sometimes extend to nearly 10 years, reflecting both the scientific complexities and regulatory caution involved in drug development for special populations.
However, as leading biopharmaceutical companies increasingly explore younger age segments, this landscape is gradually improving. Taking Dupilumab as an example, to date, the drug has been approved globally for a range of type 2 inflammation-related diseases in infants as young as 6 months, children, and adolescents. These include atopic dermatitis, asthma, eosinophilic esophagitis, chronic spontaneous urticaria, and chronic rhinosinusitis with nasal polyps (with approved age indications varying by market).
In the field of pediatric drug development—widely recognized as a scientifically and regulatorily demanding "deep-water zone"—how has Dupilumab consistently pushed the boundaries of age and disease, securing successive approvals through rigorous reviews? What underlying scientific rationale supports its commercial breakthroughs?
Challenge and Breakthrough: The Most Difficult Hurdle in Pediatric Indication Development
To understand the breakthrough significance of innovative therapies "reaching down" to younger age groups, one must first acknowledge the unique challenges in pediatric drug development.
For a long time, developing new drugs for immune-related diseases in children—especially infants and toddlers—has been akin to "walking a tightrope," facing multidimensional challenges in both science and regulation.
First, there are physiological challenges. A child's immune system is continuously developing and maturing, and its immune response patterns differ significantly from those of adults. The immune systems of infants aged six months to six years are not yet fully mature, making their responses to external stimuli more sensitive and unpredictable. This means that a drug's pharmacokinetic and pharmacodynamic profiles may differ entirely from those in adults. Immunomodulatory drugs could have long-term effects on a developing immune system, requiring longer follow-up periods and stricter monitoring for safety assessment.
Second, clinical trial design poses its own set of challenges. Disease presentation, severity assessment, and efficacy endpoints in children differ from those in adults. Researchers need to develop and validate age-appropriate disease assessment tools. Evaluating subjective symptoms such as itching or shortness of breath depends on age-related communication abilities. Growth and development indicators must be incorporated into safety assessments, and long-term follow-up is essential to evaluate impacts on growth. These special requirements make pediatric clinical trials far more complex than adult studies.
Moreover, regulatory standards for pediatric drugs are much stricter than those for adults. Sufficient adult data are typically required before pediatric studies can begin. Dose exploration must be more cautious and systematic. Safety databases demand larger sample sizes and longer follow-up periods. Different age subgroups may require independent studies. These stringent regulatory requirements extend development timelines and increase costs.
The above factors, combined with commercial cost-benefit considerations, have deterred many pharmaceutical companies from entering the younger-age market. This can create a non-virtuous cycle: a lack of adequate pediatric data leads to cautious regulatory approval, which in turn discourages further investment in pediatric R&D, ultimately limiting treatment options for children.
Yet, the clinical need for early intervention in pediatric immune diseases remains clear and urgent.
Take atopic dermatitis as an example. More than 85% of patients develop the condition before the age of five, and about 50% continue to experience symptoms into adulthood. Without timely and effective control, the disease can affect a child's sleep quality, learning ability, and social development, and may even set the stage for other type 2 inflammatory diseases later in life. The value of early intervention extends far beyond symptom relief: it helps improve atopic dermatitis symptoms while supporting normal growth, development, and learning opportunities, and reducing the risk of comorbidities.
Who will develop therapies for this "deep-water zone"? It requires leaders with both scientific vision and a long-term commitment to investment.
Scientific Foundation: Precisely Targeting the Core of Type 2 Inflammation
The confidence of Dupilumab in tackling the "deep waters of younger age groups" stems from a profound understanding of disease mechanisms.
Traditional systemic treatment approaches, such as corticosteroids and immunosuppressants, primarily rely on "broad-spectrum anti-inflammatory" effects to alleviate symptoms. This method, akin to "carpet bombing," often yields limited efficacy in young pediatric patients and raises significant safety concerns.
In contrast, the development of Dupilumab is grounded in a deep understanding of the type 2 inflammatory pathway. Extensive research has demonstrated that IL-4 (interleukin-4) and IL-13 (interleukin-13) serve as the "core drivers" of type 2 inflammation, exerting their effects through the shared IL-4Rα receptor and playing a critical role in the pathogenesis of various allergic and inflammatory diseases. Importantly, their role in pediatric type 2 inflammatory diseases is similar to that in adults. This provides a solid theoretical foundation for the application of Dupilumab in pediatric populations.
Dupilumab is a fully human monoclonal antibody targeting IL-4Rα. By blocking both the IL-4 and IL-13 signaling pathways, it precisely inhibits the type 2 inflammatory response.
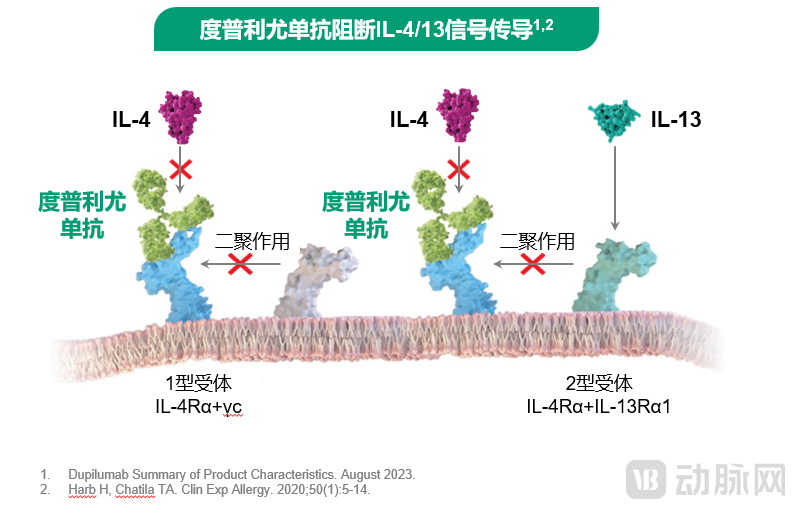 Figure: Dupilumab precisely blocks IL-4 and IL-13 signaling
Figure: Dupilumab precisely blocks IL-4 and IL-13 signaling
Specifically, Dupilumab selectively binds to the IL-4Rα receptor subunit without cross-reacting with other cytokine receptors, ensuring high specificity and precise targeting. Compared to broad-spectrum immunosuppressants, this approach significantly reduces the risk of widespread immune suppression.
Moreover, leveraging a fully human antibody technology platform, Dupilumab is composed of 100% fully human antibodies, which significantly lowers its immunogenicity and greatly reduces the risk of anti-drug antibody development. This characteristic not only ensures long-term medication safety but also provides a distinct advantage in pediatric populations, who often require extended treatment—offering stable efficacy and predictable safety.
From a scientific perspective, the application of Dupilumab in younger age groups demonstrates three key clinical benefits: First, by precisely targeting the type 2 inflammatory pathway, it minimizes the risk of broad immune suppression and reduces interference with the developing immune system in young children. Second, its low immunogenicity and well-defined mechanism provide a foundation for the long-term management of type 2 inflammatory diseases, avoiding fluctuations in efficacy that could disrupt growth and development. Third, by intervening in the atopic march, it holds the potential to reduce the risk of future comorbidities.
Expansion to Younger Ages: A Systematic and Progressive "Age-Decrement" Strategy
For the clinical development of indications targeting younger age groups, Dupilumab has adopted a scientifically rigorous "age-decrement" strategy. Its development pathway for atopic dermatitis in China precisely follows this systematic approach:
● In June 2020, Dupilumab was approved in China for moderate-to-severe atopic dermatitis in adults—the first step supported by comprehensive global clinical trial data.
● In September 2021, the indication was extended to adolescents aged 12 and above with moderate-to-severe atopic dermatitis, establishing a bridge from adults to adolescents.
● In February 2022, Dupilumab was approved for children aged 6 and above with moderate-to-severe atopic dermatitis, extending treatment to younger patient populations.
● In May 2023, Dupilumab was approved for infants and young children aged 6 months to 5 years with moderate-to-severe atopic dermatitis, making it the first biologic in China to cover atopic dermatitis patients across all age groups from 6 months and above.
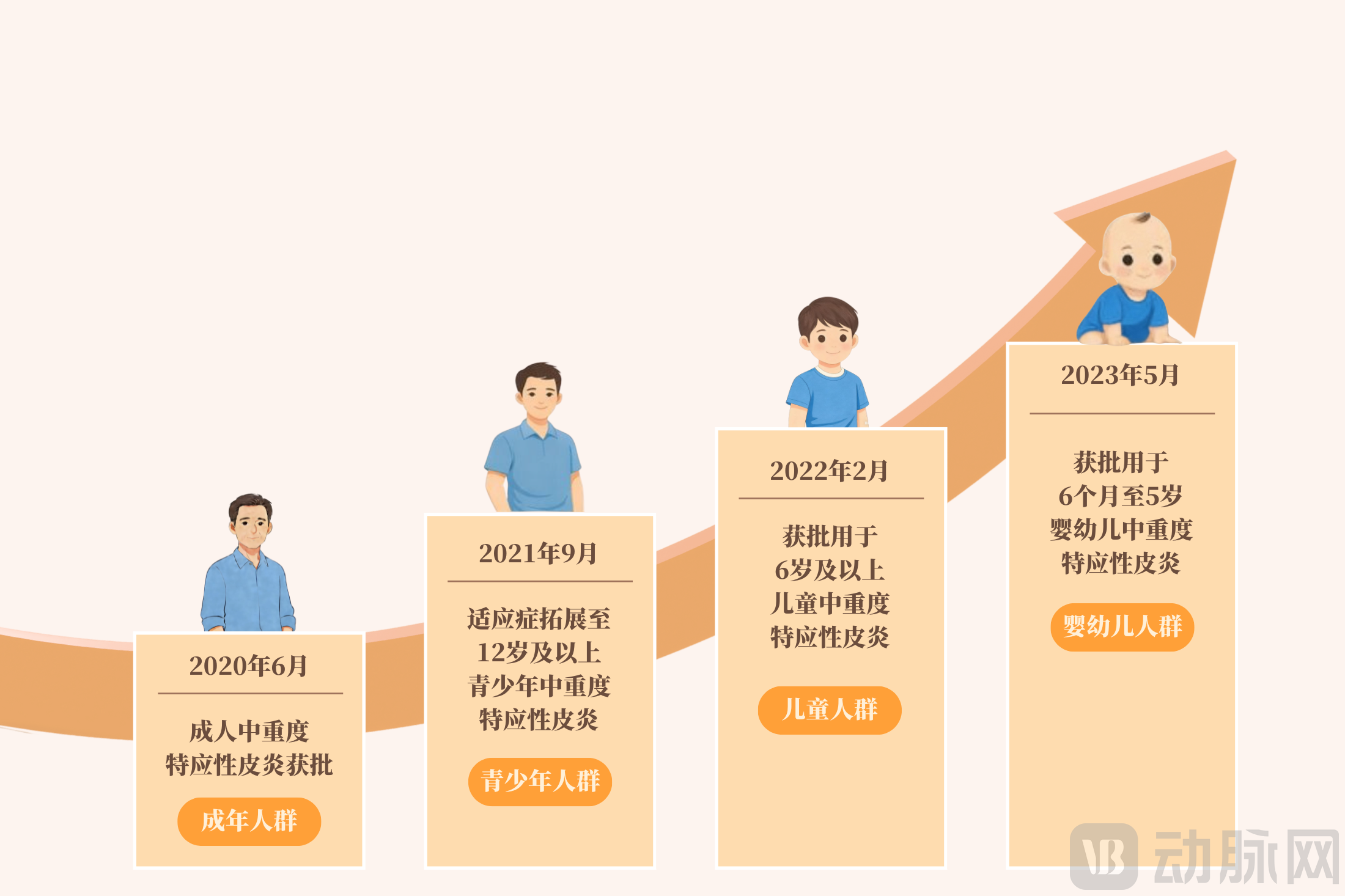
From adults to adolescents, and from children to infants, Dupilumab achieved full age-group coverage for atopic dermatitis in China in just three years. Each age-group expansion was based on robust safety and efficacy data from the previous age cohort, ensuring maximum protection for the safety of younger patients.
This rigorous scientific approach has also been carried forward into real-world studies conducted after the approval of the indications.
At the 2025 European Academy of Dermatology and Venereology Congress (EADV Congress 2025), the 24-week results of the ADOPED-STAD study were presented. ADOPED-STAD is a multicenter, prospective study conducted in China. As of the data cutoff date, 580 patients with atopic dermatitis (AD) were screened, of whom 563 were successfully enrolled (age distribution: 75 patients [13.3%] aged ≥6 months to <6 years, 366 patients [65.0%] aged ≥6 to <12 years, and 122 patients [21.7%] aged ≥12 to <18 years). This represents the largest real-world study of atopic dermatitis in children and adolescents conducted in China to date.
The study results validated the efficacy and safety of Dupilumab in real-world settings. At week 24, 75.6% of patients achieved EASI-75 (a 75% improvement in the Eczema Area and Severity Index), with attainment rates exceeding 70% across all age groups: 70.0% in the 6 months to 6 years group, 76.4% in the 6 to 12 years group, and 74.6% in the 12 to 18 years group. The affected body surface area decreased by an average of 27.5%, the itch score improved by an average of 4.5 points, the sleep disturbance score improved by 3.8 points, and the Children's Dermatology Life Quality Index improved by an average of 7.0 points.
More encouragingly, as Dupilumab treatment progressed, patients' dependence on topical medications significantly decreased. The usage rate of topical corticosteroids dropped from 69.1% at baseline to 43.9% at week 24, and the usage rate of topical antimicrobials decreased from 31.1% to 21.0%. This finding indicates that Dupilumab not only effectively controls disease symptoms but also reduces reliance on topical medications, thereby lowering potential risks.
The safety data are equally encouraging. Among the 563 pediatric patients, only 2.5% experienced serious adverse events, none of which were considered related to Dupilumab. The most common adverse events were fever and upper respiratory tract infections, consistent with previous clinical trial results.
The study also revealed disease characteristics across different age groups. Although children aged 6 months to 6 years had relatively lower baseline EASI scores and affected body surface areas, their itch scores and quality-of-life impacts were more severe. This suggests that when assessing the severity of atopic dermatitis in young children, clinicians should not only focus on the extent of skin lesions but also fully consider the impact of subjective symptoms such as itching on the patients. These findings provide clinicians with valuable age-stratified management strategies.
Conclusion
Pediatric drug development faces the dual challenges of scientific complexity and regulatory caution. The case of Dupilumab demonstrates that the "10-year lag" in new drug availability for children can be overcome through deep insights into disease mechanisms, a systematic "age-decrement" clinical strategy, and continuous accumulation of evidence-based data.
When innovation benefits children, the value of science finds its most profound expression.
